ASTM F3368-19
(Guide)Standard Guide for Cell Potency Assays for Cell Therapy and Tissue Engineered Products
Standard Guide for Cell Potency Assays for Cell Therapy and Tissue Engineered Products
SIGNIFICANCE AND USE
5.1 Cell Therapy Products may be used to treat clinical conditions, for example in regenerative medicine (e.g. type I diabetes, acute myocardial infarction, pediatric congenital heart disease, chronic ischemic heart failure, cancer, Crohn’s disease, chronic wound repair, nerve and spinal cord injury, musculoskeletal repair), and may be used for immunotherapy (e.g. graft versus host disease, CAR-T therapy).
5.2 Autologous, allogeneic, and xenogeneic cells may be used to make a product.
5.3 A product may be cells only, cells combined with an inert carrier, cells within an extracellular matrix, or cells within a synthetic scaffold, and will include tissue engineered medical products containing cells.
5.4 Cells may be gene-modified cells.
5.5 Cells may be adult or embryonic stem cells.
5.6 Cells may be minimally manipulated.
SCOPE
1.1 This guide is intended as a resource for individuals and organizations involved in the development, production, delivery, and regulation of cellular therapy products (CTPs) including genetically modified cells, tissue engineered medical products (TEMPs) and combination products where cell activity is a functional component of the final product.
1.2 This Guide was developed to include input derived from several previously published guidance documents and standards (section 2.4). It is the intent of this Guide is to reflect the current perspectives for CTP potency assays.
1.3 CTPs can provide therapy by localized or systemic treatment of a disease or pathology.
1.4 The products may provide a relatively short therapy, may be transient, or may be permanent and provide long-term therapy.
1.5 The products may be cells alone, cells combined with a carrier that is transient, or cells combined with a scaffold or other components that function in the overall therapy.
1.6 Potency assays may be in-vitro or in-vivo assays designed to determine the potency of a specific product. In-vivo assays are likely to be particularly useful to study the mechanism of action (MOA) of the therapy, but may not be desirable for final product quality control where they may be time-consuming and expensive, and where in-vitro assays may be preferable.
1.7 It is likely that multiple assays, and possibly both in-vitro and in-vivo assays, will be required to provide a broad measure of potency. However, in-vitro assays are likely to be preferred as release assays for products, and so studies to identify potency assays should emphasize in-vitro assays that are correlative or predictive of preclinical or clinical results.
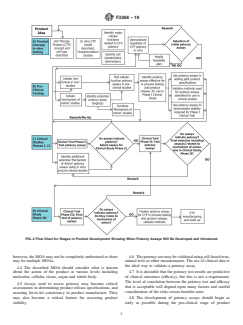
1.8 Potency assays should be developed during the product development cycle and therefore are likely to be more comprehensive at the end of that cycle compared to the beginning of product development and testing. It is recommended that potency assays be developed as early as possible in the product development cycle (Figs. 1 and 2).
FIG. 1 Progressive Implementation of Potency Assays
FIG. 2 Flow Chart for Stages in Product Development Showing When Potency Assays Will Be Developed and Introduced
1.9 Potency measurements are used as part of the testing for cell and cell-based products to demonstrate that product lots meet defined specifications when released for clinical use.
1.10 Shelf life specifications should be developed during the product development process to include potency measurements.
1.11 This standard guide is not intended to apply to drug or gene therapy products. However, genetically modified cell therapies, for example the chimeric antigen receptor-T (CAR-T) cell therapy, which the United States FDA classifies as gene therapy, are applicable.
1.12 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limit...
General Information
- Status
- Published
- Publication Date
- 14-May-2019
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.44 - Assessment for TEMPs
Relations
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Apr-2011
- Effective Date
- 01-Jun-2010
- Effective Date
- 01-Nov-2004
- Effective Date
- 10-Sep-2003
Overview
ASTM F3368-19 is the internationally recognized standard guide for cell potency assays used in cell therapy products (CTPs) and tissue engineered medical products (TEMPs). Published by ASTM International, this guide provides essential recommendations for the development, validation, and application of potency assays to ensure the consistency, quality, and efficacy of cellular therapy products. The standard supports a broad spectrum of regenerative medicine applications, including therapies for diabetes, cardiovascular diseases, cancer, wound repair, neurological conditions, and musculoskeletal disorders.
The standard applies to various types of cell therapy products, including those derived from autologous (self), allogeneic (donor), or xenogeneic (animal) sources, and covers products composed solely of cells or in combination with carriers, matrices, or scaffolds. It also encompasses gene-modified and stem cell-based therapies.
Key Topics
Potency Assay Fundamentals
- Definitions of potency, mechanism of action (MOA), and relevant terminology
- Importance of potency in demonstrating CTP efficacy and compliance with regulatory requirements
- Recommended types of assays: in-vitro (laboratory-based) and in-vivo (animal-based), with emphasis on in-vitro assays for ease and reproducibility
Assay Development and Validation
- Progressive implementation of potency assays throughout the product development cycle
- Necessity for phase-appropriate current Good Manufacturing Practice (cGMP) compliance
- Validation requirements: sensitivity, specificity, accuracy, and representation of the product’s MOA
- Use of quality management systems for reagents and protocols
Assay Selection and Implementation
- Utility of multiple complementary assays (assay matrix) to assess different aspects of CTP potency
- Considerations for surrogate assays, biomarkers, and functional tests that correlate with biological activity
- Importance of early assay development, starting from pre-clinical stages
Control and Quality Parameters
- Use of appropriate assay controls and well-characterized comparison materials
- Measures to ensure consistency, batch-to-batch reproducibility, and product shelf life
Applications
Product Development and Clinical Translation
- Guiding the development of effective potency assays, from early pre-clinical research through late-stage clinical trials
- Assisting in the establishment of product specifications for batch release, stability, and quality control
- Supporting regulatory submissions and compliance across different international jurisdictions
Therapeutic Areas
- Applicable to a wide range of regenerative medicine and advanced therapy medicinal products, including:
- Cardiovascular repair (myocardial infarction, heart failure)
- Diabetes (Type I)
- Oncology (CAR-T cancer therapies)
- Neurological injury (nerve and spinal cord injury)
- Musculoskeletal and wound repair
- Immunotherapy (graft versus host disease)
- Applicable to a wide range of regenerative medicine and advanced therapy medicinal products, including:
Manufacturing and Quality Assurance
- Ensuring potency assays are incorporated into process validation, product release testing, and ongoing quality assurance
- Addressing practical challenges such as limited batch sizes, time-to-release, and product variability
Related Standards
- ASTM F2312 – Terminology Relating to Tissue Engineered Medical Products
- ISO 17025 – General Requirements for the Competence of Testing and Calibration Laboratories
- FDA Guidance – Potency Tests for Cellular and Gene Therapy Products
- ICH Q2(R1) and Q6B – Validation of Analytical Procedures and Biotechnological Product Quality
- USP <1034> – Analysis of Biological Assays
- EMEA/CHMP Guidelines – Potency Testing for Cell-Based Medicinal Products
These standards and guidelines collectively provide a robust framework for the design, performance, and regulatory acceptance of potency assays in the field of cell therapy and tissue engineering.
ASTM F3368-19 is vital for ensuring that cell therapy and tissue engineered products meet stringent safety, quality, and efficacy expectations through reliable potency assessment methods, supporting industry growth and patient safety.
Buy Documents
ASTM F3368-19 - Standard Guide for Cell Potency Assays for Cell Therapy and Tissue Engineered Products
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F3368-19 is a guide published by ASTM International. Its full title is "Standard Guide for Cell Potency Assays for Cell Therapy and Tissue Engineered Products". This standard covers: SIGNIFICANCE AND USE 5.1 Cell Therapy Products may be used to treat clinical conditions, for example in regenerative medicine (e.g. type I diabetes, acute myocardial infarction, pediatric congenital heart disease, chronic ischemic heart failure, cancer, Crohn’s disease, chronic wound repair, nerve and spinal cord injury, musculoskeletal repair), and may be used for immunotherapy (e.g. graft versus host disease, CAR-T therapy). 5.2 Autologous, allogeneic, and xenogeneic cells may be used to make a product. 5.3 A product may be cells only, cells combined with an inert carrier, cells within an extracellular matrix, or cells within a synthetic scaffold, and will include tissue engineered medical products containing cells. 5.4 Cells may be gene-modified cells. 5.5 Cells may be adult or embryonic stem cells. 5.6 Cells may be minimally manipulated. SCOPE 1.1 This guide is intended as a resource for individuals and organizations involved in the development, production, delivery, and regulation of cellular therapy products (CTPs) including genetically modified cells, tissue engineered medical products (TEMPs) and combination products where cell activity is a functional component of the final product. 1.2 This Guide was developed to include input derived from several previously published guidance documents and standards (section 2.4). It is the intent of this Guide is to reflect the current perspectives for CTP potency assays. 1.3 CTPs can provide therapy by localized or systemic treatment of a disease or pathology. 1.4 The products may provide a relatively short therapy, may be transient, or may be permanent and provide long-term therapy. 1.5 The products may be cells alone, cells combined with a carrier that is transient, or cells combined with a scaffold or other components that function in the overall therapy. 1.6 Potency assays may be in-vitro or in-vivo assays designed to determine the potency of a specific product. In-vivo assays are likely to be particularly useful to study the mechanism of action (MOA) of the therapy, but may not be desirable for final product quality control where they may be time-consuming and expensive, and where in-vitro assays may be preferable. 1.7 It is likely that multiple assays, and possibly both in-vitro and in-vivo assays, will be required to provide a broad measure of potency. However, in-vitro assays are likely to be preferred as release assays for products, and so studies to identify potency assays should emphasize in-vitro assays that are correlative or predictive of preclinical or clinical results. 1.8 Potency assays should be developed during the product development cycle and therefore are likely to be more comprehensive at the end of that cycle compared to the beginning of product development and testing. It is recommended that potency assays be developed as early as possible in the product development cycle (Figs. 1 and 2). FIG. 1 Progressive Implementation of Potency Assays FIG. 2 Flow Chart for Stages in Product Development Showing When Potency Assays Will Be Developed and Introduced 1.9 Potency measurements are used as part of the testing for cell and cell-based products to demonstrate that product lots meet defined specifications when released for clinical use. 1.10 Shelf life specifications should be developed during the product development process to include potency measurements. 1.11 This standard guide is not intended to apply to drug or gene therapy products. However, genetically modified cell therapies, for example the chimeric antigen receptor-T (CAR-T) cell therapy, which the United States FDA classifies as gene therapy, are applicable. 1.12 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limit...
SIGNIFICANCE AND USE 5.1 Cell Therapy Products may be used to treat clinical conditions, for example in regenerative medicine (e.g. type I diabetes, acute myocardial infarction, pediatric congenital heart disease, chronic ischemic heart failure, cancer, Crohn’s disease, chronic wound repair, nerve and spinal cord injury, musculoskeletal repair), and may be used for immunotherapy (e.g. graft versus host disease, CAR-T therapy). 5.2 Autologous, allogeneic, and xenogeneic cells may be used to make a product. 5.3 A product may be cells only, cells combined with an inert carrier, cells within an extracellular matrix, or cells within a synthetic scaffold, and will include tissue engineered medical products containing cells. 5.4 Cells may be gene-modified cells. 5.5 Cells may be adult or embryonic stem cells. 5.6 Cells may be minimally manipulated. SCOPE 1.1 This guide is intended as a resource for individuals and organizations involved in the development, production, delivery, and regulation of cellular therapy products (CTPs) including genetically modified cells, tissue engineered medical products (TEMPs) and combination products where cell activity is a functional component of the final product. 1.2 This Guide was developed to include input derived from several previously published guidance documents and standards (section 2.4). It is the intent of this Guide is to reflect the current perspectives for CTP potency assays. 1.3 CTPs can provide therapy by localized or systemic treatment of a disease or pathology. 1.4 The products may provide a relatively short therapy, may be transient, or may be permanent and provide long-term therapy. 1.5 The products may be cells alone, cells combined with a carrier that is transient, or cells combined with a scaffold or other components that function in the overall therapy. 1.6 Potency assays may be in-vitro or in-vivo assays designed to determine the potency of a specific product. In-vivo assays are likely to be particularly useful to study the mechanism of action (MOA) of the therapy, but may not be desirable for final product quality control where they may be time-consuming and expensive, and where in-vitro assays may be preferable. 1.7 It is likely that multiple assays, and possibly both in-vitro and in-vivo assays, will be required to provide a broad measure of potency. However, in-vitro assays are likely to be preferred as release assays for products, and so studies to identify potency assays should emphasize in-vitro assays that are correlative or predictive of preclinical or clinical results. 1.8 Potency assays should be developed during the product development cycle and therefore are likely to be more comprehensive at the end of that cycle compared to the beginning of product development and testing. It is recommended that potency assays be developed as early as possible in the product development cycle (Figs. 1 and 2). FIG. 1 Progressive Implementation of Potency Assays FIG. 2 Flow Chart for Stages in Product Development Showing When Potency Assays Will Be Developed and Introduced 1.9 Potency measurements are used as part of the testing for cell and cell-based products to demonstrate that product lots meet defined specifications when released for clinical use. 1.10 Shelf life specifications should be developed during the product development process to include potency measurements. 1.11 This standard guide is not intended to apply to drug or gene therapy products. However, genetically modified cell therapies, for example the chimeric antigen receptor-T (CAR-T) cell therapy, which the United States FDA classifies as gene therapy, are applicable. 1.12 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limit...
ASTM F3368-19 is classified under the following ICS (International Classification for Standards) categories: 11.020.20 - Medical science. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F3368-19 has the following relationships with other standards: It is inter standard links to ASTM F2312-11(2020), ASTM F2312-11, ASTM F2312-10, ASTM F2312-04, ASTM F2312-03. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F3368-19 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F3368 − 19
Standard Guide for
Cell Potency Assays for Cell Therapy and Tissue
Engineered Products
This standard is issued under the fixed designation F3368; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope prehensive at the end of that cycle compared to the beginning
of product development and testing. It is recommended that
1.1 This guide is intended as a resource for individuals and
potency assays be developed as early as possible in the product
organizations involved in the development, production,
development cycle (Figs. 1 and 2).
delivery, and regulation of cellular therapy products (CTPs)
including genetically modified cells, tissue engineered medical 1.9 Potency measurements are used as part of the testing for
products (TEMPs) and combination products where cell activ- cell and cell-based products to demonstrate that product lots
ity is a functional component of the final product. meet defined specifications when released for clinical use.
1.2 This Guide was developed to include input derived from 1.10 Shelflifespecificationsshouldbedevelopedduringthe
several previously published guidance documents and stan- product development process to include potency measure-
dards (section 2.4). It is the intent of this Guide is to reflect the ments.
current perspectives for CTP potency assays.
1.11 This standard guide is not intended to apply to drug or
1.3 CTPs can provide therapy by localized or systemic gene therapy products. However, genetically modified cell
treatment of a disease or pathology. therapies, for example the chimeric antigen receptor-T
(CAR-T) cell therapy, which the United States FDA classifies
1.4 The products may provide a relatively short therapy,
as gene therapy, are applicable.
may be transient, or may be permanent and provide long-term
1.12 This standard does not purport to address all of the
therapy.
safety concerns, if any, associated with its use. It is the
1.5 The products may be cells alone, cells combined with a
responsibility of the user of this standard to establish appro-
carrier that is transient, or cells combined with a scaffold or
priate safety, health, and environmental practices and deter-
other components that function in the overall therapy.
mine the applicability of regulatory limitations prior to use.
1.6 Potency assays may be in-vitro or in-vivo assays de-
1.13 This international standard was developed in accor-
signed to determine the potency of a specific product. In-vivo
dance with internationally recognized principles on standard-
assays are likely to be particularly useful to study the mecha-
ization established in the Decision on Principles for the
nism of action (MOA) of the therapy, but may not be desirable
Development of International Standards, Guides and Recom-
for final product quality control where they may be time-
mendations issued by the World Trade Organization Technical
consuming and expensive, and where in-vitro assays may be
Barriers to Trade (TBT) Committee.
preferable.
2. Referenced Documents
1.7 It is likely that multiple assays, and possibly both
in-vitro and in-vivo assays, will be required to provide a broad
2.1 ASTM Standards:
measure of potency. However, in-vitro assays are likely to be
F2312 Terminology Relating to Tissue Engineered Medical
preferred as release assays for products, and so studies to
Products
identify potency assays should emphasize in-vitro assays that
2.2 ISO Document:
are correlative or predictive of preclinical or clinical results.
ISO 17025 General Requirements for the Competence of
1.8 Potency assays should be developed during the product
Testing and Calibration Laboratories
development cycle and therefore are likely to be more com-
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
This guide is under the jurisdiction of ASTM Committee F04 on Medical and contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Surgical Materials and Devices and is the direct responsibility of Subcommittee Standards volume information, refer to the standard’s Document Summary page on
F04.44 on Assessment for TEMPs. the ASTM website.
Current edition approved May 15, 2019. Published July 2019. DOI: 10.1520/ Available from American National Standards Institute (ANSI), 25 W. 43rd St.,
F3368-19 4th Floor, New York, NY 10036, http://www.ansi.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F3368 − 19
FIG. 1 Progressive Implementation of Potency Assays
2.3 U.S. Regulations: 3.2.3 potency, n—may also be the quantitative measure of
21 CFR 600.3(s) biological activity based on the attribute of the product, which
21 CFR 3.2(k) is linked to the relevant biological properties and functions.
21 CFR 1271 Current Good Manufacturing Practice for The assay demonstrating the biological activity should be
Human Cells, Tissues, and Cellular and Tissue-Based based on the intended biological effect that should ideally be
Products related to the expected clinical response. (ICH Q6B)
2.4 Other Documents:
3.2.4 strength, n—the potency, or therapeutic activity of the
FDA Guidance for Industry: Potency Tests for Cellular and
product.
Gene Therapy Products. January 2011
3.2.5 tests for potency, n—either in-vitro or in-vivo tests, or
Europeans Medicines Agency, Committee for Medicinal
both, which have been specifically designed for each product
Product for Human Use, Guideline on human cell-based
so as to indicate its potency in a manner adequate to satisfy the
medicinal products. Doc. Ref. EMEA/CHMP/410869/
interpretation of potency given by the definition in 21 CFR
2006, 2008
600.3(s).
ICH Q2(R1) Validation of Analytical Procedures: Text and
Methodology
3.2.6 validation, n—the confirmation by examination and
ICH Q6B Note for guidance on specifications: test proce-
the provision of objective evidence that the particular require-
dures and acceptance criteria for biotechnological/
ments for a specific intended use are fulfilled ISO 17025:2005
biological products (CPMP/ICH/365/96)
(5.4.5.1).
Points to Consider in the Manufacture and Testing of
Monoclonal Antibody Products for Human Use FDA
4. Summary of Guide
CBER, February 1997
4.1 It is the intent of this guide to provide a compendium of
EMEA/CHMP Guideline on potency testing of cell-based
information that may be related to the potency assays used in
immunotherapy medicinal products for the treatment of
the development and post-approval use of CTPs where activi-
cancer (CHMP/BWP/271475/06 rev.1)
ty(s) of the cell are related to function of the product. It is
USP 1034 Analysis of Biological Assays
expected that for many products multiple assays will be
3. Terminology required to provide a comprehensive assessment of potency. It
is also possible that the potency assays for a particular product
3.1 Unless provided otherwise in 3.2, terminology shall be
will continue to develop through the product development
in conformance with Terminology F2312.
cycle.
3.2 Definitions of Terms Specific to This Standard:
4.2 CTPs are complex and their therapeutic effectiveness is
3.2.1 mechanism of action (MOA), n—the means by which
dependent on multiple features and characteristics of the cell,
a product achieves its intended therapeutic effect or action, as
and it is likely that these can only be assessed using multiple
defined in 21 CFR 3.2(k).
assays. These assessments preferably will be related to mecha-
3.2.2 potency, n—the specific ability or capacity of the
nism of action.
product, as demonstrated by appropriate laboratory tests or by
adequatelycontrolledclinicaldataobtainedthroughtheadmin- 4.3 It is important to note that a potency assay is not a
istrationoftheproductinthemannerintended,toeffectagiven measure of clinical efficacy. The only way to measure clinical
result. (CFR 600.3(s)) efficacy in humans is through a human clinical trial. Potency
assays evaluate the article’s biological activity based on the
hypothesized MOA (see 3.2.1 and 3.2.2). Ideally, potency
Available from U.S. Government Printing Office, Superintendent of
assaysareameasurementofthebiologicalactivityofaproduct
Documents, 732 N. Capitol St., NW, Washington, DC 20401-0001, http://
www.access.gpo.gov. based on the hypothesized MOA (see 3.2.1 and 3.2.2);
F3368 − 19
FIG. 2 Flow Chart for Stages in Product Development Showing When Potency Assays Will Be Developed and Introduced
however, the MOAmay not be completely understood or there 4.6 Thepotencytestmaybevalidatedusingcell-basedtests,
may be multiple MOAs. animal tests or other measurements. The use of clinical data is
the ideal way to validate a potency assay.
4.4 The described MOA should consider what is known
about the action of the product at various levels including 4.7 It is desirable that the potency test results are predictive
molecular, cellular, tissue, organ and whole body. of clinical outcomes (efficacy), but this is not a requirement.
The level of correlation between the potency test and efficacy
4.5 Assays used to assess potency may become critical
that is acceptable will depend upon many factors and careful
assessments in determining product release specifications, and
consideration of the risks-versus-benefits ratio.
ensuring lot-to-lot consistency in product manufacture. They
may also become a critical feature for assessing product 4.8 The development of potency assays should begin as
stability. early as possible during the pre-clinical stage of product
F3368 − 19
development. It is desirable for the potency assays to be complex.Thereforeitisimportanttogenerateacomprehensive
predictiveofclinicalefficacyandsothismayrequirecontinued set of characterization data for these products. These data may
development throughout the product development cycle. or may not be used in the final potency assays but will guide
the identification of potential potency assays at the early stages
4.9 Potency assays should comply with phase-appropriate
of product development as well as final selection of potency
current Good Manufacturing Practice (cGMP) regulations, and
assays for manufacture and release.
should be validated to demonstrate suitability for use.
6.2 Product characterization data should be derived from a
4.10 Potency assay reagent variability may affect assay
wide range of CTP attributes that may include physical,
performance, and so reagents should be managed under an
phenotypic, molecular, biochemical, immunologic, and bio-
appropriate quality management system to minimize changes
logic properties. These data may be gathered during specific
and lot-to-lot variability, to ensure reliable and consistent assay
in-vitro characterization studies, pre-clinical animal studies,
performance.
and clinical studies. They may include those types of charac-
4.11 Potency assays are a regulatory requirement for the
teristics that are commonly used for lot release for CTPs, but
release of cell and cell-based products, It is the intent of this
should also include characteristics that will allow investigators
guide to provide information appropriate for multiple interna-
to identify product features that may best correlate with
tional regulatory agencies.
potency. This broad understanding of the CTP may help
4.12 Throughout this guide, the reader is referred to other
identify potency assays used early in the product development
documents that may provide specific information that can be
cycle, and may help improve or optimize the potency assay
applied in the manufacture and testing of cell-containing
selection as the product moves closer to marketing.
products. Most of the potentially applicable position papers
6.3 Product characterization may include assays and assess-
and guidance documents from many regions of the world can
ments that are not appropriate for setting specification for lot
be accessed via the internet. New documents are continually
release; however they will be informative in helping select
produced.
product characteristics that are related to biological activity
4.13 The application of this guide does not guarantee
and/or clinical efficacy.
clinical success of a finished product. This guide may help to
6.4 Potency assays may be used in conjunction with product
ensure consistency in the properties and characterization of a
given cell-containing product developed for the purpose of characterization assays in process verification and validation,
or qualification of proposed process changes.
clinical therapy.
4.14 This guide does not suggest that all the listed tests be
7. Measurement of Potency
conducted. The decision regarding applicability or suitability
of any particular test method remains the responsibility of the
7.1 Desired characteristics of potency assays include that
supplier, user, or regulator of the material based on applicable
they should be accurate, representative of mechanism(s) of
regulations, characterizations, and preclinical/clinical testing.
action, and predictive of clinical efficacy. They should (1)
ideally provide quantitative results allowing product release
5. Significance and Use
per acceptance criteria, and (2) should be sensitive and specific
5.1 Cell Therapy Products may be used to treat clinical
enough to detect changes relevant to potency (including
conditions, for example in regenerative medicine (e.g. type I
degradation, subpotent material). Where appropriate, potency
diabetes, acute myocardial infarction, pediatric congenital
assays may also be used in quantifying batch-to-batch consis-
heart disease, chronic ischemic heart failure, cancer, Crohn’s
tency.
disease, chronic wound repair, nerve and spinal cord injury,
7.2 Potency assays may be qualitative, but ideally should be
musculoskeletal repair), and may be used for immunotherapy
linked to a quantitative analytical assay.
(e.g. graft versus host disease, CAR-T therapy).
5.2 Autologous, allogeneic, and xenogeneic cells may be 7.3 Potency assays may be in-vitro assays or in-vivo assays.
used to make a product.
7.4 Apotency test may include measurement of a biological
5.3 A product may be cells only, cells combined with an
action of the test article, such as release of a soluble factor into
inert carrier, cells within an extracellular matrix, or cells within
the medium by the test article, or the generation of an
a synthet
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...