ASTM F2312-11(2020)
(Terminology)Standard Terminology Relating to Tissue Engineered Medical Products
Standard Terminology Relating to Tissue Engineered Medical Products
SIGNIFICANCE AND USE
3.1 The need for standards regarding TEMPs has also prompted a need for definitions. This terminology sets forth definitions of the most commonly used terms and specifies the relationship among the sciences and components applied in tissue engineering to develop TEMPs. Use of these terms and an understanding of these relationships will unify the ASTM TEMPs standards with a common language such that the users of these standards can understand and interpret the standards more precisely. Terms specific to a TEMP standard will also be defined within the respective standard as appropriate.
3.2 Defining Terms—Terms are defined with a broad scope to encompass these new products known as TEMPs. For instance, the definition for somatic cell therapy as stated in the “Guidance for Human Somatic Cell Therapy and Gene Therapy” (1)3 is recognized in this terminology. However, for the purposes of TEMPs that contain cells, we have added the definition of “cell” which is much broader and not limited to the use of living cells.
3.3 Clinical Effects of TEMPs—The users of this terminology should note that terms used regarding the clinical effects of TEMPs, for instance, “modify or modification” of the patient's condition, may also be interpreted to “enhance, augment, transform, alter, improve, or supplement.” Similarly, “repair” may also serve to mean “restore.”
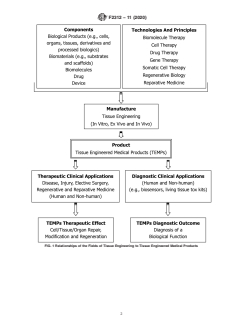
3.4 The diagram in Fig. 1 shows the relationships of components of TEMPs and of the fields of science (for example, technologies and principles) used in tissue engineering to create TEMPs. Certain TEMPs may be tissue engineered or produced in vitro by using specific components and sciences to create an off-the-shelf TEMP for the users. Other TEMPs may by design require the users to place the components inside the patient, (that is, in vivo) to rely upon the patient's regenerative potential to achieve the product's primary intended purpose. The expectation of a TEMP used for therapeutic clinical applications is to have ...
SCOPE
1.1 This terminology defines basic terms and presents the relationships of the scientific fields related to Tissue Engineered Medical Products (TEMPs). Committee F04 has defined these terms for the specific purpose of unifying the language used in standards for TEMPs.
1.2 The terms and relationships defined here are limited to TEMPs. They do not apply to any medical products of human origin regulated by the U.S. Food and Drug Administration under 21 CFR Parts 16 and 1270 and 21 CFR Parts 207, 807, and 1271.
1.3 The terms and nomenclature presented in this standard are for the specific purposes of unifying the language used in TEMP standards and are not intended for labeling of regulated medical products.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Jan-2020
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.41 - Classification and Terminology for TEMPs
Relations
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Feb-2020
Overview
ASTM F2312-11(2020): Standard Terminology Relating to Tissue Engineered Medical Products provides a unified vocabulary for the rapidly evolving field of tissue engineered medical products (TEMPs). Developed by ASTM Committee F04, this standard establishes precise definitions for terms commonly used in tissue engineering, clarifying the relationships among biological, chemical, and engineering components employed to develop TEMPs. By standardizing terminology, ASTM F2312 helps create a common language that supports accurate communication, interpretation, and implementation of other TEMP-related standards worldwide.
Key Topics
- Unified Terminology: Defines a broad set of terms covering the essential sciences, materials, and processes in tissue engineering, including definitions for cells, biomaterials, scaffolds, somatic and gene therapies, and various clinical applications such as “repair,” “regenerate,” and “modify.”
- Scope & Application: Focuses specifically on tissue engineered medical products, excluding certain human-origin medical products regulated under specific US FDA regulations (21 CFR Parts 16, 1270, 207, 807, 1271).
- Component Relationships: Clarifies the interconnectedness of scientific fields such as regenerative biology, materials science, clinical medicine, and engineering within TEMP development.
- Clinical Concepts: Expands key clinical terms to capture a wide array of therapeutic effects, e.g., “modify” may mean enhance, supplement, or restore function, while “repair” aligns with “restore.”
- Comprehensive Definitions: Offers broad definitions, such as “cell” encompassing living and non-living, modified, or unmodified sources, to account for all potential TEMP component variations.
- Organ and Tissue Systems: Identifies major body systems (e.g., cardiovascular, musculoskeletal, integumentary) to contextualize TEMP applications.
Applications
ASTM F2312 serves as a foundational resource for professionals in the tissue engineering and regenerative medicine sectors:
- Standard Development: Used as a reference for drafting and harmonizing other ASTM standards related to TEMPs, ensuring consistency.
- Product Research & Development: Guides manufacturers, engineers, and researchers in using agreed definitions, promoting clearer communication and documentation for innovative TEMP products and technologies.
- Regulatory and Quality Assurance: Aids regulatory affairs specialists and quality managers in interpreting TEMP standards, facilitating compliance, and supporting accurate assessment and comparison of new devices or therapeutics.
- Clinical Settings: Benefits clinicians and healthcare providers by ensuring the language used in clinical studies, product documentation, and training materials aligns with international standards, reducing ambiguity and enhancing patient safety.
- Education & Training: Serves as an educational tool for students and trainees entering the tissue engineering field, offering clarity and consistency in terminology.
Related Standards
- ASTM F2603 – Standard Guide for Interpreting Cellular Responses to Biomaterials: Provides further guidance on biological response assessment for prospective TEMP components.
- ASTM F2450 – Standard Guide for Assessing Microstructure of Polymeric Scaffolds Used in TEMPs: Details methods for evaluating scaffold properties in tissue-engineered constructs.
- ASTM F2027 – Standard Guide for Characterization and Testing of Hydrogels for TEMPs: Related to terminology concerning hydrogels outlined in F2312.
- ISO 10993 Series: International standards for biological evaluation of medical devices, overlapping in the definitions of biocompatibility and bioactive agents.
- 21 CFR Parts 1270 & 1271: US regulations referenced for exclusion of certain human-origin products from this terminology standard.
Keywords: tissue engineered medical products, TEMPs, tissue engineering, regenerative medicine, ASTM F2312, standard terminology, biomaterials, cell therapy, gene therapy, clinical applications, unified language, medical device standards.
Buy Documents
ASTM F2312-11(2020) - Standard Terminology Relating to Tissue Engineered Medical Products
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F2312-11(2020) is a standard published by ASTM International. Its full title is "Standard Terminology Relating to Tissue Engineered Medical Products". This standard covers: SIGNIFICANCE AND USE 3.1 The need for standards regarding TEMPs has also prompted a need for definitions. This terminology sets forth definitions of the most commonly used terms and specifies the relationship among the sciences and components applied in tissue engineering to develop TEMPs. Use of these terms and an understanding of these relationships will unify the ASTM TEMPs standards with a common language such that the users of these standards can understand and interpret the standards more precisely. Terms specific to a TEMP standard will also be defined within the respective standard as appropriate. 3.2 Defining Terms—Terms are defined with a broad scope to encompass these new products known as TEMPs. For instance, the definition for somatic cell therapy as stated in the “Guidance for Human Somatic Cell Therapy and Gene Therapy” (1)3 is recognized in this terminology. However, for the purposes of TEMPs that contain cells, we have added the definition of “cell” which is much broader and not limited to the use of living cells. 3.3 Clinical Effects of TEMPs—The users of this terminology should note that terms used regarding the clinical effects of TEMPs, for instance, “modify or modification” of the patient's condition, may also be interpreted to “enhance, augment, transform, alter, improve, or supplement.” Similarly, “repair” may also serve to mean “restore.” 3.4 The diagram in Fig. 1 shows the relationships of components of TEMPs and of the fields of science (for example, technologies and principles) used in tissue engineering to create TEMPs. Certain TEMPs may be tissue engineered or produced in vitro by using specific components and sciences to create an off-the-shelf TEMP for the users. Other TEMPs may by design require the users to place the components inside the patient, (that is, in vivo) to rely upon the patient's regenerative potential to achieve the product's primary intended purpose. The expectation of a TEMP used for therapeutic clinical applications is to have ... SCOPE 1.1 This terminology defines basic terms and presents the relationships of the scientific fields related to Tissue Engineered Medical Products (TEMPs). Committee F04 has defined these terms for the specific purpose of unifying the language used in standards for TEMPs. 1.2 The terms and relationships defined here are limited to TEMPs. They do not apply to any medical products of human origin regulated by the U.S. Food and Drug Administration under 21 CFR Parts 16 and 1270 and 21 CFR Parts 207, 807, and 1271. 1.3 The terms and nomenclature presented in this standard are for the specific purposes of unifying the language used in TEMP standards and are not intended for labeling of regulated medical products. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 3.1 The need for standards regarding TEMPs has also prompted a need for definitions. This terminology sets forth definitions of the most commonly used terms and specifies the relationship among the sciences and components applied in tissue engineering to develop TEMPs. Use of these terms and an understanding of these relationships will unify the ASTM TEMPs standards with a common language such that the users of these standards can understand and interpret the standards more precisely. Terms specific to a TEMP standard will also be defined within the respective standard as appropriate. 3.2 Defining Terms—Terms are defined with a broad scope to encompass these new products known as TEMPs. For instance, the definition for somatic cell therapy as stated in the “Guidance for Human Somatic Cell Therapy and Gene Therapy” (1)3 is recognized in this terminology. However, for the purposes of TEMPs that contain cells, we have added the definition of “cell” which is much broader and not limited to the use of living cells. 3.3 Clinical Effects of TEMPs—The users of this terminology should note that terms used regarding the clinical effects of TEMPs, for instance, “modify or modification” of the patient's condition, may also be interpreted to “enhance, augment, transform, alter, improve, or supplement.” Similarly, “repair” may also serve to mean “restore.” 3.4 The diagram in Fig. 1 shows the relationships of components of TEMPs and of the fields of science (for example, technologies and principles) used in tissue engineering to create TEMPs. Certain TEMPs may be tissue engineered or produced in vitro by using specific components and sciences to create an off-the-shelf TEMP for the users. Other TEMPs may by design require the users to place the components inside the patient, (that is, in vivo) to rely upon the patient's regenerative potential to achieve the product's primary intended purpose. The expectation of a TEMP used for therapeutic clinical applications is to have ... SCOPE 1.1 This terminology defines basic terms and presents the relationships of the scientific fields related to Tissue Engineered Medical Products (TEMPs). Committee F04 has defined these terms for the specific purpose of unifying the language used in standards for TEMPs. 1.2 The terms and relationships defined here are limited to TEMPs. They do not apply to any medical products of human origin regulated by the U.S. Food and Drug Administration under 21 CFR Parts 16 and 1270 and 21 CFR Parts 207, 807, and 1271. 1.3 The terms and nomenclature presented in this standard are for the specific purposes of unifying the language used in TEMP standards and are not intended for labeling of regulated medical products. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F2312-11(2020) is classified under the following ICS (International Classification for Standards) categories: 01.040.11 - Health care technology (Vocabularies); 11.120.99 - Other standards related to pharmaceutics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F2312-11(2020) has the following relationships with other standards: It is inter standard links to ASTM F2312-11, ASTM F3163-22, ASTM F3369-19e1, ASTM F3224-17, ASTM F3209-16, ASTM F3504-21, ASTM F3223-17, ASTM F2997-21, ASTM F3106-22, ASTM F2150-19, ASTM F3274-21, ASTM F2664-19e1, ASTM F3368-19, ASTM F3142-16, ASTM F3225-17(2022). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F2312-11(2020) is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F2312 − 11 (Reapproved 2020)
Standard Terminology Relating to
Tissue Engineered Medical Products
This standard is issued under the fixed designation F2312; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 3. Significance and Use
1.1 This terminology defines basic terms and presents the 3.1 The need for standards regarding TEMPs has also
relationships of the scientific fields related to Tissue Engi- prompted a need for definitions. This terminology sets forth
neered Medical Products (TEMPs). Committee F04 has de- definitions of the most commonly used terms and specifies the
fined these terms for the specific purpose of unifying the relationship among the sciences and components applied in
language used in standards for TEMPs. tissue engineering to develop TEMPs. Use of these terms and
an understanding of these relationships will unify the ASTM
1.2 The terms and relationships defined here are limited to
TEMPs standards with a common language such that the users
TEMPs. They do not apply to any medical products of human
of these standards can understand and interpret the standards
origin regulated by the U.S. Food and Drug Administration
more precisely.Terms specific to aTEMPstandard will also be
under 21 CFR Parts 16 and 1270 and 21 CFR Parts 207, 807,
defined within the respective standard as appropriate.
and 1271.
3.2 Defining Terms—Terms are defined with a broad scope
1.3 The terms and nomenclature presented in this standard
to encompass these new products known as TEMPs. For
are for the specific purposes of unifying the language used in
instance, the definition for somatic cell therapy as stated in the
TEMPstandards and are not intended for labeling of regulated
“Guidance for Human Somatic Cell Therapy and Gene
medical products.
Therapy” (1) is recognized in this terminology. However, for
1.4 This standard does not purport to address all of the
the purposes of TEMPs that contain cells, we have added the
safety concerns, if any, associated with its use. It is the
definition of “cell” which is much broader and not limited to
responsibility of the user of this standard to establish appro-
the use of living cells.
priate safety, health, and environmental practices and deter-
3.3 Clinical Effects of TEMPs—The users of this terminol-
mine the applicability of regulatory limitations prior to use.
ogyshouldnotethattermsusedregardingtheclinicaleffectsof
1.5 This international standard was developed in accor-
TEMPs, for instance, “modify or modification” of the patient’s
dance with internationally recognized principles on standard-
condition, may also be interpreted to “enhance, augment,
ization established in the Decision on Principles for the
transform, alter, improve, or supplement.” Similarly, “repair”
Development of International Standards, Guides and Recom-
may also serve to mean “restore.”
mendations issued by the World Trade Organization Technical
Barriers to Trade (TBT) Committee.
3.4 The diagram in Fig. 1 shows the relationships of
components of TEMPs and of the fields of science (for
2. Referenced Documents
example, technologies and principles) used in tissue engineer-
ing to createTEMPs. CertainTEMPs may be tissue engineered
2.1 Government Documents:
or produced in vitro by using specific components and sciences
21 CFR Parts 16 and 1270 Human Tissues, Intended for
to create an off-the-shelf TEMP for the users. Other TEMPs
Transplantation (July 29, 1997)
may by design require the users to place the components inside
21 CFR Parts 207, 807, and 1271 Human Cells,Tissues, and
the patient, (that is, in vivo) to rely upon the patient’s
Cellular and Tissue-Based Products; Establishment Reg-
regenerative potential to achieve the product’s primary in-
istration and Listing (January 19, 2001)
tended purpose. The expectation of a TEMPused for therapeu-
tic clinical applications is to have a therapeutic effect, specifi-
ThisterminologyisunderthejurisdictionofASTMCommitteeF04onMedical
cally to repair, modify or regenerate the recipient’s cells,
and Surgical Materials and Devicesand is the direct responsibility of Subcommittee
tissues, and organs or their structure and function. Such a
F04.41 on Classification and Terminology for TEMPs.
Current edition approved Feb. 1, 2020. Published April 2020. Originally TEMPmay be used for human and non-human applications. In
approved in 2003. Last previous edition approved in 2011 as F2312 – 11. DOI:
10.1520/F2312-11R20.
AvailablefromU.S.GovernmentPrintingOfficeSuperintendentofDocuments,
732 N. Capitol St., NW, Mail Stop: SDE, Washington, DC 20401, http:// The boldface numbers in parentheses refer to the list of references at the end of
www.access.gpo.gov. thisstandard.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2312 − 11 (2020)
FIG. 1 Relationships of the Fields of Tissue Engineering to Tissue Engineered Medical Products
F2312 − 11 (2020)
other applications, a TEMP may be used in diagnostic clinical biomaterial, n—anysubstance(otherthanadrug),syntheticor
applications, or both, to achieve an investigative outcome of natural, that can be used as a system or part of a system that
the function of the cells, tissues, and organs. treats, augments, or replaces any tissue, organ, or function of
the body.
4. Terminology
biomolecule, n—a biologically active peptide, protein,
adventitious agents, n—an unintentionally introduced micro- carbohydrate,vitamin,lipid,ornucleicacidproducedbyand
purified from naturally occurring or recombinant organisms,
biological or other infectious contaminant. In the production
of TEMPs, these agents may be unintentionally introduced tissues or cell lines or synthetic analogs of such molecules.
A biomolecule may be used as a component of a TEMP.
into the process stream or the final product, or both.
alginate, n—polysaccharide obtained from some of the more biomolecule therapy, n—the use of biomolecules to repair,
modify, or regenerate the recipient’s cells, tissues, or organs
common species of marine algae, consisting of an insoluble
mix of calcium, magnesium, sodium, and potassium salts. or their structure and function, or both. Biomolecule therapy
DISCUSSION—Alginate exists in brown algae as its most abundant
technologies can be applied in tissue engineering to generate
polysaccharide, mainly occurring in the cell walls and intercellular
TEMPs.
spaces of brown seaweed and kelp. Alginate’s main function is to
contribute to the strength and flexibility of the seaweed plant.Alginate cell, n—“the smallest structural unit of an organism that is
is classified as a hydrocolloid. The most commonly used alginate is
capable of independent functioning, consisting of one or
sodium alginate. Sodium alginate and, in particular, calcium cross-
more nuclei, cytoplasm, and various organelles, all sur-
linked alginate gels are used in Tissue Engineered Medical Products
rounded by a semipermeable cell membrane” (4).
(TEMPs)asbiomedicalmatrices,controlleddrugdeliverysystems,and
DISCUSSION—Cells are highly variable and specialized in both struc-
for immobilizing living cells.
tureandfunction,thoughallmustatsomestagesynthesizeproteinsand
nucleic acids, use energy, and reproduce.Acell or cells may be of any
allogeneic or allogenic, adj—cells, tissues, and organs in
origin (that is, organism), tissue type, developmental stage, and may be
which the donor and recipient are genetically different
living, non-living, and genetically or otherwise modified. Cells may be
individuals of the same species. Synonyms: allograft and
used as a component of a TEMP.
homograft.
cell culture, n—the in vitro growth or maintenance of cells.
allograft, n—a graft of tissue between individuals of the same
cell therapy, n—the administration of cells (any kind and
species but of disparate genotype. Called also allogeneic
form) to repair, modify or regenerate the recipient’s cells,
graft and homograft.
tissues, and organs or their structure and function, or both.
APA bead, n—alginate-poly-L-lysine-alginate bead.
Cell therapy technologies can be applied in tissue engineer-
ing to generate TEMPs.
autograft, n—a graft of tissue derived from another site in or
on the body of the organism receiving it.
channelyzer, n—a pulse height analyzer; places voltage pulses
into appropriate size bins for the size distribution data.
autologous, adj—cells, tissues, and organs in which the donor
and recipient is the same individual. Synonyms: autogenous,
chitosan, n—a linear polysaccharide consisting of β(1→4)
autograft,or autotransfusion,a self-to-self graft.
linked 2-acetamido-2-deoxy-D-glucopyranose (GlcNAc)
and 2-amino-2-deoxy-D-glucopyranose (GlcN). Chitosan is
bioactive agents, n—any molecular component in, on, or with
a polysaccharide derived by N-deacetylation of chitin.
the interstices of a device that is intended to elicit a desired
tissue or cell response.
coincidence, n—morethanonecelltransversingtheapertureat
DISCUSSION—Growth factors, antibiotics, and antimicrobials are typi-
the same time.
cal examples of bioactive agents. Device structural components or
degradation byproducts that evoke limited localized bioactivity are not
collagen, n—Type I collagen is a member of a family of
included.
structural proteins found in animals.
DISCUSSION—Type I collagen is part of the fibrillar group of colla-
biocompatibility, n—a material may be considered biocom-
gens. It derives from the COL1A1 and COL1A2 genes, which express
patible if the materials perform with an appropriate host
the alpha chains of the collagen. All collagens have a unique triple
response in a specific application.
helical structure configuration of three polypeptide units known as
alpha-chains. Proper alignment of the alpha chains of the collagen
biological product, n—“any virus, therapeutic serum, toxin,
moleculerequiresahighlycomplexenzymaticandchemicalinteraction
antitoxin, vaccine, blood, blood component or derivative,
in vivo.As such, preparation of the collagen by alternate methods may
allergenicproduct,oranalogousproduct,orarsphenamineor
result in improperly aligned alpha chains and, putatively, increase the
its derivatives (or any trivalent organic arsenic compound)
immunogenicity of the collagen. Collagen is high in glycine, L-alanine,
applicabletotheprevention,treatment,orcureofdiseasesor
L-proline, and 4-hydroxyproline, low in sulfur, and contains no
injuries of man.” (2). L-tryptophan. Natural, fibrillar Type I collagen is normally soluble in
DISCUSSION—Thetermanalogousproductisinterpretedtoencompass dilute acids and alkalis. When heated (for example, above approxi-
somatic cell and gene therapy (3).Abiological product may be used as mately 40°C), collagen is denatured to single alpha chains (gelatin).At
a component of a TEMP. For the purposes of TEMPs, these biological each end of the chains are short non-helical domains called
products may be of any origin (that is, organism), tissue type, telopeptides, which are removed in some collagen preparations.
developmental stage, and may be living, non-living, and genetically or Through non-covalent interactions with sites on adjacent helixes,
otherwise modified. fibrillogenesis is achieved. Subsequently, non-reducible cross-links are
F2312 − 11 (2020)
formed. Type I collagen can be associated with Type III and Type V
achievement of its primary intended purposes.” Devices are
collagen and also with the other non-collagenous proteins like elastin
“intendedtoaffectthestructureoranyfunctionofthebody.”
and other structural molecules like glycosaminoglycans and complex
(Section 201(h)(1) (6)).
lipoproteins and glycoproteins.
DISCUSSION—Device Criteria: “A liquid, powder, or other similar
formulation intended only to serve as a component, part or accessory to
combination product, n—as defined in 21 CFR § 3.2(e), the
a device with a primary mode of action that is physical in nature” (7).
term combination product includes: (1)Aproduct comprised
A device may be used as a component of a TEMP.
of two or more regulated components, that is, drug/device,
biologic/device, drug/biologic, or drug/device/biologic, that disinfection, n—the destruction or reduction of pathogenic and
are physically, chemically, or otherwise combined or mixed other kinds of microorganisms by thermal or chemical
and produced as a single entity; (2) Two or more separate means (for example, alcohol, antibiotics, germicides).
products packaged together in a single package or as a unit
donor, n—a living or deceased organism who is the source of
and comprised of drug and device products, device and
cellsortissues,orboth,forresearchorfurtherprocessingfor
biological products, or biological and drug products; (3)A
transplantation in accordance with established medical cri-
drug, device, or biological product packaged separately that
teria and procedures.
according to its investigational plan or proposed labeling is
intended for use only with an approved individually speci-
dressing, n—any of various materials utilized for covering and
fied drug, device, or biological product where both are protecting a wound.
requiredtoachievetheintendeduse,indication,oreffectand
drug, n—“articles intended for use in the diagnosis, cure,
where upon approval of the proposed product the labeling of
mitigation, treatment, or prevention of disease in man or
the approved product would need to be changed, for
other animals.” Drugs are “intended to affect the structure or
example, to reflect a change in intended use, dosage form,
any function of the body of man or other animals.” (Section
strength, route of administration, or significant change in
201(g)(1) (6) ).
dose; or (4) Any investigational drug, device, or biological
DISCUSSION—Drug Criteria: “Aliquid, powder, tablet or other similar
product packaged separately that according to its proposed
formulation that achieves its primary intended purpose through chemi-
labeling is for use only with another individually specified
cal action within or on the body, or by being metabolized” (7). A drug
investigational drug, device, or biological product where
may be used as a component of a TEMP.
both are required to achieve the intended use, indication, or
drug therapy, n—is the delivery of drug(s) that stimulate a
effect.” Furthermore, “many somatic cell products adminis-
specific physiologic (metabolic) response. Drug therapy
tered to patients will be combinations of a biological product
technologies can be applied in tissue engineering to generate
and a device or of a drug, a biological product, and a
TEMPs.
device.” (5). The term “combination product” may apply to
TEMPs.
electrolyte, n—diluent, offering slight conductivity, in which
cells are suspended.
corrected count, n—the cell count corrected for coincidence.
encapsulation, n—a procedure by which biological materials,
cross-contamination, n—the unintended presence of a cell or
such as cells, tissues, or proteins, are enclosed within a
a material with another cell or material.
microscopic or macroscopic semipermeable barrier.
degree of deacetylation, n—the fraction or percentage of
engraftment, n—incorporation of grafted tissue into the body
glucosamine units (GlcN: deacetylated monomers) in a
of the host.
chitosan polymer molecule.
endotoxin, n—pyrogenic high molar mass lipopolysaccharide
depolymerization, n—reduction in length of a polymer chain
(LPS) complex associated with the cell wall of gram-
to form shorter polymeric units. Depolymerization may
negative bacteria.
reduce the polymer chain to oligomeric or monomeric units,
DISCUSSION—Though endotoxins are pyrogens, not all pyrogens are
or both.
endotoxins. Endotoxins are specifically detected through a Limulus
Amebocyte Lysate (LAL) test.
dermal autograft, n—a skin [autograft] from which epidermis
and subcutaneous fat have been removed; used instead of
epidermal autograft, n—an autograft consisting primarily of
fascia in various plastic [surgery] procedures.
epidermal tissue, including keratinocyte stem cells, but with
little dermal tissue.
device, n—“an instrument, apparatus, implement, machine,
contrivance, implant, in vitro reagent, or other similar or
extracellular matrix, n—“(ECM), any material produced by
relatedarticle.intendedforuseinthediagnosisofdiseaseor
cells and excreted to the extracellular space within the
other conditions, or in the cure, mitigation, treatment, or
tissues. It takes the form of both ground substance and fibers
prevention of disease, in man or other animals,.which does
not achieve its primary intended purposes through chemical
action within or on the body of man or other animals and
For practical details, see Fang , P., Engrav, L. H., Gibran, N. S., Horani, S.,
which is not dependent upon being metabolized for the
Kiriluk,D.B.,Cole,J.K.,Fleckman,P.,Heimbach,D.M.,Gauer,G.J.,Matsumura,
H., and Warner, P., “Dermatome Steeing forAutografts to Cover Integra®,” J Burn
Care Rehabil,Vol 23, 2002, pp. 327–332; and Kagan, R. J., Invited editorial, J Burn
“a sheet or band of fibrous tissue such as lies deep to the skin …” (Dorland’s). Care Rehibil, Vol 23, 2002, p. 326.
F2312 − 11 (2020)
and is composed chiefly of fibrous elements, proteins in- ultimately forming the cicatrix [scar]; it consists of small,
volved in cell adhesion, and glycosaminoglycans and other translucent, red, nodular masses or granulations that have a
space-filling molecules. It serves as a scaffolding holding velvety appearance.
tissuestogetheranditsformandcompositionhelpdetermine
heal, v—to restore wounded parts or to make healthy.
tissue characteristics.” (8) Extracellular matrix, a biological
material or tissue derivative, may be used as a component of
healing, n—the restoration of integrity to injured tissue.
a TEMP.
DISCUSSION—In the surgical wound closure, an important distinction
is made according to whether the surgeon expects the healing to be
femtolitre, n—a cubic micron; a measurement of cell volume.
accomplished by granulation tissue.This distinction is made because in
the normal physiology of wound healing, granulation tissue matures
full thickness skin autograft, n—a skin [auto]graft consisting
into scar with wound contracture, which is an undesirable outcome.
of the epidermis and the full thickness of the dermis.
Wound closure “by approximating the wound edges or performing a
skin autograft” is called “healing by first intention,” and wound closure
full-thickness skin wound, n—a skin wound with the loss of
by“allowingspontaneoushealingfromtheedges”iscalled“healingby
epidermis, and all of the dermis or at least the depth of
second intention.”
dermis that includes most or all sources of epidermal cells
from epidermal adnexae (glands and follicles).
healing by first intention, n—healing in which union or
restoration of continuity occurs directly without intervention
GDF, n—growth and differentiation factor.
of granulations.
gel, n—the three-dimensional network structure arising from
healing by second intention, n—union by closure of a wound
intermolecular polymer chain interactions.
with granulations which form from the base and both sides
DISCUSSION—Such chain interactions may be covalent, ionic, hydro-
toward the surface of the wound.
gen bond, or hydrophobic in nature. See also Terminology F1251.
hydrocolloid, n—a water-soluble polymer of colloidal nature
genetic material, n—is nucleic acid (either deoxyribonucleic
when hydrated.
acid or ribonucleic acid).
DISCUSSION—Genetic material is also known as DNA, RNA, genetic
immobilization, n—the entrapment of materials, such as cells,
element, gene, factor, allele, operon, structural gene, regulator gene,
tissues, or proteins within, or bound to, a matrix.
operator gene, gene complement, genome, genetic code, codon,
anticodon, messenger RNA(mRNA), transfer RNA(tRNA), ribosomal
implantation, n—the procedure of inserting materials such as
extrachromosomal genetic element, plasmagene, plasmid, transposon,
a cell(s), tissue(s), or organ(s) for therapeutic purposes.
gene mutation, gene sequence, exon, intron (modified version, (9)).
Genetic material may be used as a component of a TEMP. Synonym: graft or grafting. TEMPs may be applied to a
recipient by implantation or grafting.
gene therapy, n—“is a medical intervention based on modifi-
cation of the genetic material of living cells. Cells may be in-process control, n—monitoring and, if necessary, adjust-
modified ex vivo for subsequent administration or may be
ments performed to ensure that the process conforms to its
altered in vivo by gene therapy products given directly to the specification. The control of the environment or equipment
subject.When the genetic manipulation is performed ex vivo
may be part of in-process control.
on cells that are then administered to the patient, this is also
lesion, n—any pathological or traumatic discontinuity of tissue
atypeofsomaticcelltherapy.Thegeneticmanipulationmay
or loss of function of a part. In this guide, “skin lesion” is
be intended to prevent, treat, cure, diagnose, or mitigate
intended to encompass
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...