ASTM F1670/F1670M-24
(Test Method)Standard Test Method for Resistance of Materials Used in Protective Clothing to Penetration by Synthetic Blood
Standard Test Method for Resistance of Materials Used in Protective Clothing to Penetration by Synthetic Blood
SIGNIFICANCE AND USE
5.1 This test method is based on Test Method F903 for measuring resistance of chemical protective clothing materials to penetration by liquids. This test method is normally used to evaluate specimens from individual finished items of protective clothing and individual samples of materials that are candidates for items of protective clothing.
5.1.1 Finished items of protective clothing include gloves, arm shields, aprons, gowns, coveralls, hoods, and boots.
5.1.2 The phrase “specimens from finished items” encompasses seamed and other discontinuous regions as well as the usual continuous regions of protective clothing items.
5.2 Medical protective clothing materials are intended to be a barrier to blood, body fluids, and other potentially infectious materials. Many factors can affect the wetting and penetration characteristics of body fluids, such as surface tension, viscosity, and polarity of the fluid, as well as the structure and relative hydrophilicity or hydrophobicity of the materials. The surface tension range for blood and body fluids (excluding saliva) is approximately 42 to 60 dyn/cm (0.042 to 0.060 N/m) (1).7 To help simulate the wetting characteristics of blood and body fluids, the surface tension of the synthetic blood is adjusted to approximate the lower end of this surface tension range. The resulting surface tension of the synthetic blood is approximately 40 ± 5 dyn/cm (0.040 ± 0.005 N/m).
5.3 The synthetic blood mixture is prepared with a red dye to aid in visual detection and a thickening agent to simulate the flow characteristics of blood.
5.4 Part of the protocol in Procedures A and B in Table 1 for exposing the protective clothing material specimens with synthetic blood involves pressurizing the test cell to 13.8 kPa [2.0 psig]. This hydrostatic pressure has been documented to discriminate between protective clothing material performance and to correlate with visual penetration results that are obtained with a human factors validati...
SCOPE
1.1 This test method is used to evaluate the resistance of materials used in protective clothing to penetration by synthetic blood under conditions of continuous liquid contact. Protective clothing pass/fail determinations are based on visual detection of synthetic blood penetration.
1.1.1 This test method is not always effective in testing protective clothing materials having thick inner liners which readily absorb the synthetic blood.
1.2 This test method is a means for selecting protective clothing materials for subsequent testing with a more sophisticated barrier test as described in Test Method F1671/F1671M.
1.3 This test method does not apply to all forms or conditions of blood-borne pathogen exposure. Users of the test method must review modes for work/clothing exposure and assess the appropriateness of this test method for their specific application.
1.4 This test method addresses only the performance of materials or certain material constructions (for example, seams) used in protective clothing. This test method does not address the design, overall construction and components, or interfaces of garments, or other factors which may affect the overall protection offered by the protective clothing.
1.5 The values stated in either SI units or inch-pound units are to be regarded separately as standard. The values stated in each system may not be exact equivalents; therefore, each system shall be used independently of the other. Combining values from the two systems may result in nonconformance with the standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.7 This international standard was developed in accordance with internationally recognize...
General Information
- Status
- Published
- Publication Date
- 31-Jan-2024
- Technical Committee
- F23 - Personal Protective Clothing and Equipment
- Drafting Committee
- F23.40 - Biological
Relations
- Effective Date
- 01-Feb-2024
- Effective Date
- 01-Feb-2024
- Effective Date
- 01-Feb-2024
- Effective Date
- 01-Feb-2024
- Effective Date
- 01-Feb-2024
Overview
ASTM F1670/F1670M-24 is the internationally recognized standard test method for evaluating the resistance of materials used in protective clothing to penetration by synthetic blood. Developed by ASTM, this test method is essential for assessing the barrier performance of medical and personal protective garments intended to protect against potential exposure to blood and body fluids. The standard specifically measures the potential of material specimens, including seams and finished items such as gowns, gloves, coveralls, and aprons, to resist synthetic blood penetration under continuous liquid contact, using a visual pass/fail approach.
This test method provides a controlled, repeatable way to evaluate material candidates for protective clothing before advancing to more stringent microbiological barrier tests, ensuring quality control and initial screening of materials for healthcare, laboratory, and industrial applications.
Key Topics
- Protective Clothing Material Testing: The focus is on evaluating the resistance of various protective clothing materials-including fabrics, seams, and finished items-against penetration from synthetic blood, which simulates human blood in terms of surface tension and viscosity.
- Test Method Application: The standard describes procedures where test specimens are exposed to synthetic blood under defined conditions of time and pressure. Pass/fail determinations are made based on visible penetration.
- Scope and Limitations: The method is primarily a preliminary screening tool and may not fully address extreme conditions or all routes of potential exposure to blood-borne pathogens. Particularly, it may not work effectively for materials with thick absorbent liners or under very high pressure scenarios that exceed those present in the test method.
- Quality Control and Sample Selection: The procedure emphasizes consistent sampling, specimen conditioning, and preparation of synthetic blood fluid. The standard facilitates confidence in material selection by providing guidance on statistical sampling for quality assurance.
Applications
ASTM F1670/F1670M-24 has significant practical value in the following applications:
- Healthcare and Medical Devices: Used for testing surgical gowns, isolation gowns, gloves, and other medical protective equipment to ensure resistance to blood and biological fluids, reducing the risk of cross-contamination and exposure to blood-borne pathogens such as HIV, HBV, and HCV.
- Industrial and Laboratory Safety: Applicable to protective apparel in laboratories and industries where employees may encounter blood or chemically similar fluids, including research, veterinary, and emergency response environments.
- Quality Assurance and Manufacturing: Supports manufacturers in product development and batch quality control of personal protective equipment (PPE) by providing a consistent, visual, and reproducible test method for synthetic blood penetration.
- Regulatory Compliance: Assists organizations with meeting occupational health and safety standards relating to blood-borne pathogen exposure (e.g., OSHA 29 CFR 1910.1030).
Related Standards
ASTM F1670/F1670M-24 is supported and complemented by several relevant standards, including:
- ASTM F1671/F1671M: Test method for resistance of materials used in protective clothing to penetration by blood-borne pathogens using a more sophisticated microbiological challenge.
- ASTM F903: Test method for resistance to penetration by liquids, which forms the basis for F1670/F1670M’s approach.
- ASTM D1777: Test method for measurement of textile thickness.
- ASTM D1331: Test methods for surface and interfacial tension of solutions.
- ISO 2859-1: Sampling procedures for inspection by attributes, used for statistical quality control.
- OSHA 29 CFR 1910.1030: Regulatory standard for occupational exposure to blood-borne pathogens.
Keywords: synthetic blood penetration, protective clothing standard, blood-borne pathogens, ASTM F1670, PPE testing, medical garment barrier, personal protective equipment, healthcare safety standard.
Buy Documents
ASTM F1670/F1670M-24 - Standard Test Method for Resistance of Materials Used in Protective Clothing to Penetration by Synthetic Blood
REDLINE ASTM F1670/F1670M-24 - Standard Test Method for Resistance of Materials Used in Protective Clothing to Penetration by Synthetic Blood
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F1670/F1670M-24 is a standard published by ASTM International. Its full title is "Standard Test Method for Resistance of Materials Used in Protective Clothing to Penetration by Synthetic Blood". This standard covers: SIGNIFICANCE AND USE 5.1 This test method is based on Test Method F903 for measuring resistance of chemical protective clothing materials to penetration by liquids. This test method is normally used to evaluate specimens from individual finished items of protective clothing and individual samples of materials that are candidates for items of protective clothing. 5.1.1 Finished items of protective clothing include gloves, arm shields, aprons, gowns, coveralls, hoods, and boots. 5.1.2 The phrase “specimens from finished items” encompasses seamed and other discontinuous regions as well as the usual continuous regions of protective clothing items. 5.2 Medical protective clothing materials are intended to be a barrier to blood, body fluids, and other potentially infectious materials. Many factors can affect the wetting and penetration characteristics of body fluids, such as surface tension, viscosity, and polarity of the fluid, as well as the structure and relative hydrophilicity or hydrophobicity of the materials. The surface tension range for blood and body fluids (excluding saliva) is approximately 42 to 60 dyn/cm (0.042 to 0.060 N/m) (1).7 To help simulate the wetting characteristics of blood and body fluids, the surface tension of the synthetic blood is adjusted to approximate the lower end of this surface tension range. The resulting surface tension of the synthetic blood is approximately 40 ± 5 dyn/cm (0.040 ± 0.005 N/m). 5.3 The synthetic blood mixture is prepared with a red dye to aid in visual detection and a thickening agent to simulate the flow characteristics of blood. 5.4 Part of the protocol in Procedures A and B in Table 1 for exposing the protective clothing material specimens with synthetic blood involves pressurizing the test cell to 13.8 kPa [2.0 psig]. This hydrostatic pressure has been documented to discriminate between protective clothing material performance and to correlate with visual penetration results that are obtained with a human factors validati... SCOPE 1.1 This test method is used to evaluate the resistance of materials used in protective clothing to penetration by synthetic blood under conditions of continuous liquid contact. Protective clothing pass/fail determinations are based on visual detection of synthetic blood penetration. 1.1.1 This test method is not always effective in testing protective clothing materials having thick inner liners which readily absorb the synthetic blood. 1.2 This test method is a means for selecting protective clothing materials for subsequent testing with a more sophisticated barrier test as described in Test Method F1671/F1671M. 1.3 This test method does not apply to all forms or conditions of blood-borne pathogen exposure. Users of the test method must review modes for work/clothing exposure and assess the appropriateness of this test method for their specific application. 1.4 This test method addresses only the performance of materials or certain material constructions (for example, seams) used in protective clothing. This test method does not address the design, overall construction and components, or interfaces of garments, or other factors which may affect the overall protection offered by the protective clothing. 1.5 The values stated in either SI units or inch-pound units are to be regarded separately as standard. The values stated in each system may not be exact equivalents; therefore, each system shall be used independently of the other. Combining values from the two systems may result in nonconformance with the standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognize...
SIGNIFICANCE AND USE 5.1 This test method is based on Test Method F903 for measuring resistance of chemical protective clothing materials to penetration by liquids. This test method is normally used to evaluate specimens from individual finished items of protective clothing and individual samples of materials that are candidates for items of protective clothing. 5.1.1 Finished items of protective clothing include gloves, arm shields, aprons, gowns, coveralls, hoods, and boots. 5.1.2 The phrase “specimens from finished items” encompasses seamed and other discontinuous regions as well as the usual continuous regions of protective clothing items. 5.2 Medical protective clothing materials are intended to be a barrier to blood, body fluids, and other potentially infectious materials. Many factors can affect the wetting and penetration characteristics of body fluids, such as surface tension, viscosity, and polarity of the fluid, as well as the structure and relative hydrophilicity or hydrophobicity of the materials. The surface tension range for blood and body fluids (excluding saliva) is approximately 42 to 60 dyn/cm (0.042 to 0.060 N/m) (1).7 To help simulate the wetting characteristics of blood and body fluids, the surface tension of the synthetic blood is adjusted to approximate the lower end of this surface tension range. The resulting surface tension of the synthetic blood is approximately 40 ± 5 dyn/cm (0.040 ± 0.005 N/m). 5.3 The synthetic blood mixture is prepared with a red dye to aid in visual detection and a thickening agent to simulate the flow characteristics of blood. 5.4 Part of the protocol in Procedures A and B in Table 1 for exposing the protective clothing material specimens with synthetic blood involves pressurizing the test cell to 13.8 kPa [2.0 psig]. This hydrostatic pressure has been documented to discriminate between protective clothing material performance and to correlate with visual penetration results that are obtained with a human factors validati... SCOPE 1.1 This test method is used to evaluate the resistance of materials used in protective clothing to penetration by synthetic blood under conditions of continuous liquid contact. Protective clothing pass/fail determinations are based on visual detection of synthetic blood penetration. 1.1.1 This test method is not always effective in testing protective clothing materials having thick inner liners which readily absorb the synthetic blood. 1.2 This test method is a means for selecting protective clothing materials for subsequent testing with a more sophisticated barrier test as described in Test Method F1671/F1671M. 1.3 This test method does not apply to all forms or conditions of blood-borne pathogen exposure. Users of the test method must review modes for work/clothing exposure and assess the appropriateness of this test method for their specific application. 1.4 This test method addresses only the performance of materials or certain material constructions (for example, seams) used in protective clothing. This test method does not address the design, overall construction and components, or interfaces of garments, or other factors which may affect the overall protection offered by the protective clothing. 1.5 The values stated in either SI units or inch-pound units are to be regarded separately as standard. The values stated in each system may not be exact equivalents; therefore, each system shall be used independently of the other. Combining values from the two systems may result in nonconformance with the standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognize...
ASTM F1670/F1670M-24 is classified under the following ICS (International Classification for Standards) categories: 11.140 - Hospital equipment; 13.340.10 - Protective clothing. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F1670/F1670M-24 has the following relationships with other standards: It is inter standard links to ASTM F1670/F1670M-17a, ASTM F1671/F1671M-22, ASTM F1359/F1359M-22, ASTM F1819-19, ASTM F1862/F1862M-24. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F1670/F1670M-24 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F1670/F1670M − 24
Standard Test Method for
Resistance of Materials Used in Protective Clothing to
Penetration by Synthetic Blood
This standard is issued under the fixed designation F1670/F1670M; the number immediately following the designation indicates the year
of original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval.
A superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
INTRODUCTION
Workers, primarily those in the healthcare profession involved in treating and caring for individuals
injured or sick, can be exposed to biological liquids capable of transmitting disease. These diseases,
which may be caused by a variety of microorganisms, can pose significant risks to life and health. This
is especially true of blood-borne hepatitis (hepatitis B virus (HBV) and hepatitis C virus (HCV)) and
acquired immune deficiency syndrome (AIDS) (human immunodeficiency viruses (HIV)). Since
engineering controls cannot eliminate all possible exposures, attention is placed on reducing the
potential of direct skin contact through the use of protective clothing that resists penetration (29 CFR
Part 1910.1030). This test method was developed to help assess the effectiveness of materials used in
protective clothing for protecting the wearer against contact with body fluids that potentially contain
blood-borne pathogens. Using synthetic blood, this test method is intended to identify protective
clothing material candidates for further testing according to a more rigorous procedure involving a
surrogate for blood-borne pathogens.
1. Scope 1.4 This test method addresses only the performance of
materials or certain material constructions (for example,
1.1 This test method is used to evaluate the resistance of
seams) used in protective clothing. This test method does not
materials used in protective clothing to penetration by synthetic
address the design, overall construction and components, or
blood under conditions of continuous liquid contact. Protective
interfaces of garments, or other factors which may affect the
clothing pass/fail determinations are based on visual detection
overall protection offered by the protective clothing.
of synthetic blood penetration.
1.1.1 This test method is not always effective in testing
1.5 The values stated in either SI units or inch-pound units
protective clothing materials having thick inner liners which
are to be regarded separately as standard. The values stated in
readily absorb the synthetic blood.
each system may not be exact equivalents; therefore, each
system shall be used independently of the other. Combining
1.2 This test method is a means for selecting protective
values from the two systems may result in nonconformance
clothing materials for subsequent testing with a more sophis-
with the standard.
ticated barrier test as described in Test Method F1671/F1671M.
1.6 This standard does not purport to address all of the
1.3 This test method does not apply to all forms or condi-
safety concerns, if any, associated with its use. It is the
tions of blood-borne pathogen exposure. Users of the test
responsibility of the user of this standard to establish appro-
method must review modes for work/clothing exposure and
priate safety, health, and environmental practices and deter-
assess the appropriateness of this test method for their specific
mine the applicability of regulatory limitations prior to use.
application.
1.7 This international standard was developed in accor-
dance with internationally recognized principles on standard-
This test method is under the jurisdiction of ASTM Committee F23 on Personal
Protective Clothing and Equipment and is the direct responsibility of Subcommittee
ization established in the Decision on Principles for the
F23.40 on Biological.
Development of International Standards, Guides and Recom-
Current edition approved Feb. 1, 2024. Published March 2024. Originally
mendations issued by the World Trade Organization Technical
approved in 1995. Last previous edition approved in 2017 as F1670/F1670M – 17a.
DOI: 10.1520/F1670_F1670M-24. Barriers to Trade (TBT) Committee.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F1670/F1670M − 24
2. Referenced Documents 3.3.1 Discussion—In this test method, synthetic blood is
2 used as a body fluid simulant.
2.1 ASTM Standards:
3.4 penetration, n—the movement of matter through
D1331 Test Methods for Surface and Interfacial Tension of
closures, porous materials, seams, and pinholes or other
Solutions of Paints, Solvents, Solutions of Surface-Active
imperfections in protective clothing on a nonmolecular level.
Agents, and Related Materials
3.4.1 Discussion—For this test method, the specific matter
D1777 Test Method for Thickness of Textile Materials
is synthetic blood.
D3776/D3776M Test Methods for Mass Per Unit Area
3.4.2 Discussion—In this test method, the penetration liquid
(Weight) of Fabric
is synthetic blood.
E105 Guide for Probability Sampling of Materials
E171/E171M Practice for Conditioning and Testing Flexible
3.5 protective clothing, n—an item of clothing that is
Barrier Packaging
specifically designed and constructed for the intended purpose
F903 Test Method for Resistance of Materials Used in
of isolating all or part of the body from a potential hazard; or,
Protective Clothing to Penetration by Liquids
isolating the external environment from contamination by the
F1671/F1671M Test Method for Resistance of Materials
wearer of the clothing.
Used in Protective Clothing to Penetration by Blood-
3.5.1 Discussion—The potential hazard is contact with
Borne Pathogens Using Phi-X174 Bacteriophage Penetra-
blood.
tion as a Test System
3.6 synthetic blood, n—a mixture of a red dye/surfactant,
2.2 Military Standard:
thickening agent, and distilled water having a surface tension
MIL-STD-105E Sampling Procedures and Tables for In-
and viscosity representative of blood and some other body
spection by Attributes
fluids, and the color of blood.
2.3 ANSI/ASQC Standards:
3.6.1 Discussion—The synthetic blood in this test method
ANSI/ASQC Z1.4 Sampling Procedures and Tables for In-
does not simulate all of the characteristics of real blood or body
spection by Attributes
fluids, for example, polarity (a wetting characteristic),
2.4 ISO Standard: coagulation, and content of cell matter.
ISO 2859-1 Sampling Plans for Inspection by Attributes
4. Summary of Test Method
2.5 OSHA Standard:
29 CFR Part 1910.1030 Occupational Exposure to Blood-
4.1 A specimen is subjected to a body fluid simulant
Borne Pathogens: Final Rule, Federal Register, Vol 56,
(synthetic blood) for a specified time and pressure.
No 235, Dec. 6, 1991, pp. 6175–64182
4.2 Visual observation is made to determine when, or if,
penetration occurs.
3. Terminology
4.3 Any evidence of synthetic blood penetration constitutes
3.1 blood-borne pathogen, n—an infectious secreted or
failure. Results are reported as pass/fail.
excreted bacterium, virus, or other disease-inducing microbe
carried in blood or other body fluids.
5. Significance and Use
3.2 body fluid, n—any liquid produced, secreted, or excreted
5.1 This test method is based on Test Method F903 for
by the human body.
measuring resistance of chemical protective clothing materials
3.2.1 Discussion—In this test method, body fluids include
to penetration by liquids. This test method is normally used to
those liquids potentially infected with blood-borne pathogens,
evaluate specimens from individual finished items of protective
including, but not limited to, blood, semen, vaginal secretions,
clothing and individual samples of materials that are candidates
cerebrospinal fluid, synovial fluid and peritoneal fluid, amni-
for items of protective clothing.
otic fluid, saliva in dental procedures, and any body fluid that
5.1.1 Finished items of protective clothing include gloves,
is visibly contaminated with blood, and all body fluids in
arm shields, aprons, gowns, coveralls, hoods, and boots.
situations where it is difficult or impossible to differentiate
5.1.2 The phrase “specimens from finished items” encom-
between body fluids (see 29 CFR Part 1910.1030).
passes seamed and other discontinuous regions as well as the
3.3 body fluid simulant, n—a liquid that is used to mimic
usual continuous regions of protective clothing items.
aspects of human body fluids.
5.2 Medical protective clothing materials are intended to be
a barrier to blood, body fluids, and other potentially infectious
For referenced ASTM standards, visit the ASTM website, www.astm.org, or materials. Many factors can affect the wetting and penetration
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
characteristics of body fluids, such as surface tension,
Standards volume information, refer to the standard’s Document Summary page on
viscosity, and polarity of the fluid, as well as the structure and
the ASTM website.
relative hydrophilicity or hydrophobicity of the materials. The
Standardization Documents Order Desk, Bldg. 4 Section D, 700 Robbins Ave.,
Philadelphia, PA 19111-5094. Document Status: Canceled.
surface tension range for blood and body fluids (excluding
Available from American Society for Quality Control, 611 E. Wisconsin Ave.,
saliva) is approximately 42 to 60 dyn/cm (0.042 to 0.060 N/m)
Milwaukee, WI 53202.
Available from American National Standards Institute (ANSI), 25 W. 43rd St.,
4th Floor, New York, NY 10036, http://www.ansi.org.
Available from U.S. Government Publishing Office, Washington, DC 20402,
http://www.gpo.gov.
F1670/F1670M − 24
TABLE 1 Specimen Exposure Procedures
synthetic blood penetration following an appropriate precon-
Procedure Pressure/Time Sequence and Retaining Screen Options ditioning technique representative of the expected conditions
A 0 kPa [0 psig] for 5 min, followed by 13.8 kPa [2.0 psig] for 1 of use.
min, followed by 0 kPa [0 psig] for 54 min.
5.7 While this test method involves a qualitative determi-
A retaining screen is not used to support the sample.
nation of the protective clothing material resistance to penetra-
B 0 kPa [0 psig] for 5 min, followed by 13.8 kPa [2.0 psig] for 1
tion by synthetic blood under specific test conditions, it is
min, followed by 0 kPa [0 psig] for 54 min.
possible to use this test method as a material quality control or
A retaining screen is used to support the sample. The type
must be specified in the report.
assurance procedure.
5.7.1 If this procedure is used for quality control, perform
proper statistical design and analysis of the data when more
than three specimens are tested. This type of analysis includes,
(1). To help simulate the wetting characteristics of blood and
but is not limited to, the number of individual specimens tested,
body fluids, the surface tension of the synthetic blood is
the average percent passing or failing, or both, with a standard
adjusted to approximate the lower end of this surface tension
deviation. Data reported in this way helps to establish confi-
range. The resulting surface tension of the synthetic blood is
dence limits concerning product performance. Examples of
approximately 40 6 5 dyn/cm (0.040 6 0.005 N/m).
acceptable sampling plans are found in references such as
5.3 The synthetic blood mixture is prepared with a red dye
MIL-STD-105E, ANSI/ASQC Z1.4, and ISO 2859-1.
to aid in visual detection and a thickening agent to simulate the
flow characteristics of blood.
5.4 Part of the protocol in Procedures A and B in Table 1 for
exposing the protective clothing material specimens with
synthetic blood involves pressurizing the test cell to 13.8 kPa
[2.0 psig]. This hydrostatic pressure has been documented to
discriminate between protective clothing material performance
and to correlate with visual penetration results that are obtained
with a human factors validation (2). Some studies, however,
suggest that mechanical pressures exceeding 345 kPa [50 psig]
can occur during clinical use (3, 4). Therefore, it is important
to understand that this test method does not simulate all the
physical stresses and pressures that are exerted on protective
clothing garments during actual use. This test method is offered
to identify those protective clothing materials that warrant
further evaluation with a microbiological challenge.
5.5 Since this test method uses visual observation rather
than analytical measurements to determine penetration, use this
test method as a preliminary evaluation for possible penetration
of blood and other body fluids. Perform subsequent testing
with a microbiological challenge and analytical technique
using Test Method F1671/F1671M.
NOTE 1—No viral resistance claims can be made based on this test
method, as materials can pass the test method and fail Test Method
F1671/F1671M.
5.6 Testing without considering degradation by physical,
chemical, and thermal stresses which could negatively impact
the performance of the protective barrier could lead to a false
FIG. 1 Exploded View of the Penetration Test Cell with Retaining
sense of security. Consider tests which assess the impact of
Screen
storage conditions and shelf life for disposable products, and
the effects of laundering and sterilization for reusable products.
The integrity of the protective barrier can also be compromised
6. Apparatus
during use by such effects as flexing and abrasion (5). It is also
6.1 Thickness Gauge, suitable for measuring thickness to
possible that prewetting by contaminating materials such as
the nearest 0.02 mm [or nearest 0.001 in.], in accordance with
alcohol and perspiration can also compromise the integrity of
Test Method D1777 (optional).
the protective barrier. If these conditions are of concern,
evaluate the performance of protective clothing materials for
Thickness of each protective clothing material specimen tested may be
determined prior to performing the test procedure, but is not required to comply with
The boldface numbers in parentheses refer to the list of references at the end of this test method. The thickness data for the material may be available from the
this standard. manufacturer.
F1670/F1670M − 24
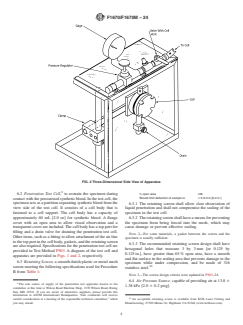
FIG. 2 Three-Dimensional Side View of Apparatus
6.2 Penetration Test Cell, to restrain the specimen during
% open area >50
Should limit deflection of sample to # 5.0 mm [0.2 in.]
contact with the pressurized synthetic blood. In the test cell, the
specimen acts as a partition separating synthetic blood from the 6.3.1 The retaining screen shall allow clear observation of
view side of the test cell. It consists of a cell body that is liquid penetration and shall not compromise the sealing of the
fastened to a cell support. The cell body has a capacity of specimen in the test cell.
approximately 60 mL [2.0 oz] for synthetic blood. A flange 6.3.2 The retaining screen shall have a means for preventing
cover with an open area to allow visual observation and a the specimen from being forced into the mesh, which may
transparent cover are included. The cell body has a top port for
cause damage or prevent effective sealing.
filling and a drain valve for draining the penetration test cell.
NOTE 2—For some materials, a gasket between the screen and the
Other items, such as a fitting to allow attachment of the air line
specimen is usually sufficient.
to the top port in the cell body, gaskets, and the retaining screen
6.3.3 The recommended retaining screen design shall have
are also required. Specifications for the penetration test cell are
hexagonal holes that measure 3 by 3 mm [or 0.125 by
provided in Test Method F903. A diagram of the test cell and
0.125 in.], have greater than 65 % open area, have a smooth
apparatus are provided in Figs. 1 and 2,
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F1670/F1670M − 17a F1670/F1670M − 24
Standard Test Method for
Resistance of Materials Used in Protective Clothing to
Penetration by Synthetic Blood
This standard is issued under the fixed designation F1670/F1670M; the number immediately following the designation indicates the year
of original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval.
A superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
Note—A correction was made to 7.1.2 and the year date was changed on Oct. 25, 2017.
INTRODUCTION
Workers, primarily those in the healthcare profession,profession involved in treating and caring for individuals
injured or sick, can be exposed to biological liquids capable of transmitting disease. These diseases, which may be
caused by a variety of microorganisms, can pose significant risks to life and health. This is especially true of
blood-borne hepatitis (hepatitis B virus (HBV) and hepatitis C virus (HCV)) and acquired immune deficiency
syndrome (AIDS) (human immunodeficiency viruses (HIV)). Since engineering controls can not cannot eliminate
all possible exposures, attention is placed on reducing the potential of direct skin contact through the use of
protective clothing that resists penetration (29 CFR Part 1910.1030). This test method was developed to help assess
the effectiveness of materials used in protective clothing for protecting the wearer against contact with body fluids
that potentially contain blood-borne pathogens. Using synthetic blood, this test method is intended to identify
protective clothing material candidates for further testing according to a more rigorous procedure involving a
surrogate for blood-borne pathogens.
1. Scope
1.1 This test method is used to evaluate the resistance of materials used in protective clothing to penetration by synthetic blood
under conditions of continuous liquid contact. Protective clothing pass/fail determinations are based on visual detection of
synthetic blood penetration.
1.1.1 This test method is not always effective in testing protective clothing materials having thick,thick inner liners which readily
absorb the synthetic blood.
1.2 This test method is a means for selecting protective clothing materials for subsequent testing with a more sophisticated barrier
test as described in Test Method F1671F1671/F1671M.
1.3 This test method does not apply to all forms or conditions of blood-borne pathogen exposure. Users of the test method must
review modes for work/clothing exposure and assess the appropriateness of this test method for their specific application.
1.4 This test method addresses only the performance of materials or certain material constructions (for example, seams) used in
This test method is under the jurisdiction of ASTM Committee F23 on Personal Protective Clothing and Equipment and is the direct responsibility of Subcommittee
F23.40 on Biological.
Current edition approved Oct. 25, 2017Feb. 1, 2024. Published October 2017March 2024. Originally approved in 1995. Last previous edition approved in 2017 as
F1670/F1670M – 17.F1670/F1670M – 17a. DOI: 10.1520/F1670_F1670M-17A.10.1520/F1670_F1670M-24.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F1670/F1670M − 24
protective clothing. This test method does not address the design, overall construction and components, or interfaces of garments,
or other factors which may affect the overall protection offered by the protective clothing.
1.5 The values stated in either SI units or inch-pound units are to be regarded separately as standard. The values stated in each
system may not be exact equivalents; therefore, each system shall be used independently of the other. Combining values from the
two systems may result in nonconformance with the standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D1331 Test Methods for Surface and Interfacial Tension of Solutions of Paints, Solvents, Solutions of Surface-Active Agents,
and Related Materials
D1777 Test Method for Thickness of Textile Materials
D3776D3776/D3776M Test Methods for Mass Per Unit Area (Weight) of Fabric
E105 Guide for Probability Sampling of Materials
E171E171/E171M Practice for Conditioning and Testing Flexible Barrier Packaging
F903 Test Method for Resistance of Materials Used in Protective Clothing to Penetration by Liquids
F1671F1671/F1671M Test Method for Resistance of Materials Used in Protective Clothing to Penetration by Blood-Borne
Pathogens Using Phi-X174 Bacteriophage Penetration as a Test System
2.2 Military Standard:
MIL-STD-105MIL-STD-105E Sampling Procedures and Tables for Inspection by Attributes
2.3 ANSI/ASQC Standards:
ANSI/ASQC Z1.4 Sampling Procedures and Tables for Inspection by Attributes
2.4 ISO Standard:
ISO 2859-1 Sampling Plans for Inspection by Attributes
2.5 OSHA Standard:
29 CFR Part 1910.1030 Occupational Exposure to Blood-Borne Pathogens: Final Rule, Federal Register, Vol 56, No 235, Dec.
6, 1991, pp. 6175–64182
3. Terminology
3.1 blood-borne pathogen, n—an infectious secreted or excreted bacterium, virus, or other disease-inducing microbe carried in
blood or other body fluids.
3.2 body fluid, n—any liquid produced, secreted, or excreted by the human body.
3.2.1 Discussion—
In this test method, body fluids include those liquids potentially infected with blood-borne pathogens, including, but not limited
to, blood, semen, vaginal secretions, cerebrospinal fluid, synovial fluid and peritoneal fluid, amniotic fluid, saliva in dental
procedures, and any body fluid that is visibly contaminated with blood, and all body fluids in situations where it is difficult or
impossible to differentiate between body fluids (see 29 CFR Part 1910.1030).
3.3 body fluid simulant, n—a liquid whichthat is used to act as a model for mimic aspects of human body fluids.
3.3.1 Discussion—
In this test method, synthetic blood is used as a body fluid simulant.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Available from Standardization Documents Order Desk, Bldg. 4 Section D, 700 Robbins Ave., Philadelphia, PA 19111-5094, Attn: NPODS.19111-5094. Document
Status: Canceled.
Available from American Society for Quality Control, 611 E. Wisconsin Ave., Milwaukee, WI 53202.
Available from American National Standards Institute (ANSI), 25 W. 43rd St., 4th Floor, New York, NY 10036, http://www.ansi.org.
Available from Supt. of Documents, U.S. Government PrintingPublishing Office, Washington, DC 20402.20402, http://www.gpo.gov.
F1670/F1670M − 24
3.4 penetration, n—the movement of matter through closures, porous materials, seams, and pinholes or other imperfections in
protective clothing on a nonmolecular level.
3.4.1 Discussion—
For this test method, the specific matter is synthetic blood.
3.4.2 Discussion—
In this test method, the penetration liquid is synthetic blood.
3.5 protective clothing, n—an item of clothing that is specifically designed and constructed for the intended purpose of isolating
all or part of the body from a potential hazard; or, isolating the external environment from contamination by the wearer of the
clothing.
3.5.1 Discussion—
The potential hazard is contact with blood.
3.6 synthetic blood, n—a mixture of a red dye/surfactant, thickening agent, and distilled water having a surface tension and
viscosity representative of blood and some other body fluids, and the color of blood.
3.6.1 Discussion—
The synthetic blood in this test method does not simulate all of the characteristics of real blood or body fluids, for example, polarity
(a wetting characteristic), coagulation, and content of cell matter.
4. Summary of Test Method
4.1 A specimen is subjected to a body fluid simulant (synthetic blood) for a specified time and pressure.
4.2 Visual observation is made to determine when, or if, penetration occurs.
4.3 Any evidence of synthetic blood penetration constitutes failure. Results are reported as pass/fail.
5. Significance and Use
5.1 This test method is based on Test Method F903 for measuring resistance of chemical protective clothing materials to
penetration by liquids. This test method is normally used to evaluate specimens from individual finished items of protective
clothing and individual samples of materials that are candidates for items of protective clothing.
5.1.1 Finished items of protective clothing include gloves, arm shields, aprons, gowns, coveralls, hoods, and boots.
5.1.2 The phrase “specimens from finished items” encompasses seamed and other discontinuous regions as well as the usual
continuous regions of protective clothing items.
5.2 Medical protective clothing materials are intended to be a barrier to blood, body fluids, and other potentially infectious
materials. Many factors can affect the wetting and penetration characteristics of body fluids, such as surface tension, viscosity, and
polarity of the fluid, as well as the structure and relative hydrophilicity or hydrophobicity of the materials. The surface tension
range for blood and body fluids (excluding saliva) is approximately 0.042 to 0.060 N/m42 to 60 dyn/cm (0.042 to 0.060 N/m) (1).
To help simulate the wetting characteristics of blood and body fluids, the surface tension of the synthetic blood is adjusted to
approximate the lower end of this surface tension range. The resulting surface tension of the synthetic blood is approximately 0.042
6 0.002 N/m.40 6 5 dyn/cm (0.040 6 0.005 N/m).
5.3 The synthetic blood mixture is prepared with a red dye to aid in visual detection and a thickening agent to simulate the flow
characteristics of blood.
5.4 Part of the protocol in Procedures A and B in Table 1 for exposing the protective clothing material specimens with synthetic
blood involves pressurization of pressurizing the test cell to 13.8 kPa [2[2.0 psig]. This hydrostatic pressure has been documented
to discriminate between protective clothing material performance and to correlate with visual penetration results that are obtained
The boldface numbers in parentheses refer to the list of references at the end of this standard.
F1670/F1670M − 24
TABLE 1 Specimen Exposure Procedures
Procedure Pressure/Time Sequence and Retaining Screen Options
A 0 kPa [0 psig] for 5 min, followed by 13.8 kPa [2 psig] for 1
min, followed by 0 kPa [0 psig] for 54 min.
A 0 kPa [0 psig] for 5 min, followed by 13.8 kPa [2.0 psig] for 1
min, followed by 0 kPa [0 psig] for 54 min.
A retaining screen is not used to support the sample.
B 0 kPa [0 psig] for 5 min, followed by 13.8 kPa [2 psig] for 1
min, followed by 0 kPa [0 psig] for 54 min.
B 0 kPa [0 psig] for 5 min, followed by 13.8 kPa [2.0 psig] for 1
min, followed by 0 kPa [0 psig] for 54 min.
A retaining screen is used to support the sample. The type
must be specified in the report.
with a human factors validation (2). Some studies, however, suggest that mechanical pressures exceeding 345 kPa [50 psig] can
occur during clinical use (3, 4). Therefore, it is important to understand that this test method does not simulate all the physical
stresses and pressures that are exerted on protective clothing garments during actual use. This test method is offered to identify
those protective clothing materials that warrant further evaluation with a microbiological challenge.
5.5 Since this test method uses visual observation rather than analytical measurements for determination of to determine
penetration, use this test method as a preliminary evaluation for possible penetration of blood and other body fluids. Perform
subsequent testing with a microbiological challenge and analytical technique using Test Method F1671F1671/F1671M.
NOTE 1—No viral resistance claims can be made based on this test method, as materials can pass the test method and fail Test Method
F1671F1671/F1671M.
5.6 Testing without considering degradation by physical, chemical, and thermal stresses which could negatively impact the
performance of the protective barrier could lead to a false sense of security. Consider tests which assess the impact of storage
conditions and shelf life for disposable products, and the effects of laundering and sterilization for reusable products. The integrity
of the protective barrier can also be compromised during use by such effects as flexing and abrasion (5). It is also possible that
prewetting by contaminating materials such as alcohol and perspiration can also compromise the integrity of the protective barrier.
If these conditions are of concern, evaluate the performance of protective clothing materials for synthetic blood penetration
following an appropriate preconditioning technique representative of the expected conditions of use.
5.7 While this test method involves a qualitative determination of the protective clothing material resistance to penetration by
synthetic blood under specific test conditions, it is possible to use this test method as a material quality control or assurance
procedure.
5.7.1 If this procedure is used for quality control, perform proper statistical design and analysis of the data when more than three
specimens are tested. This type of analysis includes, but is not limited to, the number of individual specimens tested, the average
percent passing or failing, or both, with a standard deviation. Data reported in this way helps to establish confidence limits
concerning product performance. Examples of acceptable sampling plans are found in references such as MIL-STD-105,MIL-
STD-105E, ANSI/ASQC Z1.4, and ISO 2859-1.
6. Apparatus
6.1 Thickness Gauge, suitable for measuring thickness to the nearest 0.02 mm [or nearest 0.001 in.], in accordance with Test
Method D1777 (optional).
6.2 Penetration Test Cell, to restrain the specimen during contact with the pressurized test synthetic blood. In the test cell, the
specimen acts as a partition separating synthetic blood from the view side of the test cell. It consists of a cell body that is fastened
to a cell support. The cell body has a capacity of approximately 60 mL [2.0 oz] for synthetic blood. A flange cover,cover with an
Thickness of each protective clothing material specimen tested may be determined prior to performing the test procedure, but is not required to comply with this test
method. The thickness data for the material may be available from the manufacturer.
The sole source of supply of the penetration test apparatus known to the committee at this time is Wilson Road Machine Shop, 1170 Wilson Road, Rising Sun, MD 21911.
If you are aware of alternative suppliers, please provide this information to ASTM International Headquarters. Your comments will receive careful consideration at a meeting
of the responsible technical committee, which you may attend.
F1670/F1670M − 24
FIG. 1 ExplodedExploded View of the Penetration Test Cell with Retaining Screen
open area to allow visual observation and a transparent cover are included. The cell body has a top port for filling and a drain valve
for draining the penetration test cell. Other items, such as a fitting to allow attachment of the air line to the top port in the cell body,
gaskets, and the retaining screen are also required. Specifications for the penetration test cell are provided in Test Method F903.
A diagram of the test cell and apparatus are provided in Figs. 1 and 2, respectively.
6.3 Retaining Screen, a smooth-finish plastic or metal square mesh screen meeting the following specifications used for Procedure
B from Table 1:
% open area >50
Should limit deflection of sample to <= 5.0 mm [0.2 in.]
Should limit deflec
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...