ASTM D7319-22
(Test Method)Standard Test Method for Determination of Existent and Potential Sulfate and Inorganic Chloride in Fuel Ethanol and Butanol by Direct Injection Suppressed Ion Chromatography
Standard Test Method for Determination of Existent and Potential Sulfate and Inorganic Chloride in Fuel Ethanol and Butanol by Direct Injection Suppressed Ion Chromatography
SIGNIFICANCE AND USE
5.1 Sulfates and chlorides can be found in filter plugging deposits and fuel injector deposits. The acceptability for use of the fuel components and the finished fuels depends on the sulfate and chloride content.
5.2 Existent and potential inorganic sulfate and total chloride content, as measured by this test method, can be used as one measure of the acceptability of gasoline components for automotive spark-ignition engine fuel use.
SCOPE
1.1 This test method covers a direct injection ion chromatographic procedure for determining existent and potential inorganic sulfate and total inorganic chloride content in hydrous and anhydrous denatured ethanol and butanol to be used in motor fuel applications. It is intended for the analysis of ethanol and butanol samples containing between 1.0 mg/kg to 20 mg/kg of existent or potential inorganic sulfate and 1.0 mg/kg to 50 mg/kg of inorganic chloride.
Note 1: Tertiary butanol is not included in this test method. 1-butanol, 2-butanol, and isobutanol are included in the testing and research report for this test method.
1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Material Safety Data Sheets are available for reagents and materials. Review them for hazards prior to usage.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Sep-2022
- Technical Committee
- D02 - Petroleum Products, Liquid Fuels, and Lubricants
- Drafting Committee
- D02.03 - Elemental Analysis
Relations
- Refers
ASTM D4175-23a - Standard Terminology Relating to Petroleum Products, Liquid Fuels, and Lubricants - Effective Date
- 15-Dec-2023
- Effective Date
- 01-Dec-2023
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Oct-2023
- Refers
ASTM D4175-23e1 - Standard Terminology Relating to Petroleum Products, Liquid Fuels, and Lubricants - Effective Date
- 01-Jul-2023
- Effective Date
- 15-Dec-2017
- Effective Date
- 15-Nov-2017
- Effective Date
- 01-Jan-2017
- Effective Date
- 01-Oct-2016
- Effective Date
- 01-Oct-2013
- Effective Date
- 15-Sep-2013
- Effective Date
- 15-Jun-2013
- Effective Date
- 01-Jul-2011
- Effective Date
- 01-Jul-2011
- Refers
ASTM D4057-06(2011) - Standard Practice for Manual Sampling of Petroleum and Petroleum Products - Effective Date
- 01-Jun-2011
Overview
ASTM D7319-22 provides a standardized test method for determining existent and potential inorganic sulfate and inorganic chloride in fuel ethanol and butanol using direct injection suppressed ion chromatography. This method is specifically designed to analyze hydrous and anhydrous denatured ethanol, as well as various forms of butanol (excluding tertiary butanol), ensuring their suitability for motor fuel applications. With a focus on the quality and performance of alternative fuels, this standard supports fuel producers, laboratories, and automotive industry stakeholders in maintaining stringent product specifications and regulatory compliance.
Key Topics
Sulfate and Chloride Detection:
The presence of sulfates and chlorides in fuel ethanol and butanol can lead to filter plugging and fuel injector deposits, negatively impacting engine performance. ASTM D7319-22 quantifies both existing and potential sulfate, as well as total inorganic chloride, providing critical data for fuel quality assessment.Direct Injection Suppressed Ion Chromatography:
This method utilizes ion chromatography with direct sample injection and an acid suppressor to enhance measurement sensitivity, particularly for sulfates. The suppressor reduces background conductivity, allowing for accurate quantification in the specified concentration ranges (1.0 mg/kg to 20 mg/kg for sulfate and 1.0 mg/kg to 50 mg/kg for chloride).Precision and Repeatability:
The standard details the expected repeatability and reproducibility for both chloride and sulfate determinations, supporting consistent quality control in laboratory settings.Sample Handling and Calibration:
It emphasizes proper sample collection, preparation, and calibration, referencing related ASTM practices for manual and automatic sampling, ensuring that results are reliable and traceable.Quality Control Measures:
Regular use of quality control samples and statistical process control is recommended to maintain analytical system performance and data integrity.
Applications
Fuel Quality Assurance:
The method is a critical tool for determining whether batches of denatured ethanol or butanol meet industry and regulatory standards for use in automotive spark-ignition engines. Monitoring sulfate and chloride levels helps prevent engine fouling and supports optimal performance of finished fuels.Production and Compliance Testing:
Used routinely by fuel producers, independent labs, and regulatory bodies, ASTM D7319-22 helps ensure compliance with local, national, and international fuel quality requirements.Research and Development:
The methodology is also applicable in research settings where the impact of inorganic contaminants in biofuels or fuel blends is being studied.Supply Chain Management:
Petroleum and alternative fuel suppliers leverage this standard to confirm product quality at various points in the distribution network.
Related Standards
ASTM D7319-22 references several other important standards and practices for accurate and consistent analysis:
ASTM D1193 – Specification for Reagent Water
Defines water quality requirements essential for preparing reagents and standards.ASTM D4052 – Density, Relative Density, and API Gravity of Liquids by Digital Density Meter
Used to convert concentration measurements between mg/L and mg/kg.ASTM D4057 / D4177 – Sampling Petroleum and Petroleum Products
Guidelines for manual and automatic sample collection.ASTM D4175 – Terminology Relating to Petroleum Products, Liquid Fuels, and Lubricants
Provides standard terminology for consistent communication.ASTM D5827, D6299, D6792, D7318, D7328
Include methods and practices for analysis and quality control of fuel components and laboratory operations.EN 15492
European standard for the determination of inorganic chloride and sulfate in ethanol, offering alignment with international testing practices.
Keywords: ASTM D7319-22, direct injection suppressed ion chromatography, sulfate in ethanol, chloride in butanol, fuel ethanol quality, inorganic contaminants in fuel, fuel testing standards, motor fuel applications, biofuel analysis, fuel additive standards.
Buy Documents
ASTM D7319-22 - Standard Test Method for Determination of Existent and Potential Sulfate and Inorganic Chloride in Fuel Ethanol and Butanol by Direct Injection Suppressed Ion Chromatography
REDLINE ASTM D7319-22 - Standard Test Method for Determination of Existent and Potential Sulfate and Inorganic Chloride in Fuel Ethanol and Butanol by Direct Injection Suppressed Ion Chromatography
Get Certified
Connect with accredited certification bodies for this standard

ABS Quality Evaluations Inc.
American Bureau of Shipping quality certification.

ECOCERT
Organic and sustainability certification.

Element Materials Technology
Materials testing and product certification.
Sponsored listings
Frequently Asked Questions
ASTM D7319-22 is a standard published by ASTM International. Its full title is "Standard Test Method for Determination of Existent and Potential Sulfate and Inorganic Chloride in Fuel Ethanol and Butanol by Direct Injection Suppressed Ion Chromatography". This standard covers: SIGNIFICANCE AND USE 5.1 Sulfates and chlorides can be found in filter plugging deposits and fuel injector deposits. The acceptability for use of the fuel components and the finished fuels depends on the sulfate and chloride content. 5.2 Existent and potential inorganic sulfate and total chloride content, as measured by this test method, can be used as one measure of the acceptability of gasoline components for automotive spark-ignition engine fuel use. SCOPE 1.1 This test method covers a direct injection ion chromatographic procedure for determining existent and potential inorganic sulfate and total inorganic chloride content in hydrous and anhydrous denatured ethanol and butanol to be used in motor fuel applications. It is intended for the analysis of ethanol and butanol samples containing between 1.0 mg/kg to 20 mg/kg of existent or potential inorganic sulfate and 1.0 mg/kg to 50 mg/kg of inorganic chloride. Note 1: Tertiary butanol is not included in this test method. 1-butanol, 2-butanol, and isobutanol are included in the testing and research report for this test method. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Material Safety Data Sheets are available for reagents and materials. Review them for hazards prior to usage. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 Sulfates and chlorides can be found in filter plugging deposits and fuel injector deposits. The acceptability for use of the fuel components and the finished fuels depends on the sulfate and chloride content. 5.2 Existent and potential inorganic sulfate and total chloride content, as measured by this test method, can be used as one measure of the acceptability of gasoline components for automotive spark-ignition engine fuel use. SCOPE 1.1 This test method covers a direct injection ion chromatographic procedure for determining existent and potential inorganic sulfate and total inorganic chloride content in hydrous and anhydrous denatured ethanol and butanol to be used in motor fuel applications. It is intended for the analysis of ethanol and butanol samples containing between 1.0 mg/kg to 20 mg/kg of existent or potential inorganic sulfate and 1.0 mg/kg to 50 mg/kg of inorganic chloride. Note 1: Tertiary butanol is not included in this test method. 1-butanol, 2-butanol, and isobutanol are included in the testing and research report for this test method. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Material Safety Data Sheets are available for reagents and materials. Review them for hazards prior to usage. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D7319-22 is classified under the following ICS (International Classification for Standards) categories: 71.040.50 - Physicochemical methods of analysis; 75.160.20 - Liquid fuels. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D7319-22 has the following relationships with other standards: It is inter standard links to ASTM D4175-23a, ASTM D6299-23a, ASTM D6792-23c, ASTM D6792-23b, ASTM D4175-23e1, ASTM D6299-17b, ASTM D6299-17a, ASTM D6299-17, ASTM D7328-16, ASTM D6299-13e1, ASTM D7328-13, ASTM D7318-13, ASTM D7318-11, ASTM D7328-11, ASTM D4057-06(2011). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D7319-22 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D7319 − 22
Standard Test Method for
Determination of Existent and Potential Sulfate and
Inorganic Chloride in Fuel Ethanol and Butanol by Direct
Injection Suppressed Ion Chromatography
This standard is issued under the fixed designation D7319; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* D4052 Test Method for Density, Relative Density, and API
Gravity of Liquids by Digital Density Meter
1.1 This test method covers a direct injection ion chromato-
D4057 Practice for Manual Sampling of Petroleum and
graphic procedure for determining existent and potential inor-
Petroleum Products
ganic sulfate and total inorganic chloride content in hydrous
D4175 Terminology Relating to Petroleum Products, Liquid
and anhydrous denatured ethanol and butanol to be used in
Fuels, and Lubricants
motor fuel applications. It is intended for the analysis of
D4177 Practice for Automatic Sampling of Petroleum and
ethanol and butanol samples containing between 1.0 mg⁄kg to
Petroleum Products
20 mg/kg of existent or potential inorganic sulfate and
D5827 Test Method for Analysis of Engine Coolant for
1.0 mg⁄kg to 50 mg/kg of inorganic chloride.
Chloride and Other Anions by Ion Chromatography
NOTE 1—Tertiary butanol is not included in this test method. 1-butanol,
D6299 Practice for Applying Statistical Quality Assurance
2-butanol, and isobutanol are included in the testing and research report
and Control Charting Techniques to Evaluate Analytical
for this test method.
Measurement System Performance
1.2 The values stated in SI units are to be regarded as
D6792 Practice for Quality Management Systems in Petro-
standard. No other units of measurement are included in this
leum Products, Liquid Fuels, and Lubricants Testing
standard.
Laboratories
1.3 This standard does not purport to address all of the
D7318 TestMethodforExistentInorganicSulfateinEthanol
safety concerns, if any, associated with its use. It is the
by Potentiometric Titration
responsibility of the user of this standard to establish appro-
D7328 Test Method for Determination of Existent and Po-
priate safety, health, and environmental practices and deter-
tential Inorganic Sulfate and Total Inorganic Chloride in
mine the applicability of regulatory limitations prior to use.
Fuel Ethanol by Ion Chromatography Using Aqueous
Material Safety Data Sheets are available for reagents and
Sample Injection
materials. Review them for hazards prior to usage.
2.2 European Norm Standards:
1.4 This international standard was developed in accor-
EN 15492 Ethanol as a blending component for petrol—
dance with internationally recognized principles on standard-
Determination of inorganic chloride and sulfate content—
ization established in the Decision on Principles for the
Ion chromatographic method
Development of International Standards, Guides and Recom-
mendations issued by the World Trade Organization Technical
3. Terminology
Barriers to Trade (TBT) Committee.
3.1 Definitions:
2. Referenced Documents
3.1.1 For definitions of terms used in this test method, refer
2.1 ASTM Standards: to Terminology D4175.
D1193 Specification for Reagent Water 3.2 Definitions of Terms Specific to This Standard:
3.2.1 existent inorganic sulfate, n—inorganic sulfate species
This test method is under the jurisdiction of ASTM Committee D02 on actually present in the sample at the time of analysis with no
Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of
oxidation treatment.
Subcommittee D02.03 on Elemental Analysis.
3.2.2 inorganic chloride, n—chloride present as hydrochlo-
Current edition approved Oct. 1, 2022. Published October 2022. Originally
approved in 2007. Last previous edition approved in 2017 as D7319 – 17. DOI:
ric acid, ionic salts of this acid, or mixtures of these.
10.1520/D7319-22.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Standards volume information, refer to the standard’s Document Summary page on Available fromAmerican National Standards Institute (ANSI), 25 W. 43rd St.,
the ASTM website. 4th Floor, New York, NY 10036, http://www.ansi.org.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D7319 − 22
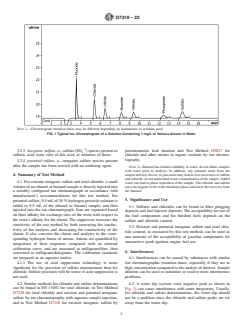
NOTE 1—Chromatogram retention times may be different depending on instruments or columns used.
FIG. 1 Typical Ion Chromatogram of a Solution Containing 1 mg/L of Various Anions in Water
-2
3.2.3 inorganicsulfate,n—sulfate(SO )speciespresentas potentiometric lead titration and Test Method D5827 for
sulfuric acid, ionic salts of this acid, or mixtures of these. chloride and other anions in engine coolants by ion chroma-
tography.
3.2.4 potential sulfate, n—inorganic sulfate species present
after the sample has been reacted with an oxidizing agent.
NOTE 2—Butanol has limited solubility in water, do not dilute samples
with water prior to analysis. In addition, any remnant water from the
sample delivery device or glassware may lead to low recoveries of sulfate
4. Summary of Test Method
andchloride.Avoidunintendedwatercontaminationofthesample.Added
4.1 For existent inorganic sulfate and total chloride, a small
water can lead to phase separation of the sample. The chloride and sulfate
volumeofanethanolorbutanolsampleisdirectlyinjectedinto ions can migrate to the water dominate phase and lower the recovery from
the butanol.
a suitably configured ion chromatograph in accordance with
manufacturer’s recommendations for this test method. For
5. Significance and Use
potentialsulfate,0.5mLof30 %hydrogenperoxidesolutionis
added to 9.5 mL of the ethanol or butanol sample, and then
5.1 Sulfates and chlorides can be found in filter plugging
injected into the ion chromatograph. Ions are separated based
deposits and fuel injector deposits. The acceptability for use of
on their affinity for exchange sites of the resin with respect to
the fuel components and the finished fuels depends on the
the resin’s affinity for the eluent. The suppressor increases the
sulfate and chloride content.
sensitivity of the test method by both increasing the conduc-
5.2 Existent and potential inorganic sulfate and total chlo-
tivity of the analytes and decreasing the conductivity of the
ride content, as measured by this test method, can be used as
eluent. It also converts the eluent and analytes to the corre-
one measure of the acceptability of gasoline components for
sponding hydrogen forms of anions. Anions are quantified by
automotive spark-ignition engine fuel use.
integration of their responses compared with an external
calibration curve, and are measured as milligrams/litre, then
6. Interferences
converted to milligrams/kilograms. The calibration standards
are prepared in an aqueous matrix. 6.1 Interferences can be caused by substances with similar
4.1.1 The use of acid suppression technology is more ion chromatographic retention times, especially if they are in
high concentration compared to the analyte of interest. Sample
significant for the precision of sulfate measurement than for
chloride. Sulfate precision will be worse if acid suppression is dilution can be used to minimize or resolve most interference
problems.
not used.
4.2 Similar methods for chloride and sulfate determinations 6.2 A water dip (system void, negative peak as shown in
can be found in EN 15492 for total chloride, in Test Method Fig. 1) can cause interference with some integrators. Usually,
D7328 for total chloride and existent and potential inorganic for chloride and sulfate determinations, the water dip should
sulfate by ion chromatography with aqueous sample injection, not be a problem since the chloride and sulfate peaks are far
and in Test Method D7318 for existent inorganic sulfate by away from the water dip.
D7319 − 22
6.3 Given the trace amounts of chloride and sulfate deter- 7.6.6 Conductivity Detector, (low volume), temperature
mined in this test method, interferences can be caused by controlled to 0.01 °C, capable of at least 0 µS⁄cm to
contamination of glassware, eluent, reagents, and the like. For 1000 µS⁄cm on a linear scale.
example, sodium leaching from glassware can precipitate with 7.6.7 Integrator or Chromatography Data System Software,
sulfate giving low sulfate results. Take care to ensure that capable of measuring peak areas and retention times, and
contamination is kept at the lowest possible levels. The use of correcting the data in accordance with the baseline of the
powder-free gloves is highly recommended to prevent sample chromatogram.
contamination.
7.7 Gloves, powder-free examination type.
7. Apparatus 8. Reagents
8.1 Purity of Reagents—Reagent grade or higher purity
7.1 Analytical Balance, capable of weighing up to 100 g
chemicals shall be used for the preparation of all samples,
accurately to 0.0001 g.
standards, eluent, and regenerator solutions. Unless otherwise
7.2 Drying Oven, controlled at 110 °C 6 5 °C for drying
indicated, it is intended that all reagents conform to the
sodium sulfate and sodium chloride.
specifications of the Committee on Analytical Reagents of the
American Chemical Society, where such specifications are
7.3 Desiccator, containing freshly activated silica gel (or
equivalent desiccant) with moisture content indicator. available. Other grades may be used, provided it is first
ascertained that the reagent is of sufficiently high purity to
7.4 Pipets or Volumetric Transferring Devices, appropri-
permit its use without lessening the accuracy of the determi-
ately sized Class A glass pipets or their equivalent or variable
nation.
volume automatic pipets fitted with disposable polypropylene
8.2 Purity of Water—Unless otherwise indicated, reference
tips.
to water shall be understood to mean reagent water as defined
7.5 Volumetric Flasks (with Glass Stoppers), Class A of
by Type I or better in Specification D1193. For eluent prepa-
10 mL, 25 mL, 50 mL, 100 mL, 1000 mL, 2000 mL capacity.
ration and handling, comply with all ion chromatograph
7.5.1 Container, equipped with a closure to prevent ethanol
instrument and column vendor requirements (for example,
evaporation, 1 L(for sample collection). See 6.3 for additional
filtering, degassing, and the like).
information.
8.3 Eluent Buffer Stock Solution—Sodium bicarbonate
7.6 Ion Chromatograph, analytical system with all required
(NaHCO ) 1.0 mM and sodium carbonate (Na CO ) 3.2 mM.
3 2 3
accessories including syringes, columns, suppressor, and de-
Dissolve8.4 g 60.0005 gofNaHCO and33.92 g 60.0005 g
tector which can be demonstrated to obtain the precision and
of Na CO in reagent water in a 1 L Type A volumetric flask
2 3
detection limits and requirements of the method.
and dilute to volume. Dilute 10.0 mL of this stock solution to
7.6.1 Injection System, capable of delivering 20 µL with a 1 L in a 1 L Type A volumetric flask with degassed reagent
precision better than 1 %, or as recommended for this deter- water.Theeluentsolutionusedmaybedifferentifothersystem
mination by the manufacturer. or analytical columns are used.
7.6.2 Pumping System, capable of delivering mobile phase
NOTE 3—Other volumes of stock solution can be prepared using
flows between 0.2 mL⁄min and 2.5 mL/min with a precision
appropriate ratios of reagents. Follow the specific guidelines for this
better than 2 %, or as recommended for this determination by solution from the vendor of the column being used. Alternatively, this
solution can be purchased from a qualified vendor.
the manufacturer.
8.4 Suppressor Regenerant Solution for Suppressor—0.1 M
7.6.3 GuardColumn,forprotectionoftheanalyticalcolumn
sulfuric acid. Carefully add 334 mL of reagent grade sulfuric
from strongly retained constituents. Better separations are
acid (relative density 1.84) to approximately 500 mL reagent
obtained with additional theoretical plates.
water in a 1 Lvolumetric flask. (Warning—This will generate
7.6.4 Anion Separator Column, 100 % solvent compatible
averyhotsolution.Allowittocoolbeforedilutingto1000mL
and capable of producing satisfactory analyte (sulfate and
volume. Never add water to concentrated acid.) Dilute to
chloride) separation (Fig. 1).
1000 mL with reagent water, and label this solution as 10.0 M
7.6.5 Anion Suppressor Device, vendor specific and capable
sulfuric acid. Dilute 10.0 mL of this concentrate to 1000 mL
of using acid suppression technology.
with reagent water for the final 0.1 M working suppressor
7.6.5.1 Tri-Chamber Approach, 100 % solvent compatible
solution.The regenerate solution used may be different if other
tri-chamber micro packed bed with cation exchange resin (or
system or analytical columns are used.
equivalent). Suppressor device should simultaneously be on-
line continuously chemically regenerated to its hydrogen form NOTE 4—Other volumes of this solution can be prepared using
appropriate ratios of reagents. Follow the specific guidelines for this
using any mineral acid providing H+ ion.
7.6.5.2 Continuous Approach, The regenerant channels are
flushedwithamineralacid(sulfuric)tosupplyhydroniumions
ACS Reagent Chemicals, Specifications and Procedures for Reagents and
Standard-Grade Reference Materials, American Chemical Society, Washington,
required for the suppression reaction. The solvent compatible
DC. For suggestions on the testing of reagents not listed by theAmerican Chemical
ionexchangemembranesprovidethetransportpathwayforthe
Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset,
hydronium ions into the eluent channel and the transport of
U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharma-
sodium and other cations out of the eluent channel. copeial Convention, Inc. (USPC), Rockville, MD.
D7319 − 22
TABLE 1 Typical Volumetric Preparation of Chloride and Sulfate Standards in Water
Chloride and Sulfate Standards Chloride Stock Solution, Sulfate Stock Solution,
mg Chloride and Sulfate (each)/1 L water mL mL
-
50 Cl only 25 .
20 10 10
10 5 5
5 2.5 2.5
1 0.5 0.5
0.5 0.25 0.25
0.3 0.15 0.15
solution from the vendor of the suppressor being used.
where:
8.5 Sodium Sulfate—anhydrous, reagent grade, 99 % mini- gNa SO = weight in grams of Na SO dissolved in 1 L,
2 4 2 4
mum purity. (Warning—Do not ingest. Avoid unnecessary and
0.6764 = weight percent sulfate in Na SO .
exposure.)
2 4
8.6 Sodium Chloride—ACS or reagent grade, 99 % mini-
9.1.2 Chloride Stock Solution (approximately 2000 mg/
mum purity.
L)—To ensure dryness, place sodium chloride (5 g) in a drying
oven at 110 °C for at least an hour, cool, and store in a
8.7 Tetrabutylamonium Bisulfate—Technical or reagent
desiccator. Accurately weigh 3.30 g dried sodium chloride to
grade, 99 % minimum purity.
thenearesttenthofamilligramandtransfertoa1 Lvolumetric
8.8 Tetrabutylammonium Chloride—Technical or reagent
flask. Add water to dissolve the sodium chloride and make to
grade 97 % minimum purity.
volume. Calculate the concentration of chloride in the solution
8.9 Butanol—ACS, 99 % minimum purity. Could consist of
in accordance with Eq 2. Other volumes of stock solution can
isobutanol, sec butanol, or n-butanol. (Warning—Flammable,
be prepared using the appropriate ratio of reagents.
toxic, and may be harmful if ingested or inhaled. Affects
Stock Chloride ~mg/L! 5 ~g NaCl!~0.6068!~1000 mg/g!/1 L (2)
central nervous system similar to excessive alcohol exposure.)
where:
8.10 Ethanol—denatured with methanol, formula 3A or
histological grade ethanol, anhydrous, denatured with ethyl
g NaCl = weight in grams of NaCl dissolved in 1 L, and
acetate, methylisobutyl ketone and hydrocarbon naphtha.
0.6068 = weight percent chloride in NaCl.
(Warning—Flammable, toxic, and may be harmful or fatal if
9.2 Chloride and Sulfate Standards in Water—Add water
ingested or inhaled. Avoid skin contact.)
andsulfateandchloridestocksolutionsareaddedtoa1 Lglass
8.11 Hydrogen Peroxide—ACS reagent grade, 30 % by
volumetric flask in accordance with Table 1 to achieve the
weight in water.
desired standard.
8.12 Hydrogen Peroxide Solution, 30 %—commercially
9.2.1 Chloride and sulfate stock solutions from 9.1 are
available 30 % hydrogen peroxide solution.
added quantitatively into the flask and mixed quantitatively
with water in accordance with Table 1. Be very careful to
9. Preparation of Standard Solutions
measure the exact volumes of the sulfate and chloride stock
9.1 Stock Solutions: solutions that are added to the flask, and fill the flask to 1.00 L
9.1.1 Sulfate Stock Solution, approximately 2000 mg/L—To
with water. The sulfate and chloride concentrations of each
ensure dryness, place anhydrous sodium sulfate (5 g) in a standard are calculated in accordance with Eq 3 and Eq 4.
drying oven at 110 °C for at least an hour, cool, and store in a
Sulfate in Standard mg/L 5 V 3C /V (3)
~ !
a a
desiccator. Accurately weigh 2.96 g anhydrous sodium sulfate
Chloride in Standard mg/L 5 V 3C /V (4)
~ !
to the nearest tenth of a milligram and transfer to a 1 L b b
volumetric flask.Add water to dissolve the sodium sulfate, and
where:
make to volume. Calculate the concentration of sulfate in the
V = volume of sulfate stock solution (9.1.1), in mL,
a
solution in accordance with Eq 1. Other volumes of stock
C = concentration of sulfate stock (Eq 1), in mg/L,
a
solution can be prepared using the appropriate ratio of re-
V = volume of chloride stock solution (9.1.2)inmL,
b
agents.
C = concentration of chloride stock (Eq 2), in mg/L,
b
V = final volume of standard solution, in L.
Stock Sulfate ~mg/L! 5 ~gNa SO !~0.6764!~1000 mg/g!/1 L (1)
2 4
D7319 − 22
9.2.2 Multiples or fractions of Table 1 values can be used to
prepare different volumes of standards, but Eq 3 and Eq 4 are
still used to calculate standard ion concentrations.
NOTE 5—Alternatively, commercially available stock calibration solu-
tions can be used, provided that the solutions are traceable to primary
stock solutions or certified reference materials and are free from other
analytes.
10. Calibration
10.1 Set up the ion chromatograph in accordance with the
manufacturer’s instructions. No specific parameters are given
here since different equip
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D7319 − 17 D7319 − 22
Standard Test Method for
Determination of Existent and Potential Sulfate and
Inorganic Chloride in Fuel Ethanol and Butanol by Direct
Injection Suppressed Ion Chromatography
This standard is issued under the fixed designation D7319; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This test method covers a direct injection ion chromatographic procedure for determining existent and potential inorganic
sulfate and total inorganic chloride content in hydrous and anhydrous denatured ethanol and butanol to be used in motor fuel
applications. It is intended for the analysis of ethanol and butanol samples containing between 1.0 mg ⁄kg to 20 mg/kg of existent
or potential inorganic sulfate and 1.0 mg ⁄kg to 50 mg/kg of inorganic chloride.
NOTE 1—Tertiary butanol is not included in this test method. 1-butanol, 2-butanol, and isobutanol are included in the testing and research report for this
test method.
1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use. Material Safety Data Sheets are available for reagents and materials. Review them for hazards
prior to usage.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D1193 Specification for Reagent Water
D4052 Test Method for Density, Relative Density, and API Gravity of Liquids by Digital Density Meter
D4057 Practice for Manual Sampling of Petroleum and Petroleum Products
D4175 Terminology Relating to Petroleum Products, Liquid Fuels, and Lubricants
D4177 Practice for Automatic Sampling of Petroleum and Petroleum Products
D5827 Test Method for Analysis of Engine Coolant for Chloride and Other Anions by Ion Chromatography
D6299 Practice for Applying Statistical Quality Assurance and Control Charting Techniques to Evaluate Analytical Measure-
ment System Performance
This test method is under the jurisdiction of ASTM Committee D02 on Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of Subcommittee
D02.03 on Elemental Analysis.
Current edition approved Oct. 1, 2017Oct. 1, 2022. Published October 2017October 2022. Originally approved in 2007. Last previous edition approved in 20132017 as
D7319 – 13.D7319 – 17. DOI: 10.1520/D7319-17.10.1520/D7319-22.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D7319 − 22
D6792 Practice for Quality Management Systems in Petroleum Products, Liquid Fuels, and Lubricants Testing Laboratories
D7318 Test Method for Existent Inorganic Sulfate in Ethanol by Potentiometric Titration
D7328 Test Method for Determination of Existent and Potential Inorganic Sulfate and Total Inorganic Chloride in Fuel Ethanol
by Ion Chromatography Using Aqueous Sample Injection
2.2 European Norm Standards:
EN 15492 Ethanol as a blending component for petrol—Determination of inorganic chloride and sulfate content—Ion
chromatographic method
3. Terminology
3.1 Definitions:
3.1.1 For definitions of terms used in this test method, refer to Terminology D4175.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 existent inorganic sulfate, n—inorganic sulfate species actually present in the sample at the time of analysis with no oxidation
treatment.
3.2.2 inorganic chloride, n—chloride present as hydrochloric acid, ionic salts of this acid, or mixtures of these.
-2
3.2.3 inorganic sulfate, n—sulfate (SO ) species present as sulfuric acid, ionic salts of this acid, or mixtures of these.
3.2.4 potential sulfate, n—inorganic sulfate species present after the sample has been reacted with an oxidizing agent.
4. Summary of Test Method
4.1 For existent inorganic sulfate and total chloride, a small volume of an ethanol or butanol sample is directly injected into a
suitably configured ion chromatograph in accordance with manufacturer’s recommendations for this test method. For potential
sulfate, 0.5 mL of 30 % hydrogen peroxide solution is added to 9.5 mL of the ethanol or butanol sample, and then injected into
the ion chromatograph. Ions are separated based on their affinity for exchange sites of the resin with respect to the resin’s affinity
for the eluent. The suppressor increases the sensitivity of the test method by both increasing the conductivity of the analytes and
decreasing the conductivity of the eluent. It also converts the eluent and analytes to the corresponding hydrogen forms of anions.
Anions are quantified by integration of their responses compared with an external calibration curve, and are measured as
milligrams/litre, then converted to milligrams/kilograms. The calibration standards are prepared in an aqueous matrix.
4.1.1 The use of acid suppression technology is more significant for the precision of sulfate measurement than for chloride. Sulfate
precision will be worse if acid suppression is not used.
4.2 Similar methods for chloride and sulfate determinations can be found in EN 15492 for total chloride, in Test Method D7328
for total chloride and existent and potential inorganic sulfate by ion chromatography with aqueous sample injection, and in Test
Method D7318 for existent inorganic sulfate by potentiometric lead titration and Test Method D5827 for chloride and other anions
in engine coolants by ion chromatography.
NOTE 2—Butanol has limited solubility in water, do not dilute samples with water prior to analysis. In addition, any remnant water from the sample
delivery device or glassware may lead to low recoveries of sulfate and chloride. Avoid unintended water contamination of the sample. Added water can
lead to phase separation of the sample. The chloride and sulfate ions can migrate to the water dominate phase and lower the recovery from the butanol.
5. Significance and Use
5.1 Sulfates and chlorides can be found in filter plugging deposits and fuel injector deposits. The acceptability for use of the fuel
components and the finished fuels depends on the sulfate and chloride content.
5.2 Existent and potential inorganic sulfate and total chloride content, as measured by this test method, can be used as one measure
of the acceptability of gasoline components for automotive spark-ignition engine fuel use.
Available from American National Standards Institute (ANSI), 25 W. 43rd St., 4th Floor, New York, NY 10036, http://www.ansi.org.
D7319 − 22
NOTE 1—Chromatogram retention times may be different depending on instruments or columns used.
FIG. 1 Typical Ion Chromatogram of a Solution Containing 1 mg/L of Various Anions in Water
6. Interferences
6.1 Interferences can be caused by substances with similar ion chromatographic retention times, especially if they are in high
concentration compared to the analyte of interest. Sample dilution can be used to minimize or resolve most interference problems.
6.2 A water dip (system void, negative peak as shown in Fig. 1) can cause interference with some integrators. Usually, for chloride
and sulfate determinations, the water dip should not be a problem since the chloride and sulfate peaks are far away from the water
dip.
6.3 Given the trace amounts of chloride and sulfate determined in this test method, interferences can be caused by contamination
of glassware, eluent, reagents, and the like. For example, sodium leaching from glassware can precipitate with sulfate giving low
sulfate results. Take care to ensure that contamination is kept at the lowest possible levels. The use of powder-free gloves is highly
recommended to prevent sample contamination.
7. Apparatus
7.1 Analytical Balance, capable of weighing up to 100 g accurately to 0.0001 g.
7.2 Drying Oven, controlled at 110 °C 6 5 °C for drying sodium sulfate and sodium chloride.
7.3 Desiccator, containing freshly activated silica gel (or equivalent desiccant) with moisture content indicator.
7.4 Pipets or Volumetric Transferring Devices, appropriately sized Class A glass pipets or their equivalent or variable volume
automatic pipets fitted with disposable polypropylene tips.
7.5 Volumetric Flasks (with Glass Stoppers), Class A of 10 mL, 25 mL, 50 mL, 100 mL, 1000 mL, 2000 mL capacity.
7.5.1 Container, equipped with a closure to prevent ethanol evaporation, 1 L (for sample collection). See 6.3 for additional
information.
7.6 Ion Chromatograph, analytical system with all required accessories including syringes, columns, suppressor, and detector
which can be demonstrated to obtain the precision and detection limits and requirements of the method.
D7319 − 22
7.6.1 Injection System, capable of delivering 20 μL with a precision better than 1 %, or as recommended for this determination
by the manufacturer.
7.6.2 Pumping System, capable of delivering mobile phase flows between 0.2 mL ⁄min and 2.5 mL/min with a precision better than
2 %, or as recommended for this determination by the manufacturer.
7.6.3 Guard Column, for protection of the analytical column from strongly retained constituents. Better separations are obtained
with additional theoretical plates.
7.6.4 Anion Separator Column, 100 % solvent compatible and capable of producing satisfactory analyte (sulfate and chloride)
separation (Fig. 1).
7.6.5 Anion Suppressor Device, vendor specific and capable of using acid suppression technology.
7.6.5.1 Tri-Chamber Approach, 100 % solvent compatible tri-chamber micro packed bed with cation exchange resin (or
equivalent). Suppressor device should simultaneously be on-line continuously chemically regenerated to its hydrogen form using
any mineral acid providing H+ ion.
7.6.5.2 Continuous Approach, The regenerant channels are flushed with a mineral acid (sulfuric) to supply hydronium ions
required for the suppression reaction. The solvent compatible ion exchange membranes provide the transport pathway for the
hydronium ions into the eluent channel and the transport of sodium and other cations out of the eluent channel.
7.6.6 Conductivity Detector, (low volume), temperature controlled to 0.01 °C, capable of at least 0 μS ⁄cm to 1000 μS ⁄cm on a
linear scale.
7.6.7 Integrator or Chromatography Data System Software, capable of measuring peak areas and retention times, and correcting
the data in accordance with the baseline of the chromatogram.
7.7 Gloves, powder-free examination type.
8. Reagents
8.1 Purity of Reagents—Reagent grade or higher purity chemicals shall be used for the preparation of all samples, standards,
eluent, and regenerator solutions. Unless otherwise indicated, it is intended that all reagents conform to the specifications of the
Committee on Analytical Reagents of the American Chemical Society, where such specifications are available. Other grades may
be used, provided it is first ascertained that the reagent is of sufficiently high purity to permit its use without lessening the accuracy
of the determination.
8.2 Purity of Water—Unless otherwise indicated, reference to water shall be understood to mean reagent water as defined by Type
I or better in Specification D1193. For eluent preparation and handling, comply with all ion chromatograph instrument and column
vendor requirements (for example, filtering, degassing, and the like).
8.3 Eluent Buffer Stock Solution—Sodium bicarbonate (NaHCO ) 1.0 mM and sodium carbonate (Na CO ) 3.2 mM. Dissolve
3 2 3
8.4 g 6 0.0005 g of NaHCO and 33.92 g 6 0.0005 g of Na CO in reagent water in a 1 L Type A volumetric flask and dilute to
3 2 3
volume. Dilute 10.0 mL of this stock solution to 1 L in a 1 L Type A volumetric flask with degassed reagent water. The eluent
solution used may be different if other system or analytical columns are used.
NOTE 3—Other volumes of stock solution can be prepared using appropriate ratios of reagents. Follow the specific guidelines for this solution from the
vendor of the column being used. Alternatively, this solution can be purchased from a qualified vendor.
8.4 Suppressor Regenerant Solution for Suppressor—0.1 M sulfuric acid. Carefully add 334 mL of reagent grade sulfuric acid
Reagent Chemicals, American Chemical Society Specifications,ACS Reagent Chemicals, Specifications and Procedures for Reagents and Standard-Grade Reference
Materials, American Chemical Society, Washington, DC. For Suggestionssuggestions on the testing of reagents not listed by the American Chemical Society, see
AnnualAnalar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharmacopeial
Convention, Inc. (USPC), Rockville, MD.
D7319 − 22
(relative density 1.84) to approximately 500 mL reagent water in a 1 L volumetric flask. (Warning—This will generate a very hot
solution. Allow it to cool before diluting to 1000 mL volume. Never add water to concentrated acid.) Dilute to 1000 mL with
reagent water, and label this solution as 10.0 M sulfuric acid. Dilute 10.0 mL of this concentrate to 1000 mL with reagent water
for the final 0.1 M working suppressor solution. The regenerate solution used may be different if other system or analytical columns
are used.
NOTE 4—Other volumes of this solution can be prepared using appropriate ratios of reagents. Follow the specific guidelines for this solution from the
vendor of the suppressor being used.
8.5 Sodium Sulfate—anhydrous, reagent grade, 99 % minimum purity. (Warning—Do not ingest. Avoid unnecessary exposure.)
8.6 Sodium Chloride—ACS or reagent grade, 99 % minimum purity.
8.7 Tetrabutylamonium Bisulfate—Technical or reagent grade, 99 % minimum purity.
8.8 Tetrabutylammonium Chloride—Technical or reagent grade 97 % minimum purity.
8.9 Butanol—ACS, 99 % minimum purity. Could consist of isobutanol, sec butanol, or n-butanol. (Warning—Flammable, toxic,
and may be harmful if ingested or inhaled. Affects central nervous system similar to excessive alcohol exposure.)
8.10 Ethanol—denatured with methanol, formula 3A or histological grade ethanol, anhydrous, denatured with ethyl acetate,
methylisobutyl ketone and hydrocarbon naphtha. (Warning—Flammable, toxic, and may be harmful or fatal if ingested or inhaled.
Avoid skin contact.)
8.11 Hydrogen Peroxide—ACS reagent grade, 30 % by weight in water.
8.12 Hydrogen Peroxide Solution, 30 %—commercially available 30 % hydrogen peroxide solution.
9. Preparation of Standard Solutions
9.1 Stock Solutions:
9.1.1 Sulfate Stock Solution, approximately 2000 mg/L—To ensure dryness, place anhydrous sodium sulfate (5 g) in a drying oven
at 110 °C for at least an hour, cool, and store in a desiccator. Accurately weigh 2.96 g anhydrous sodium sulfate to the nearest tenth
of a milligram and transfer to a 1 L volumetric flask. Add water to dissolve the sodium sulfate, and make to volume. Calculate the
concentration of sulfate in the solution in accordance with Eq 1. Other volumes of stock solution can be prepared using the
appropriate ratio of reagents.
Stock Sulfate ~mg/L!5 ~g Na SO ! ~0.6764! ~1000 mg/g!/1 L (1)
2 4
D7319 − 22
TABLE 1 Typical Volumetric Preparation of Chloride and Sulfate Standards in Water
Chloride and Sulfate Standards Chloride Stock Solution, Sulfate Stock Solution,
mg Chloride and Sulfate (each)/1 L water mL mL
-
50 Cl only 25 .
20 10 10
10 5 5
5 2.5 2.5
1 0.5 0.5
0.5 0.25 0.25
0.3 0.15 0.15
where:
g Na SO = weight in grams of Na SO dissolved in 1 L, and
2 4 2 4
0.6764 = weight percent sulfate in Na SO .
2 4
9.1.2 Chloride Stock Solution (approximately 2000 mg/L)—To ensure dryness, place sodium chloride (5 g) in a drying oven at
110 °C for at least an hour, cool, and store in a desiccator. Accurately weigh 3.30 g dried sodium chloride to the nearest tenth of
a milligram and transfer to a 1 L volumetric flask. Add water to dissolve the sodium chloride and make to volume. Calculate the
concentration of chloride in the solution in accordance with Eq 2. Other volumes of stock solution can be prepared using the
appropriate ratio of reagents.
Stock Chloride mg/L 5 g NaCl 0.6068 1000 mg/g /1 L (2)
~ ! ~ ! ~ ! ~ !
where:
g NaCl = weight in grams of NaCl dissolved in 1 L, and
0.6068 = weight percent chloride in NaCl.
9.2 Chloride and Sulfate Standards in Water—Add water and sulfate and chloride stock solutions are added to a 1 L glass
volumetric flask in accordance with Table 1 to achieve the desired standard.
9.2.1 Chloride and sulfate stock solutions from 9.1 are added quantitatively into the flask and mixed quantitatively with water in
accordance with Table 1. Be very careful to measure the exact volumes of the sulfate and chloride stock solutions that are added
to the flask, and fill the flask to 1.00 L with water. The sulfate and chloride concentrations of each standard are calculated in
accordance with Eq 3 and Eq 4.
Sulfate in Standard ~mg/L!5 V 3C /V (3)
a a
Chloride in Standard ~mg/L!5 V 3C /V (4)
b b
where:
V = volume of sulfate stock solution (9.1.1), in mL,
a
C = concentration of sulfate stock (Eq 1), in mg/L,
a
V = volume of chloride stock solution (9.1.2) in mL,
b
C = concentration of chloride stock (Eq 2), in mg/L,
b
V = final volume of standard solution, in L.
9.2.2 Multiples or fractions of Table 1 values can be used to prepare different volumes of standards, but Eq 3 and Eq 4 are still
used to calculate standard ion concentrations.
NOTE 5—Alternatively, commercially available stock calibration solutions can be used, provided that the solutions are traceable to primary stock solutions
or certified reference materials and are free from other analytes.
10. Calibration
10.1 Set up the ion chromatograph in accordance with the manuf
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...