ASTM E1806-23
(Practice)Standard Practice for Sampling Steel and Iron for Determination of Chemical Composition
Standard Practice for Sampling Steel and Iron for Determination of Chemical Composition
SIGNIFICANCE AND USE
4.1 This practice covers all aspects of sampling and preparing steel and iron for chemical analysis as defined in Test Methods, Practices, and Definitions A751 and Specification A48/A48M. Such subjects as sampling location and the sampling of lots are defined.
4.2 This practice includes most requirements for sampling steel and iron for analysis. Standard test methods that reference this practice need contain only special modifications and exceptions.
4.3 All who use these procedures should be trained samplers capable of performing common sampling operations skillfully and safely. Only proper sampling equipment should be used.
SCOPE
1.1 This practice covers the sampling of all grades of steel, both cast and wrought, and all types (grades) of cast irons and blast furnace iron for chemical and spectrochemical determination of composition. This practice is similar to ISO 14284.
1.2 This practice is divided into the following sections.
Sections
Requirements for Sampling and Sample Preparation
6
General
6.1
Sample
6.2
Selection of a Sample
6.3
Preparation of a Sample
6.4
Liquid Iron for Steelmaking and Pig Iron Production
7
General
7.1
Spoon Sampling
7.2
Probe Sampling
7.3
Preparation of a Sample for Analysis
7.4
Liquid Iron for Cast Iron Production
8
General
8.1
Spoon Sampling
8.2
Probe Sampling
8.3
Preparation of a Sample for Analysis
8.4
Sampling and Sample Preparation for the Determination of
8.5
Oxygen and Hydrogen
Liquid Steel for Steel Production
9
General
9.1
Probe Sampling
9.2
Spoon Sampling
9.3
Preparation of a Sample for Analysis
9.4
Sampling and Sample Preparation for the Determination
9.5
of Oxygen
Sampling and Sample Preparation for the Determination
9.6
of Hydrogen
Pig Irons
10
General
10.1
Increment Sampling
10.2
Preparation of a Sample for Analysis
10.3
Cast Iron Products
11
General
11.1
Sampling and Sample Preparation
11.2
Sections
Steel Products
12
General
12.1
Selection of a Laboratory Sample or a Sample for
12.2
Analysis from a Cast Product
Selection of a Laboratory Sample or a Sample for
12.3
Analysis from a Wrought Product
Preparation of a Sample for Analysis
12.4
Sampling of Leaded Steel
12.5
Sampling and Sample Preparation for the Determination
12.6
of Oxygen
Sampling and Sample Preparation for the Determination
12.7
of Hydrogen
Keywords
13
Annexes
Sampling Probes for Use with Liquid Iron and Steel
Annex A1
Sampling Probes for Use with Liquid Steel for the
Annex A2
Determination of Hydrogen
1.3 The values stated in SI units are regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific statements, see 6.4.3.5, 9.4.4.3, 12.5.1, and Section 5.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-May-2023
- Technical Committee
- E01 - Analytical Chemistry for Metals, Ores, and Related Materials
- Drafting Committee
- E01.01 - Iron, Steel, and Ferroalloys
Relations
- Effective Date
- 01-Jan-2020
- Effective Date
- 15-May-2019
- Effective Date
- 15-May-2016
- Effective Date
- 01-Jul-2015
- Effective Date
- 15-May-2015
- Effective Date
- 15-Aug-2014
- Effective Date
- 01-Apr-2014
- Effective Date
- 01-Mar-2014
- Effective Date
- 01-Mar-2014
- Effective Date
- 15-Feb-2014
- Effective Date
- 01-Dec-2013
- Effective Date
- 01-Oct-2012
- Effective Date
- 01-Nov-2011
- Effective Date
- 15-Sep-2011
- Effective Date
- 15-Jun-2011
Overview
ASTM E1806-23, titled Standard Practice for Sampling Steel and Iron for Determination of Chemical Composition, is a comprehensive guideline developed by ASTM International. This standard covers all procedures related to sampling and preparing both steel and iron-regardless of grade or production method-for subsequent chemical and spectrochemical analysis. By following ASTM E1806-23, users ensure rigor, repeatability, and reliability in determining chemical composition, which is essential for manufacturing, quality assurance, and materials research.
The standard applies to all grades of cast and wrought steel, cast irons, and blast furnace iron. It addresses sampling methods in various production settings, ensuring representativeness and integrity of the analytical sample. ASTM E1806-23 is closely aligned with ISO 14284, promoting international harmonization.
Key Topics
Sampling Locations and Lots
- Defines best practices for where and how to sample within production runs or lots.
- Ensures representative sampling for accurate chemical composition analysis.
Sample Types and Preparation
- Outlines the selection and preparation of samples-from liquid iron and steel to solid products.
- Covers methods such as spoon sampling, probe sampling, and incremental sampling.
- Guidance on removal of surface contamination, use of suitable sampling tools, and proper labeling.
Applicable Materials
- Applies to all forms of steel (cast and wrought), all grades of cast irons, and pig iron.
Safety and Skill
- Emphasizes the requirement for trained personnel and proper use of sampling equipment.
- Notes the need for suitable personal protective equipment and safe sampling procedures.
Sample Homogeneity and Integrity
- Ensures samples are sufficiently homogeneous in chemical composition.
- Provides instruction to avoid contamination, loss, or alteration during and after sampling.
Applications
Adhering to ASTM E1806-23 is critical in several industrial contexts, including but not limited to:
Steel and Iron Production
- Used to monitor and control chemical composition during steelmaking, iron casting, and pig iron production.
- Supports process validation and product certification.
Laboratory Analysis
- Ensures labs receive representative and uncontaminated samples for accurate analysis by atomic emission spectrometry, X-ray fluorescence, and other analytical methods.
Quality Assurance
- Guarantees that products meet required specifications, including compliance with customer or regulatory requirements.
Material Certification and Dispute Resolution
- Provides clear guidelines for arbitration samples jointly selected and sealed by involved parties.
Global Trade
- Facilitates international commerce by providing a harmonized approach for sampling, complementary to globally recognized standards like ISO 14284.
Related Standards
For comprehensive implementation and cross-referencing within industry and laboratory practices, consider the following standards:
- ASTM A751 - Test Methods and Practices for Chemical Analysis of Steel Products
- ASTM A48/A48M - Specification for Gray Iron Castings
- ASTM E415 - Test Method for Analysis of Carbon and Low-Alloy Steel by Spark Atomic Emission Spectrometry
- ASTM E1010/E1306 - Practice for Preparation of Disk Specimens by Electric Arc Melting (for spectrochemical analysis)
- ASTM E135 - Terminology Relating to Analytical Chemistry for Metals, Ores, and Related Materials
- ISO 14284 - Steel and Iron–Sampling and Preparation of Samples for Determination of Chemical Composition
Practical Value
ASTM E1806-23 helps ensure valid, consistent, and safe sampling of steel and iron for chemical determination, resulting in analytical results that support process control, material traceability, and industry best practice. Its adoption benefits manufacturers, testing laboratories, and buyers seeking high-quality, standardized sampling methods for ferrous metals. By following this standard, organizations can reduce variability, improve data quality, and facilitate international and domestic trade in steel and iron products.
Keywords: ASTM E1806-23, sampling steel, sampling iron, chemical composition, sample preparation, metallurgical analysis, quality assurance, ISO 14284, steelmaking standards.
Buy Documents
ASTM E1806-23 - Standard Practice for Sampling Steel and Iron for Determination of Chemical Composition
REDLINE ASTM E1806-23 - Standard Practice for Sampling Steel and Iron for Determination of Chemical Composition
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM E1806-23 is a standard published by ASTM International. Its full title is "Standard Practice for Sampling Steel and Iron for Determination of Chemical Composition". This standard covers: SIGNIFICANCE AND USE 4.1 This practice covers all aspects of sampling and preparing steel and iron for chemical analysis as defined in Test Methods, Practices, and Definitions A751 and Specification A48/A48M. Such subjects as sampling location and the sampling of lots are defined. 4.2 This practice includes most requirements for sampling steel and iron for analysis. Standard test methods that reference this practice need contain only special modifications and exceptions. 4.3 All who use these procedures should be trained samplers capable of performing common sampling operations skillfully and safely. Only proper sampling equipment should be used. SCOPE 1.1 This practice covers the sampling of all grades of steel, both cast and wrought, and all types (grades) of cast irons and blast furnace iron for chemical and spectrochemical determination of composition. This practice is similar to ISO 14284. 1.2 This practice is divided into the following sections. Sections Requirements for Sampling and Sample Preparation 6 General 6.1 Sample 6.2 Selection of a Sample 6.3 Preparation of a Sample 6.4 Liquid Iron for Steelmaking and Pig Iron Production 7 General 7.1 Spoon Sampling 7.2 Probe Sampling 7.3 Preparation of a Sample for Analysis 7.4 Liquid Iron for Cast Iron Production 8 General 8.1 Spoon Sampling 8.2 Probe Sampling 8.3 Preparation of a Sample for Analysis 8.4 Sampling and Sample Preparation for the Determination of 8.5 Oxygen and Hydrogen Liquid Steel for Steel Production 9 General 9.1 Probe Sampling 9.2 Spoon Sampling 9.3 Preparation of a Sample for Analysis 9.4 Sampling and Sample Preparation for the Determination 9.5 of Oxygen Sampling and Sample Preparation for the Determination 9.6 of Hydrogen Pig Irons 10 General 10.1 Increment Sampling 10.2 Preparation of a Sample for Analysis 10.3 Cast Iron Products 11 General 11.1 Sampling and Sample Preparation 11.2 Sections Steel Products 12 General 12.1 Selection of a Laboratory Sample or a Sample for 12.2 Analysis from a Cast Product Selection of a Laboratory Sample or a Sample for 12.3 Analysis from a Wrought Product Preparation of a Sample for Analysis 12.4 Sampling of Leaded Steel 12.5 Sampling and Sample Preparation for the Determination 12.6 of Oxygen Sampling and Sample Preparation for the Determination 12.7 of Hydrogen Keywords 13 Annexes Sampling Probes for Use with Liquid Iron and Steel Annex A1 Sampling Probes for Use with Liquid Steel for the Annex A2 Determination of Hydrogen 1.3 The values stated in SI units are regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific statements, see 6.4.3.5, 9.4.4.3, 12.5.1, and Section 5. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 This practice covers all aspects of sampling and preparing steel and iron for chemical analysis as defined in Test Methods, Practices, and Definitions A751 and Specification A48/A48M. Such subjects as sampling location and the sampling of lots are defined. 4.2 This practice includes most requirements for sampling steel and iron for analysis. Standard test methods that reference this practice need contain only special modifications and exceptions. 4.3 All who use these procedures should be trained samplers capable of performing common sampling operations skillfully and safely. Only proper sampling equipment should be used. SCOPE 1.1 This practice covers the sampling of all grades of steel, both cast and wrought, and all types (grades) of cast irons and blast furnace iron for chemical and spectrochemical determination of composition. This practice is similar to ISO 14284. 1.2 This practice is divided into the following sections. Sections Requirements for Sampling and Sample Preparation 6 General 6.1 Sample 6.2 Selection of a Sample 6.3 Preparation of a Sample 6.4 Liquid Iron for Steelmaking and Pig Iron Production 7 General 7.1 Spoon Sampling 7.2 Probe Sampling 7.3 Preparation of a Sample for Analysis 7.4 Liquid Iron for Cast Iron Production 8 General 8.1 Spoon Sampling 8.2 Probe Sampling 8.3 Preparation of a Sample for Analysis 8.4 Sampling and Sample Preparation for the Determination of 8.5 Oxygen and Hydrogen Liquid Steel for Steel Production 9 General 9.1 Probe Sampling 9.2 Spoon Sampling 9.3 Preparation of a Sample for Analysis 9.4 Sampling and Sample Preparation for the Determination 9.5 of Oxygen Sampling and Sample Preparation for the Determination 9.6 of Hydrogen Pig Irons 10 General 10.1 Increment Sampling 10.2 Preparation of a Sample for Analysis 10.3 Cast Iron Products 11 General 11.1 Sampling and Sample Preparation 11.2 Sections Steel Products 12 General 12.1 Selection of a Laboratory Sample or a Sample for 12.2 Analysis from a Cast Product Selection of a Laboratory Sample or a Sample for 12.3 Analysis from a Wrought Product Preparation of a Sample for Analysis 12.4 Sampling of Leaded Steel 12.5 Sampling and Sample Preparation for the Determination 12.6 of Oxygen Sampling and Sample Preparation for the Determination 12.7 of Hydrogen Keywords 13 Annexes Sampling Probes for Use with Liquid Iron and Steel Annex A1 Sampling Probes for Use with Liquid Steel for the Annex A2 Determination of Hydrogen 1.3 The values stated in SI units are regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific statements, see 6.4.3.5, 9.4.4.3, 12.5.1, and Section 5. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E1806-23 is classified under the following ICS (International Classification for Standards) categories: 71.040.50 - Physicochemical methods of analysis. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E1806-23 has the following relationships with other standards: It is inter standard links to ASTM E135-20, ASTM E135-19, ASTM E135-16, ASTM E135-15a, ASTM E135-15, ASTM E135-14b, ASTM E135-14a, ASTM A751-14, ASTM E415-14, ASTM E135-14, ASTM E135-13a, ASTM A48/A48M-03(2012), ASTM E1306-11, ASTM E135-11b, ASTM E135-11a. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E1806-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E1806 − 23
Standard Practice for
Sampling Steel and Iron for Determination of Chemical
Composition
This standard is issued under the fixed designation E1806; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
Sections
1.1 This practice covers the sampling of all grades of steel,
Steel Products 12
both cast and wrought, and all types (grades) of cast irons and General 12.1
Selection of a Laboratory Sample or a Sample for 12.2
blast furnace iron for chemical and spectrochemical determi-
Analysis from a Cast Product
nation of composition. This practice is similar to ISO 14284.
Selection of a Laboratory Sample or a Sample for 12.3
Analysis from a Wrought Product
1.2 This practice is divided into the following sections.
Preparation of a Sample for Analysis 12.4
Sampling of Leaded Steel 12.5
Sections
Requirements for Sampling and Sample Preparation 6 Sampling and Sample Preparation for the Determination 12.6
of Oxygen
General 6.1
Sample 6.2
Sampling and Sample Preparation for the Determination 12.7
Selection of a Sample 6.3
of Hydrogen
Preparation of a Sample 6.4
Keywords 13
Liquid Iron for Steelmaking and Pig Iron Production 7
General 7.1
Annexes
Spoon Sampling 7.2
Probe Sampling 7.3 Sampling Probes for Use with Liquid Iron and Steel Annex A1
Sampling Probes for Use with Liquid Steel for the Annex A2
Preparation of a Sample for Analysis 7.4
Determination of Hydrogen
Liquid Iron for Cast Iron Production 8
1.3 The values stated in SI units are regarded as standard.
General 8.1
No other units of measurement are included in this standard.
Spoon Sampling 8.2
Probe Sampling 8.3
1.4 This standard does not purport to address all of the
Preparation of a Sample for Analysis 8.4
safety concerns, if any, associated with its use. It is the
Sampling and Sample Preparation for the Determination of 8.5
Oxygen and Hydrogen
responsibility of the user of this standard to establish appro-
priate safety, health, and environmental practices and deter-
Liquid Steel for Steel Production 9
mine the applicability of regulatory limitations prior to use.
General 9.1
Probe Sampling 9.2
For specific statements, see 6.4.3.5, 9.4.4.3, 12.5.1, and Section
Spoon Sampling 9.3
5.
Preparation of a Sample for Analysis 9.4
Sampling and Sample Preparation for the Determination 9.5 1.5 This international standard was developed in accor-
of Oxygen
dance with internationally recognized principles on standard-
Sampling and Sample Preparation for the Determination 9.6
ization established in the Decision on Principles for the
of Hydrogen
Development of International Standards, Guides and Recom-
Pig Irons 10
mendations issued by the World Trade Organization Technical
General 10.1
Barriers to Trade (TBT) Committee.
Increment Sampling 10.2
Preparation of a Sample for Analysis 10.3
2. Referenced Documents
Cast Iron Products 11
2.1 ASTM Standards:
General 11.1
Sampling and Sample Preparation 11.2
A48/A48M Specification for Gray Iron Castings
A751 Test Methods and Practices for Chemical Analysis of
Steel Products
This practice is under the jurisdiction of ASTM Committee E01 on Analytical
Chemistry for Metals, Ores, and Related Materials and is the direct responsibility of
Subcommittee E01.01 on Iron, Steel, and Ferroalloys. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved June 1, 2023. Published July 2023. Originally approved contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
in 1996. Last previous edition approved in 2018 as E1806 – 18. DOI: 10.1520/ Standards volume information, refer to the standard’s Document Summary page on
E1806-23. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E1806 − 23
E135 Terminology Relating to Analytical Chemistry for 3.2.14 test portion, n—part of the sample for analysis, or
Metals, Ores, and Related Materials part of the sample taken from the melt, actually analyzed.
E415 Test Method for Analysis of Carbon and Low-Alloy 3.2.14.1 Discussion—Sometimes, the test portion may be
Steel by Spark Atomic Emission Spectrometry selected from the sample product itself.
E1306 Practice for Preparation of Metal and Alloy Samples
3.2.15 thermal method of analysis, n—method for the de-
by Electric Arc Melting for Spectrochemical Analysis
termination of chemical composition in which the sample is
submitted to a process of heating, combustion, or fusion.
2.2 ISO Standards:
ISO 9147 Pig irons–Definition and classification
3.2.16 wrought product, n—item of steel which has been
ISO 14284 Steel and iron—Sampling and preparation of
subject to deformation by rolling, drawing, forging, or some
samples for the determination of chemical composition
other method, for example, a bar, billet, plate, strip, tube, or
wire.
3. Terminology
4. Significance and Use
3.1 Definitions—For definitions of terms in this practice,
refer to Terminology E135.
4.1 This practice covers all aspects of sampling and prepar-
3.2 Definitions of Terms Specific to This Standard:
ing steel and iron for chemical analysis as defined in Test
3.2.1 cast product, n—item of iron or steel which has not
Methods, Practices, and Definitions A751 and Specification
been subject to deformation, for example, an ingot, a semi
A48/A48M. Such subjects as sampling location and the sam-
finished product obtained by continuous casting, or a shaped
pling of lots are defined.
casting.
4.2 This practice includes most requirements for sampling
3.2.2 consignment, n—quantity of metal delivered at one
steel and iron for analysis. Standard test methods that reference
time.
this practice need contain only special modifications and
exceptions.
3.2.3 grinding, n—method of preparing a sample of metal
for a spectrochemical method of analysis in which the surface
4.3 All who use these procedures should be trained samplers
of the sample is abraded using an abrasive wheel.
capable of performing common sampling operations skillfully
3.2.4 linishing, n—method of preparing a sample of metal and safely. Only proper sampling equipment should be used.
for a spectrochemical method of analysis in which the surface
5. Hazards and Safety Precautions
of the sample is abraded using a rotating disk or belt coated
with an abrasive substance.
5.1 Provide personal protective equipment to minimize the
risk of injury during sampling and sample preparation meth-
3.2.5 melt, n—liquid metal from which a sample is re-
ods. Provisions shall include protective clothing, hand
moved.
protection, and face visors resistant to splashes of liquid metal
3.2.6 sample, probe, n—sample taken from the melt using a
for use during the sampling of liquid metal; protective clothing
sampling probe.
and hand, eye, and hearing protection for use during the
3.2.7 sample, product, n—specific item of iron or steel
sampling and sample preparation of solid metal; and respira-
selected from a supplied quantity for the purpose of obtaining
tory protection for use where necessary.
a sample.
5.2 The use of machinery for sampling and sample prepa-
3.2.8 sample, spoon, n—sample taken from the melt using a
ration shall be in accordance with appropriate national stan-
spoon and cast into a small mold.
dards. Grinding operations used for surface preparation may be
3.2.9 sampling, immersion, n—method of probe sampling in
covered by national legislation.
which the probe is immersed in the melt and the sample
5.3 Refer to appropriate national regulations regarding the
chamber in the probe fills by ferrostatic pressure or gravity.
use of solvents for cleaning and drying of samples and test
3.2.10 sampling, probe, n—method in which the sample is
portions.
taken using a sampling probe inserted into the melt.
6. Requirements for Sampling and Sample Preparation
3.2.11 sampling, spoon, n—method in which the sample is
taken from the melt, or during the pouring of the melt, using a
6.1 General:
long-handled spoon and cast into a small mold.
6.1.1 This section describes the general requirements for the
3.2.12 sampling, stream, n—method of probe sampling in sample and for the sampling and sample preparation of iron
and steel. Special requirements apply to each category of liquid
which the probe is inserted into a stream of liquid metal and the
sample chamber in the probe fills by force of metal flow. and solid metal, and these requirements are described in the
relevant section.
3.2.13 sampling, suction, n—method of probe sampling in
6.1.2 The sequence of sampling and sample preparation of
which the probe is inserted into the melt and the sample
liquid iron and steel and cast iron and steel products is shown
chamber in the probe fills by aspiration.
in Fig. 1. Special considerations apply to pig irons (see Section
10).
6.2 Sample:
Available from American National Standards Institute (ANSI), 25 W. 43rd St.,
4th Floor, New York, NY 10036, http://www.ansi.org. 6.2.1 Quality:
E1806 − 23
FIG. 1 Sequence of Sampling and Sample Preparation of (a) Liquid Iron and Steel and (b) Cast Iron and Steel Products
6.2.1.1 Sampling practices shall be designed to provide an 6.2.1.6 A sample taken from a melt shall be cooled in such
analytical sample that is representative of the mean chemical a manner that the chemical composition and metallurgical
composition of the melt or of the sample product. structure of the sample is consistent from sample to sample.
6.2.1.2 Ensure that the sample is sufficiently homogeneous 6.2.1.7 It is important to recognize that analysis by some
regarding chemical composition so that inhomogeneity does spectrochemical methods may be influenced by the metallur-
not appreciably contribute to the error variability of the method gical structure of the sample, particularly for irons with white
of analysis. For a sample taken from a melt, however, some and grey iron structures, and steels in the as-cast and wrought
variability in analysis, both within and between samples, may conditions.
be unavoidable. This variability will form an inherent part of 6.2.2 Size:
the repeatability and reproducibility of the analysis. 6.2.2.1 Ensure that the dimensions of a laboratory sample in
6.2.1.3 Ensure that the sample is free from surface coatings the form of a solid mass are sufficient to permit additional
and from moisture, dirt, or other forms of contamination. samples to be taken for reanalysis when necessary using an
6.2.1.4 The sample should be free from voids, cracks, and alternative method of analysis.
porosity and from fins, laps, or other surface defects. 6.2.2.2 A sample shall be prepared consisting of a sufficient
6.2.1.5 Take particular care when selecting and preparing mass to provide for any reanalysis necessary. Generally, a mass
the sample if a sample taken from a melt is expected to be of 100 g will be sufficient for a sample in the form of chips or
heterogeneous or contaminated in any way. powder.
E1806 − 23
include dimensions for information only.
6.2.2.3 The dimensional requirements for a sample in the
form of a solid mass will depend upon the method selected for
6.3.2 Sample from a Product:
analysis. For spark atomic emission and X-ray fluorescence
6.3.2.1 The laboratory sample can be selected from the
spectrometric methods, the shape and size of the sample will be
sample product at the location indicated in the product speci-
determined by the dimensions of the sample chamber. The
fication for the selection of material for mechanical testing
dimensions for samples given in this practice are for guidance
when available.
only. Refer to Test Method E415 for sample size requirements
6.3.2.2 For an iron casting, the sample can be selected from
for spark atomic emission spectrometry.
a bar or block cast-on to the casting.
6.2.3 Identification:
6.3.2.3 For a forging, the sample can be selected from the
6.2.3.1 Assign the sample a unique identification to deter-
initial starting material from which the forging has been made
mine the melt or sample product from which it was taken, and
or from prolongations of the forging or from additional
if necessary, the process conditions of the melt or the location
forgings.
of the laboratory sample in the sample product. For pig iron,
6.3.2.4 In the absence of requirements given in the product
this shall identify the consignment or part of a consignment and
standard or of a specification when ordering the product, the
the increment from which it was taken.
sample may, following agreement between the supplier and the
6.2.3.2 Use labeling or some equivalent method of marking
purchaser, be selected from the sample for mechanical testing
to ensure that the assigned identification remains associated
or from the test piece, or directly from the sample product.
with the sample.
6.3.2.5 The laboratory sample can be obtained from the
6.2.3.3 Record the identification of the sample to ensure that
sample product by machining or by using a cutting torch.
confusion is prevented regarding the identity of the item to
Special considerations apply to sampling for the determination
which the analysis and records refer.
of certain elements.
6.2.4 Conservation:
6.4 Preparation of a Sample:
6.2.4.1 Provide adequate storage facilities to segregate and
6.4.1 Preliminary Preparation:
protect the sample. During and after preparation, store the
6.4.1.1 If any part of the sample is potentially nonrepresen-
sample in a manner which prevents contamination or chemical
tative in chemical composition, remove those parts that have
change.
changed. Following this operation, the sample shall be pro-
6.2.4.2 Keep the sample, or the laboratory sample in a solid
tected from any change in composition.
mass form, for a sufficient period of time to permit retesting if
6.4.1.2 Remove any coating that has been applied during
necessary.
manufacturing. If necessary, degrease the surface of the metal
6.2.5 Arbitration:
with a suitable solvent, taking care to ensure that the manner of
6.2.5.1 Samples intended for arbitration shall be prepared
degreasing does not affect the accuracy of analysis.
jointly by the supplier and purchaser or by their representa-
6.4.2 Sample in the Form of Chips:
tives. Keep a record of the methods used for preparing samples.
6.4.2.1 Obtain the sample by drilling, milling or turning to
6.2.5.2 Both parties or their representatives shall seal con-
produce chips of a consistent size and shape. Do not machine
tainers with the samples intended for arbitration. Unless agreed
on a part of the sample that has been affected by the heat of a
to the contrary, the representatives of each party responsible for
cutting torch.
the preparation of samples will keep these containers.
6.4.2.2 Before preparation, clean the tools, machines, and
6.3 Selection of a Sample:
containers to be used during preparation to prevent any
6.3.1 Sample from a Melt:
contamination of the sample.
6.3.1.1 Melts are sampled at various stages in the manufac-
6.4.2.3 Machine in such a way that the chips are not subject
turing process for the purposes of monitoring and controlling
to overheating as indicated by a change in the color (blueing or
the process. Samples can be taken during the casting of the blackening). Unavoidable coloration of chips obtained from
melt to verify chemical composition in accordance with the
some types of alloy steels, for example, manganese and
specification of the cast product. For liquid metal intended for austenitic steels, can be minimized by selection of appropriate
the production of a casting, the analytical sample may be
tools and cutting speeds.
selected from test bars or blocks specially cast from the same
6.4.2.4 Heat treatment may be required to soften the sample
metal as that of the casting for purposes of mechanical testing
for machining.
in accordance with the product standard.
6.4.2.5 The use of coolants during machining is only per-
6.3.1.2 Sampling practices shall be designed for melts to
mitted in exceptional cases, after which the chips shall be
provide samples during a particular manufacturing process in
cleaned by means of a suitable solvent that does not leave any
accordance with requirements for sample quality (see 6.2.1).
residue.
The sample obtained from a melt is usually a small ingot, a
6.4.2.6 Mix the chips thoroughly before weighing the test
cylindrical or rectangular block, or a chill-cast disk or a
portion. For most purposes, it is satisfactory to mix the chips by
combination of a disk with one or more attached pins.
rolling the container on a level surface, gently tumbling the
Sometimes small lugs are attached to a disk sample.
chips.
6.4.3 Sample in the Form of a Powder or Fragments:
NOTE 1—Sampling probes for use with liquid iron and steel may be
6.4.3.1 If drilling the sample to obtain chips is
obtained from a number of suppliers. The main features of the different
types of probe are distinguished in Annex A1 and Annex A2, which impracticable, cut or break the sample into pieces. Crush the
E1806 − 23
pieces using a percussion mortar or a vibratory grinding mill, samples (see A2.3) and for the punching of slugs as test
also known as a disk mill or ring mill, to obtain a sample in the portions may incorporate operations for the sandblasting of the
form of a powder the whole of which passes through a sieve of sample and for heat treatment to soften the sample before
a specified aperture size. punching.
6.4.3.2 In some applications the sample is crushed in a
6.4.5.7 Select the abrasive substances used in the final stage
percussion mortar to obtain a sample in the form of fragments
of preparing the sample that do not contaminate the surface
suitable for use with the selected method of analysis.
with elements that are to be determined by the analysis method.
6.4.3.3 Equipment used for pulverization shall be con-
Ensure that the grit size of the abrasive is in accordance with
structed from material that does not alter the sample compo-
the grade of surface finish required for the analysis method.
sition. Tests may be necessary to show that such equipment
6.4.5.8 For spark atomic emission spectrometric methods,
does not affect the composition of the sample.
an abrasive with a size of 60 grit to 120 grit normally is
6.4.3.4 Sieve the sample taking precautions to avoid con-
suitable. For X-ray fluorescence spectrometric methods, it is
tamination or loss of material. Take care when sieving hard
essential to ensure that the method selected for surface prepa-
materials to avoid damaging the fabric of the sieve.
ration produces a grade of surface finish that is sufficiently
6.4.3.5 Homogenize the sample before weighing the test
smooth and reproducible from sample to sample. Also, there
portion. Powders can be homogenized by stirring. (Warning—
should be no smearing of the surface.
Finely divided metals of particle size less than approximately
6.4.5.9 The effect of abrasive materials depends on the
150 μm can present a fire risk. Ensure adequate ventilation
analytical method. When using spark atomic emission spectro-
during pulverization.)
metric methods, the action of pre-sparking normally will clean
6.4.4 Sample in the Form of a Solid Mass:
the surface of the sample for analysis by volatilizing any
6.4.4.1 Obtain the sample by cutting from the sample
grinding contaminants. Particular care is required, however, to
product or laboratory sample a piece of suitable size and shape
avoid surface contamination when using new abrasive media.
for the method of analysis. Cut samples by sawing, abrasive
6.4.5.10 When using X-ray fluorescence spectrometric
cutting, shearing, or punching.
methods, examine all phases of surface preparation for poten-
6.4.4.2 In the absence of any indication in the product
tial surface contamination effects.
standard, perform a spectrochemical analysis on that part of the
6.4.5.11 Examine the sample visually after preparation to
sample corresponding to a transverse section of the product,
establish that the surface is dry and free from particulate matter
provided that the material has sufficient thickness.
and that there are no defects. Resurface or discard the sample
6.4.4.3 Prepare the sample to expose a surface suitable for
if defects are present. Protect the prepared surface from
the analytical method. Do not prepare a surface for analysis on
contamination.
part of a sample that has been affected by the heat of a cutting
6.4.6 Preparation of a Sample for Analysis by Melting:
torch. The equipment used for sample preparation shall be
6.4.6.1 A sample in the form of small pieces or chips, or a
designed to minimize overheating the sample, and, where
part of the sample product itself, can be melted in an atmo-
appropriate, shall incorporate systems of cooling.
sphere of argon using commercially available melting equip-
6.4.5 Main Types of Equipment Used for Surface Prepara-
ment of the type described in Practice E1306. The sample is
tion:
converted into a disk, 40-mm to 30-mm diameter by 6 mm
6.4.5.1 A milling machine capable of removing a prese-
thick, which is suitable for analysis by a spectrochemical
lected depth of metal in a reproducible manner, for use with
method. Some types of melting equipment incorporate opera-
samples that are within a hardness range suitable for milling.
tions for the centrifugal casting of the disk sample.
The equipment should be capable of use, if required, with a
6.4.6.2 Partial losses of some elements can be experienced
sample taken from a melt while the sample is still hot.
during the melting process. It is essential to ensure that any
6.4.5.2 A grinding machine with a fixed, rotating, or oscil-
selective volatilization or segregation of elements, or any other
lating head capable of removing a preselected depth of metal in
change in composition, which occur are known quantitatively
a reproducible manner.
and do not significantly influence the analysis results. Conduct
6.4.5.3 A flatbed linishing machine with abrasive grinding
suitable tests to show that any change in composition is both
disks, or machines with continuous abrasive belts, capable of
small in magnitude and reproducible.
preparing the surface of the sample to varying grades of finish.
6.4.6.3 The equipment used and the method adopted for
6.4.5.4 A machine for blasting with sand or grit, or metal
shot, capable of cleaning the surface of the sample or the test melting shall be designed to prevent or minimize change in
composition and to ensure that any change is reproducible. A
portion when required.
deoxidant, for example 0.1 % (m ⁄m) zirconium, should be
6.4.5.5 Ensure that the surface of the sample after prepara-
used during melting. Consider any change that does occur
tion is flat and free from defects that could affect accuracy of
when using the method for calibration of analytical measure-
analysis.
ment.
6.4.5.6 Cutting and surface preparation can be performed
either manually or automatically. For samples taken from 6.4.6.4 Not all ferrous metals can be melted in this manner.
melts, commercially available systems may be used that Do not use melting as a method of sample preparation for the
perform each stage of preparation automatically. Systems for determination of an element that is subject to a significant and
the automatic surface preparation of dualthickness probe nonreproducible change in composition.
E1806 − 23
7. Liquid Iron for Steelmaking and Pig Iron Production
7.1 General:
7.1.1 The following methods are applicable for sampling
liquid blast-furnace iron intended for steelmaking, and com-
monly described as hot metal, or for the casting of pig iron. The
liquid iron is normally sampled from the blast furnace runner
during the pouring of the melt into torpedo ladles, or from
transfer vessels or during secondary treatment processes in the
ladle, or during the casting of the melt into pig iron.
7.1.2 The chemical composition of iron may fluctuate dur-
ing run-out from the blast furnace. Two or more samples
should be taken from the melt at timed intervals and an average
analysis determined.
7.1.3 When spectrochemical methods are used for analysis,
the method of sampling should be designed to chill the liquid
metal in a manner that ensures that the metallurgical structure
of the sample is suitable for the requirements of the analysis
method selected.
7.2 Spoon Sampling: All dimensions are in millimetres.
The flat chill plate (not shown) has similar overall dimensions.
7.2.1 Hold Types:
FIG. 2 Combination-Type Vertical Mold Used for Sampling Liquid
7.2.1.1 A disk-shaped sample, commonly described as a
Iron Intended for Cast Iron Production
coin sample, can be obtained using a two-piece steel mold. The
dimensions of the sample are typically 35 mm to 40 mm in
7.2.3.1 It is essential to maintain sampling spoons and
diameter with thickness varying from 6 mm to 12 mm. The
molds in a clean and dry condition. After use, remove any slag
mold is constructed in two pieces that are clamped together in
and skull and brush the faces of the mold with a wire brush.
use: one piece is a flat chill plate and the other is a block with
7.2.3.2 Molds should be re-machined if the internal surfaces
the mold cavity. The edge of the mold cavity may be tapered,
become worn. This avoids the need for additional machining of
for example, from 38 mm to 32 mm, to facilitate removal of the
the sample during surface preparation.
sample from the mold.
7.2.1.2 A coin sample with one or more attached pins can be
7.3 Probe Sampling:
obtained using a combination-type mold. The pins are broken
7.3.1 General:
off from the disk and used, if required, as test portions for
7.3.1.1 The different types of probes used for sampling blast
analysis by a thermal method. A combination-type mold for use
furnace iron are described in Annex A1. Probes are designed to
with liquid iron intended for the production of cast iron is
provide a disk-shaped sample with a depth of white iron
shown in Fig. 2.
structure that is sufficient for the requirements of the spectro-
7.2.1.3 A thin slab-shaped sample with a rounded end can
chemical method selected for analysis.
be obtained using a cast iron or steel split-mold. The dimen-
7.3.1.2 Probe sampling is influenced by such factors as the
sions of the sample are typically 70 mm by 35 mm with a
angle and depth of immersion of the sampler in the melt, and
thickness of 4 mm. The two halves of the mold are bevelled at
immersion times can vary depending on the temperature of the
the top to give a feeder head and are clamped together in use.
liquid iron. These factors should be determined for the particu-
This type of mold may be preferred for use with liquid iron
lar iron-making practice, and thereafter, strictly controlled to
containing high amounts of carbon.
maintain the standard of quality of the sample.
7.2.2 Procedures:
7.3.2 Procedures:
7.2.2.1 For sampling from a melt, immerse a preheated steel 7.3.2.1 For sampling from a melt, immerse a suitable
spoon into the melt and fill with liquid iron. Withdraw the
immersion probe sampler into the melt at an angle as near as
spoon and remove any slag by skimming the surface of the possible to the vertical plane.
liquid iron.
7.3.2.2 When sampling from the runner of a blast furnace,
7.2.2.2 For sampling from a stream, introduce a pre-heated select the position of immersion to give sufficient depth of
steel spoon into the stream from the ladle and fill with liquid
liquid metal for the use of a probe sampler. A depth of
iron. approximately 200 mm is adequate for most types of sampling
7.2.2.3 Pour the liquid iron from the spoon without delay
probe.
into a metal mold to chill the iron as rapidly as possible. 7.3.2.3 For sampling from a stream of liquid iron, introduce
Remove the sample from the mold and break off any riser.
a suitable suction probe sampler into the metal flow from a
7.2.2.4 It is essential that the liquid iron be poured into a ladle, at an angle of approximately 45° to the vertical plane, at
mold that is cold to ensure adequate chilling. If necessary, the
a position as near as possible to the nozzle of the vessel.
mold should be air-cooled before use. The mold should be free 7.3.2.4 Withdraw the probe sampler from the melt after a
from moisture.
predetermined interval of time, break it apart, and allow the
7.2.3 Maintenance of Equipment: sample to cool in air.
E1806 − 23
7.4 Preparation of a Sample for Analysis: average analysis determined. In continuous processes, samples
should be taken at regular intervals of time.
7.4.1 Preliminary Preparation—Remove any surface oxida-
tion from a sample taken from the melt that may contaminate
8.1.4 Methods of sampling normally are designed to chill
the sample during subsequent preparation. the liquid metal of a sample cast from a spoon, as rapidly as
7.4.2 Sample for Analysis by a Chemical Method: possible, to produce a metallurgical structure of white iron free
from graphite. A white iron structure obtained by chill-casting
7.4.2.1 Break the sample into small pieces and crush pieces
is required generally for analysis by spectrochemical methods.
using a percussion mortar or a vibratory grinding mill to obtain
8.1.5 Non-chilled samples also may be used. These samples
a sufficient mass of sample of particle size less than 150 μm.
can be cast from a spoon, or selected from a test bar or keel
7.4.2.2 Alternatively, obtain chips by drilling the sample at
block intended for mechanical testing. Test bars or blocks are
a low speed as described in 10.3.1.2.
cast separately from the same metal as that used to produce the
7.4.3 Sample for Analysis by a Thermal Method:
castings.
7.4.3.1 Break the pin of a disk-shaped sample into pieces of
8.1.6 By agreement with the customer, when large castings
a suitable mass for use as test portions or use the lugs of a
or large numbers of castings are produced, two or more
probe sample. Analyze a representative number of test portions
samples should be obtained.
to obtain an average value.
8.1.7 Special considerations apply to the sampling and
7.4.3.2 Alternatively, crush the pin or lugs using a percus-
sample preparation of liquid iron for the determination of
sion mortar to obtain a sufficient mass of sample of particle size
hydrogen, oxygen, and nitrogen (see 8.5).
approximately 1 mm to 2 mm. Avoid the production of fine
material during crushing. For a slab-shaped sample, break the
8.2 Spoon Sampling:
sample into small pieces and crush the pieces in a similar
8.2.1 General:
manner.
8.2.1.1 Sampling should take place before any additions of
7.4.4 Sample for Analysis by a Spectrochemical Method:
inoculating agents to the melt.
7.4.4.1 In the case of a disk-shaped sample, remove any lugs
or pin, as necessary, and grind the surface of the sample to NOTE 2—When sampling takes place before addition of inoculating
agents, recognize that the sample obtained will not be representative of the
expose a white iron structure that is representative of the
chemical composition of the cast product.
sample. The amount of material to be removed in this way
should be determined for the chemical composition of the 8.2.1.2 Alternatively, sufficient time should elapse for the
particular iron, and the conditions of sampling. The thickness immediate effect of the additions to fade and the melt should be
of the layer to be removed is normally 0.5 mm to 1 mm. thoroughly stirred before sampling. Failure to allow adequate
standing time before taking the sample will seriously impair
7.4.4.2 For a slab-shaped sample, break the slab into two
the representivity of sampling.
pieces to obtain a sample of suitable size.
7.4.4.3 Prepare the surface of the sample by grinding and 8.2.1.3 Ductile iron is difficult to sample due to the possi-
bility of contamination by dross during the production process.
linishing. Grinding should be performed wet to avoid overheat-
ing the sample but the final surface preparation should be dry A suitable sample may be obtained by filtering the iron using
a ceramic disk.
linishing. Alternatively, cool the sample after grinding by
immersion in water, and then finish by dry linishing.
8.2.2 Methods—A graphite spoon or a steel spoon lined with
7.4.4.4 Particular care is required when preparing the sur- a layer of a refractory, such as ganister, is suitable for use in
face of thin samples. A chuck should be specially designed to accordance with one of the following methods.
hold the sample securely during grinding and linishing opera-
8.2.2.1 Remove any slag from the surface of the melt by
tions.
skimming, and then immerse a preheated spoon into the melt
and fill with liquid iron.
8. Liquid Iron for Cast Iron Production
8.2.2.2 Introduce a preheated spoon into the stream during
pouring and fill with liquid iron.
8.1 General:
8.2.3 Chilled Sample:
8.1.1 The following methods are applicable to the sampling
8.2.3.1 Pour the liquid iron from the spoon without delay
of liquid iron from cupola furnaces and electric furnaces,
into a split mold made from graphite, hematite iron, or copper
holding furnaces in duplex processes, and ladles and treatment
to obtain a sample in the form of a small, flat plate, 4 mm to
vessels.
8 mm in thickness. Remove the sample from the mold when
8.1.2 Liquid iron intended for the production of iron cast-
solidified to avoid overheating the mold and the risk of sample
ings may be subject to inhomogeneity and particular care is
breakage. Break off any riser.
required in the design of strategies and methods of sampling to
meet the requirements of the particular production process. For 8.2.3.2 The sample, commonly described as a coin sample,
may be circular, rectangular, or square with typical sizes,
example, liquid iron in holding furnaces tends to stratify, and
sampling should ensure that analysis is representative of the respectively, 35-mm to 40-mm diameter, (50 by 27) mm and
(50 by 50) mm. Generally, disk samples are cast vertically and
melt as a whole.
rectangular and square samples are cast horizontally.
8.1.3 In batch processes, two or more samples should be
taken from melting furnaces preferably when approximately 8.2.3.3 The mold is constructed in two pieces that are
one third and two thirds of the melt has been discharged and an clamped together in use: one piece is a flat chill plate, and the
E1806 − 23
other is a block with the mold cavity. The edge of the mold using a tungsten-carbide-tipped tool adjusting the speed and
cavity may be tapered to facilitate removal of the sample from feed to produce chips of uniform size avoiding the production
the mold. of fine particles. Avoid overheating the sample and the tool.
8.2.3.4 A coin sample with one or more attached pins can be Chips should be as solid and compact as possible with a mass
obtained using a combination-type mold. The pins are broken of approximately 10 mg to prevent crumbling and loss of
off from the disk and used, if required, as test portions for graphite. Chips should not be cleaned with a solvent or treated
analysis by a thermal method. A vertical mold of this type, magnetically because of the risk of altering the distribution of
commonly described as a book mold and made from low- metal and graphite. A tool with a diameter of 10 mm is suitable
phosphorus, high-carbon grey iron, or from copper or graphite, for use when obtaining chips by drilling.
is shown in Fig. 2. The sample obtained is a disk, 38 mm in 8.4.2.2 The size range of chips intended for the determina-
diameter by 6 mm thick, with three 5-mm diameter pins.
tion of total carbon should be 1 mm to 2 mm.
8.2.3.5 The temperature of the liquid iron in the spoon
8.4.2.3 When machining is not practicable, the sample can
should be as high as possible and compatible with the mold
be broken into pieces and the pieces crushed using a percussion
material. It is essential that the mold is cold to ensure adequate
mortar or vibratory grinding mill to obtain a sufficient mass of
chilling for the production of a sample with a white iron
sample of particle size less than 150 μm. Use this method
structure. If necessary, the mold should be air-cooled before
where it can be shown that pulverization does not lead to
use. The mold should be free from moisture.
contamination of the sample.
8.2.3.6 For processes where samples are required to be
8.4.2.4 For a chilled sample, drill the sample, if practicable,
taken at frequent intervals, provide several molds to ensure the
discarding chips obtained from the surface.
availability of a mold that is cold.
8.4.2.5 For a non-chilled sample, like a cylindrical block,
8.2.3.7 Thermal stress due to overheating of the mold can
drill a hole transversely at a position one third along the length
cause breakage of the coin sample and should be avoided.
of the block. Then drill another hole from the opposite side.
8.2.4 Non-Chilled Sample:
Discard chips from one third of the radial depth in both
8.2.4.1 Pour the liquid iron from the spoon without delay
directions. Continue drilling through the center of the block to
into a sand mold to obtain a cylindrical block-shaped sample
obtain the sample.
approximately 50 mm in diameter by 40 mm to 50 mm long.
8.4.2.6 For a test bar, either grind two flats on opposite sides
8.2.4.2 Alternatively, a sample can be selected from a test
of the bar and drill from one side to the other at a position one
bar or keel block intended for mechanical testing. Test bars or
third along the bar or turn the test bar using a lathe with a
blocks are cast either from liquid iron taken from a ladle using
maximum cut of 0.25 mm. Do not use a cutting fluid or
a pouring spoon, or, if a small hand-ladle is used for pouring,
coolant. Use a radial cut from edge to center, or face turn a
directly from the ladle itself. Bars are typically 30 mm in
cross section of the bar, but do not confine turning to the
diameter by 150 mm long and may be cast vertically or
surface of the bar. Discard chips obtained from the surface of
horizontally in a sand mold.
the bar. For a non-machinable sample, break pieces from the
8.2.4.3 Allow samples to cool completely before removal
sample or cut a 3-mm slice or disk from the cross section near
from the mold.
the bottom of a test bar. Crush these pieces using a percussion
8.2.5 Maintenance of Equipment:
mortar or vibratory grinding mill to obtain a sufficient mass of
8.2.5.1 It is essential to maintain pouring spoons and molds
sample of particle size less than 150 μm.
in a clean and dry condition. After use, remove any slag and
8.4.3 Sample in the Form of a Solid Mass for Analysis by a
skull and brush the faces of the mold.
Thermal Method:
8.2.5.2 Molds should be remachined if the surfaces become
8.4.3.1 For a chilled sample, remove a pin from the sample
worn. This avoids the need for additional machining of the
and break or cut the pin into pieces for use as test portions.
sample during surface preparation.
8.4.3.2 Alternatively, crush the pin in a percussion mortar to
8.3 Probe Sampling—Probe sampling has limited use in the
provide a sample of particle size range approximately 1 mm to
manufacture of cast iron products. Sampling probes, if
2 mm. Avoid the production of fine material.
required, should be designed to provide samples from the melt
8.4.3.3 For a non-chilled sample, use a saw to cut a 3-mm
of a quality and metallurgical structure required by the analysis
disk or slice from the cross section of a cylindrical block or test
method.
bar and cut pieces of a suitable mass for use as test portions.
8.4.3.4 Analyze a representative number of test portions to
8.4 Preparation of a Sample for Analysis:
obtain an average value. The mass of a piece selected as a test
8.4.1 Preliminary Preparation:
portion should not be less than approximately 0.3 g.
8.4.1.1 Remove any sand adhering to the surface of a
sand-cast sample by scratch brushing or shot-blasting. Remove 8.4.4 Sample for Analysis by a Spectrochemical Method:
any surface oxidation by grinding. 8.4.4.1 For a chilled sample, remove any pins and then use
8.4.1.2 Prepare the sample in accordance with one of the
a fixed-head grinder to expose a white iron structure which is
following procedures depending upon the method selected for representative of the sample. The amount of material to be
analysis.
removed in this way should be determined for the chemical
8.4.2 Sample for Analysis by a Chemical Method: composition of the particular iron and the conditions of
8.4.2.1 Machining to obtain chips should be conducted by sampling. The thickness of the layer to be removed is normally
drilling or turning at a low speed (100 r ⁄min to 150 r ⁄min) at least 1 mm.
E1806 − 23
8.4.4.2 Air-cooling is recommended during grinding. Grind- 9. Liquid Steel for Steel Production
ing can be conducted wet to avoid overheating the sample, but
9.1 General:
the final treatment should be dry grinding or linishing. Exces-
9.1.1 The following methods are applicable to the sampling
sive grinding may lead to analysis errors if the chill region of
of liquid steel from furnaces, ladles, and other vessels, and
the sample is exceeded. Chilled samples should be examined
from tundishes and molds during the melting, secondary
regularly in routine practice to ensure the suitability of the
treatment, and casting of the steel.
metallurgical structure of the prepared sample for the analysis
9.1.2 Special considerations apply to sampling and sample
method.
preparation of liquid steel for the determination of oxygen and
8.4.4.3 For a non-chilled sample, use a grinding or linishing
hydrogen. See 9.5 and 9.6, respectively.
machine to remove a layer approximately 1 mm in thickness
from the surface of the sample. Air cooling is recommended
9.2 Probe Sampling:
during grinding and, liquid coolants should not be used.
9.2.1 General:
8.4.4.4 For irons that are subject to segregation effects, for
9.2.1.1 The main features of the different types of commer-
example, high-phosphorus engineering iron, high-silicon duc-
cially available sampling probes for use with liquid steel are
tile iron, malleable iron, prepare the surfaces of two sides of the
distinguished in Annex A1.
sample to obtain an average value.
9.2.1.2 Probe sampling is influenced by such factors as the
8.4.4.5 Avoid overheating of the sample during surface
angle and depth of immersion of the sampler and the time of
preparation as this can result in surface crazing that will affect
immersion in the melt. It is essential that these factors be
correctness of analysis.
determined for the particular conditions of composition and
8.4.4.6 Care is required when preparing the surface of a thin temperature of the steel involved, and thereafter, strictly
coin-sample. A chuck should be designed to hold the sample controlled to maintain the standard of quality required for the
securely during grinding operations.
analysis.
9.2.1.3 Take precautions to ensure that the operation of
NOTE 3—A fixed-head grinder is preferred to a swing grinder for
probe sampling does not contaminate the sample of liquid steel
surface preparation; the latter type of equipment may not give a flat
surface to the sample. particularly when sampling for the determination of elements
present in low amounts. The selection of materials used for
8.5 Sampling and Sample Preparation for the Determina-
construction of the sampling probe, the design of the capping
tion of Oxygen, Nitrogen, and Hydrogen:
and entrance system, and the method of deoxidation should be
8.5.1 General—The determination of oxygen, nitrogen, and
such as to minimize risk of contamination, other than from the
hydrogen is required infrequently in the production of castings.
deoxidant itself.
Sampling and sample preparation methods should minimize
9.2.2 Procedures:
hydrogen losses and avoid contamination of the sample by
9.2.2.1 For sampling from deep melts, such as melting
oxygen, nitrogen, or hydrogen.
furnaces and ladles, quickly immerse a suitable probe sampler
8.5.2 Procedure:
through the slag layer into the melt as near as possible to the
8.5.2.1 It is essential that a sample for the determination of
c
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E1806 − 18 E1806 − 23
Standard Practice for
Sampling Steel and Iron for Determination of Chemical
Composition
This standard is issued under the fixed designation E1806; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This practice covers the sampling of all grades of steel, both cast and wrought, and all types (grades) of cast irons and blast
furnace iron for chemical and spectrochemical determination of composition. This practice is similar to ISO 14284.
1.2 This practice is divided into the following sections.
Sections
Requirements for Sampling and Sample Preparation 6
General 6.1
Sample 6.2
Selection of a Sample 6.3
Preparation of a Sample 6.4
Liquid Iron for Steelmaking and Pig Iron Production 7
General 7.1
Spoon Sampling 7.2
Probe Sampling 7.3
Preparation of a Sample for Analysis 7.4
Liquid Iron for Cast Iron Production 8
General 8.1
Spoon Sampling 8.2
Probe Sampling 8.3
Preparation of a Sample for Analysis 8.4
Sampling and Sample Preparation for the Determination of 8.5
Oxygen and Hydrogen
Liquid Steel for Steel Production 9
General 9.1
Probe Sampling 9.2
Spoon Sampling 9.3
Preparation of a Sample for Analysis 9.4
Sampling and Sample Preparation for the Determination 9.5
of Oxygen
Sampling and Sample Preparation for the Determination 9.6
of Hydrogen
Pig Irons 10
General 10.1
Increment Sampling 10.2
Preparation of a Sample for Analysis 10.3
This practice is under the jurisdiction of ASTM Committee E01 on Analytical Chemistry for Metals, Ores, and Related Materials and is the direct responsibility of
Subcommittee E01.01 on Iron, Steel, and Ferroalloys.
Current edition approved March 15, 2018June 1, 2023. Published April 2018July 2023. Originally approved in 1996. Last previous edition approved in 20162018 as
E1806 – 09 (2016).E1806 – 18. DOI: 10.1520/E1806-18.10.1520/E1806-23.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E1806 − 23
Cast Iron Products 11
General 11.1
Sampling and Sample Preparation 11.2
Sections
Steel Products 12
General 12.1
Selection of a Laboratory Sample or a Sample for 12.2
Analysis from a Cast Product
Selection of a Laboratory Sample or a Sample for 12.3
Analysis from a Wrought Product
Preparation of a Sample for Analysis 12.4
Sampling of Leaded Steel 12.5
Sampling and Sample Preparation for the Determination 12.6
of Oxygen
Sampling and Sample Preparation for the Determination 12.7
of Hydrogen
Keywords 13
Annexes
Sampling Probes for Use with Liquid Iron and Steel Annex A1
Sampling Probes for Use with Liquid Steel for the Annex A2
Determination of Hydrogen
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use. For specific statements, see 6.4.3.5, 9.4.4.3, and 12.5.1 as well as , and Section 5.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
A48/A48M Specification for Gray Iron Castings
A751 Test Methods and Practices for Chemical Analysis of Steel Products
E135 Terminology Relating to Analytical Chemistry for Metals, Ores, and Related Materials
E415 Test Method for Analysis of Carbon and Low-Alloy Steel by Spark Atomic Emission Spectrometry
E1010E1306 Practice for Preparation of Disk Specimens of Steel and Iron by RemeltingMetal and Alloy Samples by Electric
Arc Melting for Spectrochemical Analysis (Withdrawn 2022)
2.2 ISO Documents:Standards:
ISO 9147 Pig irons–Definition and classification
ISO 14284 Steel and iron—Sampling and preparation of samples for the determination of chemical composition
3. Terminology
3.1 Definitions—For definitions of terms in this practice, refer to Terminology E135.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 cast product, n—item of iron or steel which has not been subject to deformation, for example, an ingot, a semi finished
product obtained by continuous casting, or a shaped casting.
3.2.2 consignment, n—quantity of metal delivered at one time.
3.2.3 grinding, n—method of preparing a sample of metal for a spectrochemical method of analysis in which the surface of the
sample for analysis is abraded using an abrasive wheel.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Available from American National Standards Institute (ANSI), 25 W. 43rd St., 4th Floor, New York, NY 10036, http://www.ansi.org.
E1806 − 23
3.2.4 linishing, n—method of preparing a sample of metal for a spectrochemical method of analysis in which the surface of the
sample for analysis is abraded using a rotating disk or continuous belt coated with an abrasive substance.
3.2.5 melt, n—liquid metal from which a sample is removed.
3.2.6 sample, probe, n—sample taken from the melt using a sampling probe.
3.2.7 sample, product, n—specific item of iron or steel selected from a supplied quantity for the purpose of obtaining a sample.
3.2.8 sample, spoon, n—sample taken from the melt using a spoon and cast into a small mold.
3.2.9 sampling, immersion, n—method of probe sampling in which the probe is immersed in the melt whereand the sample
chamber in the probe fills by ferrostatic pressure or gravity.
3.2.10 sampling, probe, n—method in which the sample is taken using a sampling probe inserted into the melt.
3.2.11 sampling, spoon, n—method in which the sample is taken from the melt, or during the pouring of the melt, using a
long-handled spoon and cast into a small mold.
3.2.12 sampling, stream, n—method of probe sampling in which the probe is inserted into a stream of liquid metal whereand the
sample chamber in the probe fills by force of metal flow.
3.2.13 sampling, suction, n—method of probe sampling in which the probe is inserted into the melt whereand the sample chamber
in the probe fills by aspiration.
3.2.14 test portion, n—part of the sample for analysis, or part of the sample taken from the melt, actually analyzed.
3.2.14.1 Discussion—
In certain cases, Sometimes, the test portion may be selected from the sample product itself.
3.2.15 thermal method of analysis, n—method for the determination of chemical composition in which the sample is submitted
to a process of heating, combustion, or fusion.
3.2.16 wrought product, n—item of steel which has been subject to deformation by rolling, drawing, forging, or some other
method, for example, a bar, billet, plate, strip, tube, or wire.
4. Significance and Use
4.1 This practice covers all aspects of sampling and preparing steel and iron for chemical analysis as defined in Test Methods,
Practices, and Definitions A751 and Specification A48/A48M. Such subjects as sampling location and the sampling of lots are
defined.
4.2 It is intended that this practice includeThis practice includes most requirements for sampling steel and iron for analysis.
Standard analyticaltest methods that reference this practice need contain only special modifications and exceptions.
4.3 It is assumed that all All who use these procedures willshould be trained samplers capable of performing common sampling
operations skillfully and safely. It is expected that only Only proper sampling equipment willshould be used.
5. Hazards and Safety Precautions
5.1 Provide personal protective equipment to minimize the risk of injury during sampling and sample preparation methods.
Provisions shall include protective clothing, hand protection, and face visors resistant to splashes of liquid metal for use during
the sampling of liquid metal; protective clothing and hand, eye, and hearing protection for use during the sampling and sample
preparation of solid metal; and respiratory protection for use where necessary.
E1806 − 23
5.2 The use of machinery for sampling and sample preparation shall be in accordance with appropriate national standards.
Grinding operations used for surface preparation may be covered by national legislation.
5.3 Reference should be made Refer to appropriate national regulations with respect to regarding the use of solvents for the
cleaning and drying of samples and test portions.
6. Requirements for Sampling and Sample Preparation
6.1 General:
6.1.1 This section describes the general requirements for the sample and for the sampling and sample preparation of iron and steel.
Special requirements apply to each category of liquid and solid metal, and these requirements are described in the relevant section.
6.1.2 The sequence of sampling and sample preparation of liquid iron and steel and cast iron and steel products is shown in Fig.
1. Special considerations apply to pig irons (see Section 10).
FIG. 1 Sequence of Sampling and Sample Preparation of (a) Liquid Iron and Steel and (b) Cast Iron and Steel Products
E1806 − 23
6.2 Sample:
6.2.1 Quality:
6.2.1.1 Sampling practices shall be designed to provide an analytical sample that is representative of the mean chemical
composition of the melt or of the sample product.
6.2.1.2 Ensure that the sample is sufficiently homogeneous with respect to regarding chemical composition so that inhomogeneity
does not appreciably contribute to the error variability of the method of analysis. In the case of For a sample taken from a melt,
however, some variability in analysis, both within and between samples for analysis, samples, may be unavoidable. This variability
will form an inherent part of the repeatability and reproducibility of the analysis.
6.2.1.3 Ensure that the sample is free from surface coatings and from moisture, dirt, or other forms of contamination.
6.2.1.4 The sample should be free from voids, cracks, and porosity and from fins, laps, or other surface defects.
6.2.1.5 Take particular care when selecting and preparing the sample whereif a sample taken from a melt is expected to be
heterogeneous or contaminated in any way.
6.2.1.6 A sample taken from a melt shall be cooled in such a manner that the chemical composition and metallurgical structure
of the sample is consistent from sample to sample.
6.2.1.7 It is important to recognize that analysis by some spectrochemical methods may be influenced by the metallurgical
structure of the sample, particularly in the case of for irons with white and grey iron structures, and steels in the as-cast and wrought
conditions.
6.2.2 Size:
6.2.2.1 Ensure that the dimensions of a laboratory sample in the form of a solid mass are sufficient to permit additional samples
for analysis to be taken for reanalysis when necessary using an alternative method of analysis.
6.2.2.2 A sample shall be prepared consisting of a sufficient mass to provide for any reanalysis necessary. Generally, a mass of
100 g will be sufficient for a sample in the form of chips or powder.
6.2.2.3 The dimensional requirements for a sample in the form of a solid mass will depend upon the method selected for analysis.
In the case of For spark atomic emission and X-ray fluorescence spectrometric methods, the shape and size of the sample will be
determined by the dimensions of the sample chamber. The dimensions for samples given in this practice are for guidance only.
Refer to Test Method E415 for sample size requirements for spark atomic emission spectrometry.
6.2.3 Identification:
6.2.3.1 Assign the sample a unique identification to determine the melt or sample product from which it was taken, and if
necessary, the process conditions of the melt or the location of the laboratory sample in the sample product. For pig iron, this shall
identify the consignment or part of a consignment and the increment from which it was taken.
6.2.3.2 Use labeling or some equivalent method of marking to ensure that the assigned identification remains associated with the
sample for analysis.sample.
6.2.3.3 Record the identification of the sample to ensure that confusion cannot arise as to is prevented regarding the identity of
the item to which the analysis and records refer.
6.2.4 Conservation:
6.2.4.1 Provide adequate storage facilities to segregate and protect the sample. During and after preparation, store the sample in
a manner which prevents contamination or chemical change.
E1806 − 23
6.2.4.2 Keep the sample, or the laboratory sample in the form of a solid mass, mass form, for a sufficient period of time to permit
retesting if necessary.
6.2.5 Arbitration:
6.2.5.1 Samples intended for arbitration shall be prepared jointly by the supplier and purchaser or by their representatives. Keep
a record of the methods used for preparing samples.
6.2.5.2 Both parties or their representatives shall seal containers with the samples intended for arbitration. Unless agreed to the
contrary, the representatives of each party responsible for the preparation of samples will keep these containers.
6.3 Selection of a Sample:
6.3.1 Sample from a Melt:
6.3.1.1 Melts are sampled at various stages in the manufacturing process for the purposes of monitoring and controlling the
process. Samples can be taken during the casting of the melt to verify chemical composition in accordance with the specification
of the cast product. In the case of For liquid metal intended for the production of a casting, the analytical sample may be selected
from test bars or blocks specially cast from the same metal as that of the casting for purposes of mechanical testing in accordance
with the product standard.
6.3.1.2 Sampling practices shall be designed for melts to provide samples during a particular manufacturing process in accordance
with requirements for sample quality (see 6.2.1). The sample obtained from a melt usually is in the form of is usually a small ingot,
a cylindrical or rectangular block, or a chill-cast disk or a combination of a disk with one or more attached pins, in some cases
pins. Sometimes small lugs are attached to a disk sample.
NOTE 1—Sampling probes for use with liquid iron and steel may be obtained from a number of suppliers. The main features of the different types of probe
are distinguished in Annex A1 and Annex A2, which include dimensions for information only.
6.3.2 Sample from a Product:
6.3.2.1 The laboratory sample can be selected from the sample product at the location indicated in the product specification for
the selection of material for mechanical testing when available.
6.3.2.2 In the case of For an iron casting, the sample can be selected from a bar or block cast-on to the casting.
6.3.2.3 In the case of For a forging, the sample can be selected from the initial starting material from which the forging has been
made or from prolongations of the forging or from additional forgings.
6.3.2.4 In the absence of requirements given in the product standard or of a specification when ordering the product, the sample
may, following agreement between the supplier and the purchaser, be selected from the sample for mechanical testing or from the
test piece, or directly from the sample product.
6.3.2.5 The laboratory sample can be obtained from the sample product by machining or by using a cutting torch. Special
considerations apply in the case of to sampling for the determination of certain elements.
6.4 Preparation of a Sample:
6.4.1 Preliminary Preparation:
6.4.1.1 If any part of the sample is liable to be potentially nonrepresentative in chemical composition, remove those parts that have
changed. Following this operation, the sample shall be protected from any change in composition.
6.4.1.2 Remove any coating that has been applied during manufacturing. If necessary, degrease the surface of the metal with a
suitable solvent, taking care to ensure that the manner of degreasing does not affect the correctnessaccuracy of analysis.
E1806 − 23
6.4.2 Sample in the Form of Chips:
6.4.2.1 Obtain the sample by drilling or drilling, milling or turning to produce chips of a regularconsistent size and shape. Do not
machine on a part of the sample that has been affected by the heat of a cutting torch.
6.4.2.2 Clean Before preparation, clean the tools, machines, and containers to be used during preparation of the sample beforehand
to prevent any contamination of the sample for analysis.sample.
6.4.2.3 Machine in such a way that the chips are not subject to overheating as indicated by a change in the color (blueing or
blackening) of the chips. blackening). Unavoidable coloration of chips obtained from some types of alloy steels, for example,
manganese and austenitic steels, can be minimized by selection of appropriate tools and cutting speeds.
6.4.2.4 Heat treatment may be required to soften the sample for machining.
6.4.2.5 The use of coolants during machining is only permitted in exceptional cases, after which the chips shall be cleaned by
means of a suitable solvent that does not leave any deposit.residue.
6.4.2.6 Mix the chips thoroughly before weighing the test portion. For most purposes, it is satisfactory to mix the chips by rolling
the container on a level surface, gently tumbling the container.chips.
6.4.3 Sample in the Form of a Powder or Fragments:
6.4.3.1 WhereIf drilling of the sample to obtain chips is impracticable, cut or break the sample into pieces. Crush the pieces using
a percussion mortar or a vibratory grinding mill, also known as a disk mill or ring mill, to obtain a sample in the form of a powder
the whole of which passes through a sieve of a specified aperture size.
6.4.3.2 In some applications the sample is crushed in a percussion mortar to obtain a sample for analysis in the form of fragments
suitable for use with the selected method of analysis.
6.4.3.3 Equipment used for pulverization shall be constructed from material that does not alter the sample composition. Tests may
be necessary to show that such equipment does not affect the composition of the sample.
6.4.3.4 Sieve the sample taking precautions to avoid contamination or loss of material. Take care when sieving hard materials to
avoid damaging the fabric of the sieve.
6.4.3.5 Homogenize the sample before weighing the test portion. Powders can be homogenized by stirring. (Warning—Finely
divided metals of particle size less than approximately 150 μm can present a fire risk. Ensure adequate ventilation during
pulverization.)
6.4.4 Sample in the Form of a Solid Mass:
6.4.4.1 Obtain the sample by cutting from the sample product or laboratory sample a piece of suitable size and shape for the
method of analysis. Cut samples by sawing, abrasive cutting, shearing, or punching.
6.4.4.2 In the absence of any indication in the product standard, perform a spectrochemical analysis on that part of the sample
corresponding to a transverse section of the product, provided that the material has sufficient thickness.
6.4.4.3 Prepare the sample to expose a surface suitable for the analytical method. Do not prepare a surface for analysis on part
of a sample that has been affected by the heat of a cutting torch. The equipment used for sample preparation shall be designed to
minimize overheating the sample, and, where appropriate, shall incorporate systems of cooling.
6.4.5 Main Types of Equipment Used for Surface Preparation:
6.4.5.1 A milling machine capable of removing a preselected depth of metal in a reproducible manner, for use with samples that
are within a hardness range suitable for milling. The equipment should be capable of use, if required, with a sample taken from
a melt wherewhile the sample is still hot.
E1806 − 23
6.4.5.2 A grinding machine with a fixed, rotating, or oscillating head capable of removing a preselected depth of metal in a
reproducible manner.
6.4.5.3 A flatbed linishing machine with abrasive grinding disks, or machines with continuous abrasive belts, capable of preparing
the surface of the sample for analysis to varying grades of finish.
6.4.5.4 A machine for blasting with sand or grit, or metal shot, capable of cleaning the surface of the sample for analysis or the
test portion when required.
6.4.5.5 Ensure that the surface of the sample after preparation is flat and free from defects that could affect correctnessaccuracy
of analysis.
6.4.5.6 Cutting and surface preparation can be performed either manually or automatically. In the case of For samples taken from
melts, commercially available systems may be used that perform each stage of preparation automatically. Systems for the
automatic surface preparation of dualthickness probe samples (see A2.3) and for the punching of slugs as test portions may
incorporate facilitiesoperations for the sandblasting of the sample and for heat treatment to soften the sample before punching.
6.4.5.7 Select the abrasive substances used in the final stage of preparing the sample that do not contaminate the surface with
elements that are to be determined by the analysis method. Ensure that the grit size of the abrasive is in accordance with the grade
of surface finish required for the analysis method.
6.4.5.8 In the case of For spark atomic emission spectrometric methods, an abrasive with a gradesize of 60 grit to 120 grit
normally is suitable. In the case of For X-ray fluorescence spectrometric methods, it is essential to ensure that the method selected
for surface preparation produces a grade of surface finish that is sufficiently smooth and reproducible from sample to sample. Also,
there should be no smearing of the surface.
6.4.5.9 The effect of abrasive materials depends on the analytical method. When using spark atomic emission spectrometric
methods, the action of pre-sparking normally will clean the surface of the sample for analysis by volatilizing any grinding
contaminants. Particular care is required, however, to avoid surface contamination when using a new abrasive disk.media.
6.4.5.10 When using X-ray fluorescence spectrometric methods, examine all phases of surface preparation for potential surface
contamination effects.
6.4.5.11 Examine the sample visually after preparation to establish that the surface is dry and free from particulate matter and that
there are no defects. Resurface or discard the sample if defects are present. Protect the prepared surface from contamination.
6.4.6 Preparation of a Sample for Analysis by Remelting:Melting:
6.4.6.1 A sample in the form of small pieces or chips, or a part of the sample product itself, can be remeltedmelted in an
atmosphere of argon using commercially available melting equipment of the type described in Practice E1010E1306. The sample
is converted into a disk, 40-mm to 30-mm diameter by 6 mm thick, which is suitable for analysis by a spectrochemical method.
Some types of remeltingmelting equipment incorporate facilitiesoperations for the centrifugal casting of the disk sample.
6.4.6.2 Partial losses of some elements can be experienced during the remeltingmelting process. It is essential to ensure that any
selective volatilization or segregation of elements, or any other change in composition, which occur are known quantitatively and
do not significantly influence the analysis results. Carry out Conduct suitable tests to show that any change in composition is both
small in magnitude and reproducible.
6.4.6.3 The equipment used and the method adopted for remeltingmelting shall be designed to prevent or minimize change in
composition and to ensure that any change is reproducible. A deoxidant, for example 0.1 % (m ⁄m) zirconium, should be used
during remelting. Take into account melting. Consider any change that does occur when using the method for calibration of
analytical measurement any change that does occur. measurement.
6.4.6.4 Not all ferrous metals can be remeltedmelted in this manner. Do not use remeltingmelting as a method of sample
preparation for the determination of an element that is subject to a significant and nonreproducible change in composition.
E1806 − 23
7. Liquid Iron for Steelmaking and Pig Iron Production
7.1 General:
7.1.1 The following methods are applicable to the sampling of for sampling liquid blast-furnace iron intended for steelmaking, and
commonly described as hot metal, or for the casting of pig iron. The liquid iron is normally sampled from the blast furnace runner
during the pouring of the melt into torpedo ladles, or from transfer vessels or during secondary treatment processes in the ladle,
or during the casting of the melt into pig iron.
7.1.2 The chemical composition of iron may fluctuate during run-out from the blast furnace. Two or more samples should be taken
from the melt at timed intervals and an average analysis determined.
7.1.3 When spectrochemical methods are used for analysis, the method of sampling should be designed to chill the liquid metal
in a manner that ensures that the metallurgical structure of the sample is suitable for the requirements of the analysis method
selected.
7.2 Spoon Sampling:
7.2.1 Hold Types:
7.2.1.1 A disk-shaped sample, commonly described as a coin sample, can be obtained using a two-piece steel mold. The
dimensions of the sample are typically 35 mm to 40 mm in diameter with thickness varying from 6 mm to 12 mm. The mold is
constructed in two pieces that are clamped together in use: one piece is a flat chill plate and the other is a block with the mold
cavity. The edge of the mold cavity may be tapered, for example, from 38 mm to 32 mm, to facilitate removal of the sample from
the mold.
7.2.1.2 A coin sample with one or more attached pins can be obtained using a combination-type mold. theThe pins are broken off
from the disk and used, if required, as test portions for analysis by a thermal method. A combination-type mold for use with liquid
iron intended for the production of cast iron is shown in Fig. 2.
7.2.1.3 A thin slab-shaped sample with a rounded end can be obtained using a cast iron or steel split-mold. The dimensions of the
sample are typically 70 mm by 35 mm with a thickness of 4 mm. The two halves of the mold are bevelled at the top to give a feeder
head and are clamped together in use. This type of mold may be preferred for use with liquid iron containing high
percentagesamounts of carbon.
NOTE 1—All dimensions are in millimetres.
All dimensions are in millimetres.
The flat chill plate (not shown) has similar overall dimensions.
NOTE 2—The flat chill plate (not shown) has similar overall dimensions.
FIG. 2 Combination-Type Vertical Mold Used for Sampling Liquid Iron Intended for Cast Iron Production
E1806 − 23
7.2.2 Procedures:
7.2.2.1 For sampling from a melt, immerse a preheated steel spoon into the melt and fill with liquid iron. Withdraw the spoon and
remove any slag by skimming the surface of the liquid iron.
7.2.2.2 For sampling from a stream, introduce a pre-heated steel spoon into the stream from the ladle and fill with liquid iron.
7.2.2.3 Pour the liquid iron from the spoon without delay into a metal mold to chill the iron as rapidly as possible. Remove the
sample from the mold and break off any riser.
7.2.2.4 It is essential that the liquid iron should be poured into a mold that is cold to ensure adequate chilling. If necessary, the
mold should be air-cooled before use. The mold should be free from moisture.
7.2.3 Maintenance of Equipment:
7.2.3.1 It is essential to maintain sampling spoons and molds in a clean and dry condition. After use, remove any slag and skull
and brush the faces of the mold with a wire brush.
7.2.3.2 Molds should be re-machined if the internal surfaces become worn. This avoids the need for additional machining of the
sample during surface preparation.
7.3 Probe Sampling:
7.3.1 General:
7.3.1.1 The different types of probes used for sampling blast furnace iron are described in Annex A1. Probes are designed to
provide a disk-shaped sample with a depth of white iron structure that is sufficient for the requirements of the spectrochemical
method selected for analysis.
7.3.1.2 Probe sampling is influenced by such factors as the angle and depth of immersion of the sampler in the melt, and
immersion times can vary depending on the temperature of the liquid iron. These factors should be determined for the particular
iron-making practice, and thereafter, strictly controlled to maintain the standard of quality of the sample for analysis.sample.
7.3.2 Procedures:
7.3.2.1 For sampling from a melt, immerse a suitable immersion probe sampler into the melt at an angle as near as possible to
the vertical plane.
7.3.2.2 When sampling from the runner of a blast furnace, select the position of immersion to give sufficient depth of liquid metal
for the use of a probe sampler. A depth of approximately 200 mm is adequate for most types of sampling probe.
7.3.2.3 For sampling from a stream of liquid iron, introduce a suitable suction probe sampler into the metal flow from a ladle, at
an angle of approximately 45° to the vertical plane, at a position as near as possible to the nozzle of the vessel.
7.3.2.4 Withdraw the probe sampler from the melt after a predetermined interval of time, break it apart, and allow the sample to
cool in air.
7.4 Preparation of a Sample for Analysis:
7.4.1 Preliminary Preparation—Remove any surface oxidation from a sample taken from the melt that may contaminate the
sample during subsequent preparation.
7.4.2 Sample for Analysis by a Chemical Method:
7.4.2.1 Break the sample into small pieces and crush pieces using a percussion mortar or a vibratory grinding mill to obtain a
sufficient mass of sample for analysis of particle size less than 150 μm.
E1806 − 23
7.4.2.2 Alternatively, obtain chips by drilling the sample at a low speed as described in 10.3.1.2.
7.4.3 Sample for Analysis by a Thermal Method:
7.4.3.1 Break the pin of a disk-shaped sample into pieces of a suitable mass for use as test portions or use the lugs of a probe
sample. Analyze a representative number of test portions to obtain an average value.
7.4.3.2 Alternatively, crush the pin or lugs using a percussion mortar to obtain a sufficient mass of sample for analysis of particle
size approximately 1 mm to 2 mm. Avoid the production of fine material during crushing. In the case of For a slab-shaped sample,
break the sample into small pieces and crush the pieces in a similar manner.
7.4.4 Sample for Analysis by a Spectrochemical Method:
7.4.4.1 In the case of a disk-shaped sample, remove any lugs or pin, as necessary, and grind the surface of the sample to expose
a white iron structure that is representative of the sample. The amount of material to be removed in this way should be determined
for the chemical composition of the particular iron, and the conditions of sampling. The thickness of the layer to be removed is
normally lies between 0.5 mm and0.5 mm to 1 mm.
7.4.4.2 In the case of For a slab-shaped sample, break the slab into two pieces to obtain a sample of suitable size for analysis.size.
7.4.4.3 Prepare the surface of the sample by grinding and linishing. Grinding should be carried out performed wet to avoid
overheating the sample but the final surface preparation should be dry linishing. Alternatively, cool the sample after grinding by
immersion in water, and then finish by dry linishing.
7.4.4.4 Particular care is required when preparing the surface of thin samples. A chuck should be specially designed to hold the
sample securely during grinding and linishing operations.
8. Liquid Iron for Cast Iron Production
8.1 General:
8.1.1 The following methods are applicable to the sampling of liquid iron from cupola furnaces and electric furnaces, holding
furnaces in duplex processes, and ladles and treatment vessels.
8.1.2 Liquid iron intended for the production of iron castings may be subject to inhomogeneity and particular care is required in
the design of strategies and methods of sampling to meet the requirements of the particular production process. For example, liquid
iron in holding furnaces tends to stratify, and sampling should ensure that analysis is representative of the melt as a whole.
8.1.3 In batch processes, two or more samples should be taken from melting furnaces preferably when approximately one third
and two thirds of the melt has been discharged and an average analysis determined. In continuous processes, samples should be
taken at regular intervals of time.
8.1.4 Methods of sampling normally are designed to chill the liquid metal of a sample cast from a spoon, as rapidly as possible,
to produce a metallurgical structure of white iron free from graphite. A white iron structure obtained by chill-casting is required
generally for analysis by spectrochemical methods.
8.1.5 Non-chilled samples also may be used. In this case, These samples can be cast specially from a spoon, or selected from a
test bar or keel block intended for mechanical testing. Test bars or blocks are cast separately from the same metal as that used to
produce the casting or castings.
8.1.6 By agreement with the customer, when large castings or large numbers of castings are produced, two or more samples should
be obtained.
8.1.7 Special considerations apply to the sampling and sample preparation of liquid iron for the determination of hydrogen,
oxygen, and nitrogen (see 8.5).
8.2 Spoon Sampling:
E1806 − 23
8.2.1 General:
8.2.1.1 Sampling should take place before any additions of inoculating agents have been made to the melt.
NOTE 2—WhereWhen sampling takes place before addition of inoculating agents, it should be recognized recognize that the sample obtained will not be
representative of the chemical composition of the cast product.
8.2.1.2 Alternatively, sufficient time should be allowed to elapse for the immediate effect of the additions to have faded fade and
the melt should be thoroughly stirred before sampling. Failure to allow adequate standing time before taking the sample will
seriously impair the representivity of sampling.
8.2.1.3 Ductile iron particularly is difficult to sample due to the possibility of contamination by dross during the production
process. In this case, a A suitable sample may be obtained by filtering the iron using a ceramic disk.
8.2.2 Methods—A graphite spoon or a steel spoon lined with a layer of a refractory, such as ganister, is suitable for use in
accordance with one of the following methods.
8.2.2.1 Remove any slag from the surface of the melt by skimming, and then immerse a preheated spoon into the melt and fill
with liquid iron.
8.2.2.2 Introduce a preheated spoon into the stream during pouring and fill with liquid iron.
8.2.3 Chilled Sample:
8.2.3.1 Pour the liquid iron from the spoon without delay into a split mold made from graphite, hematite iron, or copper to obtain
a sample in the form of a small, flat plate, 4 mm to 8 mm in thickness. Remove the sample from the mold, as soon as it is solid,
mold when solidified to avoid overheating the mold and the risk of breakage of the sample. sample breakage. Break off any riser.
8.2.3.2 The sample, commonly described as a coin sample, may be circular, rectangular, or square in shape with typical sizes,
respectively, 35-mm to 40-mm diameter, (50 by 27) mm and (50 by 50) mm. Generally, disk samples are cast vertically and
rectangular and square samples are cast horizontally.
8.2.3.3 The mold is constructed in two pieces that are clamped together in use: one piece is a flat chill plate, and the other is a
block with the mold cavity. The edge of the mold cavity may be tapered to facilitate removal of the sample from the mold.
8.2.3.4 A coin sample with one or more attached pins can be obtained using a combination-type mold. The pins are broken off
from the disk and used, if required, as test portions for analysis by a thermal method. A vertical mold of this type, commonly
described as a book mold and made from low-phosphorus, high-carbon grey iron, or from copper or graphite, is shown in Fig. 2.
The sample obtained is a disk, 38 mm in diameter by 6 mm thick, with three 5-mm diameter pins.
8.2.3.5 The temperature of the liquid iron in the spoon should be as high as possible and compatible with the mold material. It
is essential that the mold is cold to ensure adequate chilling for the production of a sample for analysis with a white iron structure.
If necessary, the mold should be air-cooled before use. The mold should be free from moisture.
8.2.3.6 In the case of a process For processes where samples are required to be taken at frequent intervals, provide several molds
to ensure the availability for use of a mold that is cold.
8.2.3.7 Thermal stress due to overheating of the mold can cause breakage of the coin sample and should be avoided.
8.2.4 Non-Chilled Sample:
8.2.4.1 Pour the liquid iron from the spoon without delay into a sand mold to obtain a cylindrical block-shaped sample
approximately 50 mm in diameter by 40 mm to 50 mm long.
8.2.4.2 Alternatively, a sample can be selected from a test bar or keel block intended for mechanical testing. Test bars or blocks
are cast either from liquid iron taken from a ladle using a pouring spoon, or, if a small hand-ladle is used for pouring, directly from
the ladle itself. Bars are typically 30 mm in diameter by 150 mm long and may be cast vertically or horizontally in a sand mold.
E1806 − 23
8.2.4.3 Allow samples to cool completely before removal from the mold.
8.2.5 Maintenance of Equipment:
8.2.5.1 It is essential to maintain pouring spoons and molds in a clean and dry condition. After use, remove any slag and skull
and brush the faces of the mold.
8.2.5.2 Molds should be remachined if the surfaces become worn. This avoids the need for additional machining of the sample
during surface preparation.
8.3 Probe Sampling—Probe sampling is used only to a limited extenthas limited use in the manufacture of cast iron products.
Sampling probes, if required, should be designed to provide samples from the melt of a quality and metallurgical structure required
by the analysis method.
8.4 Preparation of a Sample for Analysis:
8.4.1 Preliminary Preparation:
8.4.1.1 Remove any sand adhering to the surface of a sand-cast sample by scratch brushing or shot-blasting. Remove any surface
oxidation by grinding.
8.4.1.2 Prepare the sample in accordance with one of the following procedures depending upon the method selected for analysis.
8.4.2 Sample for Analysis by a Chemical Method:
8.4.2.1 Machining to obtain chips should be carried out conducted by drilling or turning at a low speed (100 r ⁄min to 150 r ⁄min)
using a tungsten-carbide-tipped tool adjusting the speed and feed to produce chips of uniform size avoiding the production of fine
particles. Avoid overheating the sample and the tool. Chips should be as solid and compact as possible with a mass of
approximately 10 mg in order to prevent crumbling and loss of graphite. Chips should not be washedcleaned with a solvent or
treated magnetically because of the risk of altering the distribution of metal and graphite. A tool with a diameter of 10 mm is
suitable for use when obtaining chips by drilling.
8.4.2.2 The size range of chips intended for the determination of total carbon should be from 1 mm to 2 mm.
8.4.2.3 When machining is not practicable, the sample can be broken into pieces and the pieces crushed using a percussion mortar
or vibratory grinding mill to obtain a sufficient mass of sample of particle size less than 150 μm. Use this method in cases where
it can be shown that pulverization does not lead to contamination of the sample.
8.4.2.4 For a chilled sample, drill the sample, if practicable, discarding chips obtained from the surface.
8.4.2.5 For a non-chilled sample, in the case of like a cylindrical block, drill a hole transversely at a position one third along the
length of the block. Then drill another hole from the opposite side. Discard chips from one third of the radial depth in both
directions. Continue drilling through the center of the block to obtain the sample.
8.4.2.6 In the case of For a test bar, either grind two flats on opposite sides of the bar and drill from one side to the other at a
position one third along the bar or turn the test bar using a lathe with a maximum cut of 0.25 mm. Do not use a cutting fluid or
coolant. Use a radial cut from edge to center, or face turn a cross section of the bar, but do not confine turning to the surface of
the bar. Discard chips obtained from the surface of the bar. For a non-machinable sample, break pieces from the sample or cut a
3-mm slice or disk from the cross section near the bottom of a test bar. Crush these pieces using a percussion mortar or vibratory
grinding mill to obtain a sufficient mass of sample for analysis of particle size less than 150 μm.
8.4.3 Sample in the Form of a Solid Mass for Analysis by a Thermal Method:
8.4.3.1 For a chilled sample, remove a pin from the sample and break or cut the pin into pieces for use as test portions.
8.4.3.2 Alternatively, crush the pin in a percussion mortar to provide a sample for analysis of particle size range from
approximately 1 mm to 2 mm. Avoid the production of fine material.
E1806 − 23
8.4.3.3 For a non-chilled sample, use a saw to cut a 3-mm disk or slice from the cross section of a cylindrical block or test bar
and cut pieces of a suitable mass for use as test portions.
8.4.3.4 Analyze a representative number of test portions to obtain an average value. The mass of a piece selected as a test portion
should not be less than approximately 0.3 g.
8.4.4 Sample for Analysis by a Spectrochemical Method:
8.4.4.1 For a chilled sample, remove any pins and then use a fixed-head grinder to expose a white iron structure which is
representative of the sample. The amount of material to be removed in this way should be determined for the chemical composition
of the particular iron and the conditions of sampling. The thickness of the layer to be removed is normally at least 1 mm.
8.4.4.2 Air-cooling is recommended during grinding. Grinding can be carried out conducted wet to avoid overheating the sample,
but the final treatment should be dry grinding or linishing. Excessive grinding may lead to analysis errors if the chill region of the
sample is exceeded. Chilled samples should be examined regularly in routine practice to ensure the suitability of the metallurgical
structure of the prepared sample for the analysis method.
8.4.4.3 For a non-chilled sample, use a grinding or linishing machine to remove a layer approximately 1 mm in thickness from
the surface of the sample. Air cooling is recommended during grinding and, liquid coolants should not be used.
8.4.4.4 For irons that are subject to segregation effects, for example, high-phosphorus engineering iron, high-silicon ductile iron,
malleable iron, prepare the surfaces of two sides of the sample for analysis to obtain an average value.
8.4.4.5 Avoid overheating of the sample during surface preparation as this can result in surface crazing that will affect correctness
of analysis.
8.4.4.6 Care is required when preparing the surface of a thin coin-sample. A chuck should be designed specially to hold the sample
securely during grinding operations.
NOTE 3—A fixed-head grinder is preferred to a swing grinder for surface p
...


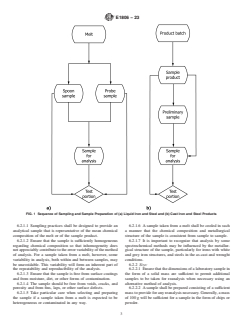




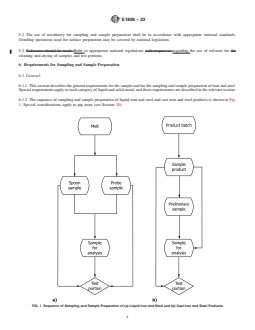
Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...