ASTM D5811-20
(Test Method)Standard Test Method for Strontium-90 in Water
Standard Test Method for Strontium-90 in Water
SIGNIFICANCE AND USE
5.1 This test method was developed to measure the concentration of 90Sr in non-process water samples. This test method may be used to determine the concentration of 90Sr in environmental samples.
SCOPE
1.1 This test method covers the determination of radioactive 90Sr in environmental water samples (for example, non-process and effluent waters) in the range of 0.037 Bq/L (1.0 pCi/L) or greater.
1.2 This test method has been used successfully with tap water. It is the user’s responsibility to ensure the validity of this test method for samples larger than 1 L and for waters of untested matrices.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific hazard statements, see Section 9.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Apr-2020
- Technical Committee
- D19 - Water
- Drafting Committee
- D19.04 - Methods of Radiochemical Analysis
Relations
- Effective Date
- 01-May-2020
- Effective Date
- 01-May-2020
- Effective Date
- 01-May-2020
- Effective Date
- 01-Feb-2019
- Effective Date
- 01-Feb-2018
- Effective Date
- 15-Dec-2017
- Effective Date
- 01-Feb-2016
- Effective Date
- 01-Jul-2015
- Effective Date
- 01-Jan-2015
- Effective Date
- 15-Jan-2014
- Effective Date
- 15-Jan-2014
- Effective Date
- 01-Apr-2013
- Effective Date
- 15-Jun-2012
- Effective Date
- 01-Jun-2012
- Effective Date
- 01-Jan-2011
Overview
ASTM D5811-20: Standard Test Method for Strontium-90 in Water provides a validated procedure to determine the concentration of radioactive strontium-90 (^90Sr) in environmental water samples, such as tap water, non-process water, and effluent waters. Developed by ASTM International, this test method specifies detection for concentrations as low as 0.037 Bq/L (1.0 pCi/L) or greater, supporting environmental monitoring and regulatory compliance efforts where accurate measurement of radioactive contaminants is essential.
The method uses solid phase extraction and beta proportional counting to isolate and measure ^90Sr activity in water, ensuring sensitive and reliable quantification. It is well-established for tap water and suitable for diverse matrices, given proper validation.
Key Topics
Scope and Purpose
- Applicable to environmental water samples, including tap water, groundwater, surface water, and effluents.
- Designed for the detection of strontium-90 at or above 0.037 Bq/L.
- Ensures results are reported in SI units for consistency in scientific and regulatory communication.
Test Method Summary
- Utilizes solid phase extraction to isolate strontium from the water matrix.
- Employs ion exchange and extraction chromatography techniques to purify strontium.
- Detection is performed via low background gas proportional beta counting, enabling precise measurement of beta particle emissions.
- Calibration and chemical yield are carefully monitored to maintain result accuracy.
Quality Control and Precision
- The method mandates use of quality control samples-such as laboratory control samples, method blanks, and duplicates-within designated sample batches.
- Requires calibration traceable to national standards (e.g., NIST, NPL) and verification of detector efficiency.
- Specifies procedures to account for potential chemical or radiochemical interferences.
- Recommends regular demonstration of laboratory and analyst proficiency to ensure precision and bias are within defined limits.
Safety and Regulatory Considerations
- Stresses the need for laboratory safety, especially when handling corrosive acids and radioactive materials.
- Users must ensure regulatory compliance for environmental, safety, and health requirements before performing the test.
Applications
Drinking Water Monitoring
- Ensures compliance with public health regulations by detecting radioactive strontium in potable water supplies.
- Useful for both routine surveillance and investigation following a nuclear incident.
Environmental Surveillance
- Essential for monitoring groundwater, surface waters, and effluents near nuclear facilities or waste sites.
- Supports assessment of radiological contamination and helps guide remediation or protection measures.
Regulatory Reporting
- Meets international and national standards for radiochemical water analysis.
- Produces data suitable for reporting to regulatory authorities or supporting environmental impact assessments.
Quality Assurance Programs
- Enables laboratories to contribute reliable data to quality assurance and accreditation programs in environmental testing.
Related Standards
- ASTM D1129 - Terminology Relating to Water
- ASTM D1193 - Specification for Reagent Water
- ASTM D1890 - Test Method for Beta Particle Radioactivity of Water
- ASTM D2777 - Practice for Determination of Precision and Bias of Applicable Test Methods
- ASTM D3370 - Practices for Sampling Water from Flowing Process Streams
- ASTM D3648 - Practices for the Measurement of Radioactivity
- ASTM D4448 - Guide for Sampling Ground-Water Monitoring Wells
- ASTM D5673 - Test Method for Elements in Water by Inductively Coupled Plasma-Mass Spectrometry
- ASTM D5847 - Practice for Writing Quality Control Specifications for Water Analysis
- ASTM D6001 - Guide for Direct-Push Groundwater Sampling
- ASTM D7282 - Practice for Set-up, Calibration, and Quality Control of Radioactivity Measurements
- ASTM D7902 - Terminology for Radiochemical Analyses
- ANSI N42.22 - Traceability of Radioactive Sources to National Standards
This comprehensive set of related standards positions ASTM D5811-20 as a core reference for laboratories and organizations focused on radioactive strontium analysis in water, ensuring robust, traceable methodologies for environmental and public health protection.
Buy Documents
ASTM D5811-20 - Standard Test Method for Strontium-90 in Water
REDLINE ASTM D5811-20 - Standard Test Method for Strontium-90 in Water
Get Certified
Connect with accredited certification bodies for this standard

NSF International
Global independent organization facilitating standards development and certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.

Kiwa BDA Testing
Building and construction product certification.
Sponsored listings
Frequently Asked Questions
ASTM D5811-20 is a standard published by ASTM International. Its full title is "Standard Test Method for Strontium-90 in Water". This standard covers: SIGNIFICANCE AND USE 5.1 This test method was developed to measure the concentration of 90Sr in non-process water samples. This test method may be used to determine the concentration of 90Sr in environmental samples. SCOPE 1.1 This test method covers the determination of radioactive 90Sr in environmental water samples (for example, non-process and effluent waters) in the range of 0.037 Bq/L (1.0 pCi/L) or greater. 1.2 This test method has been used successfully with tap water. It is the user’s responsibility to ensure the validity of this test method for samples larger than 1 L and for waters of untested matrices. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific hazard statements, see Section 9. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 This test method was developed to measure the concentration of 90Sr in non-process water samples. This test method may be used to determine the concentration of 90Sr in environmental samples. SCOPE 1.1 This test method covers the determination of radioactive 90Sr in environmental water samples (for example, non-process and effluent waters) in the range of 0.037 Bq/L (1.0 pCi/L) or greater. 1.2 This test method has been used successfully with tap water. It is the user’s responsibility to ensure the validity of this test method for samples larger than 1 L and for waters of untested matrices. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific hazard statements, see Section 9. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D5811-20 is classified under the following ICS (International Classification for Standards) categories: 13.060.50 - Examination of water for chemical substances; 13.060.60 - Examination of physical properties of water. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D5811-20 has the following relationships with other standards: It is inter standard links to ASTM D5811-08(2013), ASTM D7902-20, ASTM D1129-13(2020)e2, ASTM D4448-01(2019), ASTM D7902-18, ASTM D1890-15(2017), ASTM D7902-16, ASTM D5673-15, ASTM D1890-15, ASTM D7902-14e1, ASTM D7902-14, ASTM D4448-01(2013), ASTM D2777-12, ASTM D1890-05(2012), ASTM D3648-04(2011). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D5811-20 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D5811 − 20
Standard Test Method for
Strontium-90 in Water
This standard is issued under the fixed designation D5811; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope D3648Practices for the Measurement of Radioactivity
D4448GuideforSamplingGround-WaterMonitoringWells
1.1 This test method covers the determination of radioac-
90 D5673Test Method for Elements in Water by Inductively
tive Sr in environmental water samples (for example, non-
Coupled Plasma—Mass Spectrometry
process and effluent waters) in the range of 0.037 Bq/L (1.0
D5847Practice for Writing Quality Control Specifications
pCi/L) or greater.
for Standard Test Methods for Water Analysis
1.2 This test method has been used successfully with tap
D6001Guide for Direct-Push Groundwater Sampling for
water.Itistheuser’sresponsibilitytoensurethevalidityofthis
Environmental Site Characterization
test method for samples larger than 1 L and for waters of
D7282Practice for Set-up, Calibration, and Quality Control
untested matrices.
of Instruments Used for Radioactivity Measurements
1.3 The values stated in SI units are to be regarded as D7902Terminology for Radiochemical Analyses
2.2 Other Documents:
standard. No other units of measurement are included in this
standard. ANSI N42.22Traceability of Radioactive Sources to the
National Institute of Standards and Technology (NIST)
1.4 This standard does not purport to address all of the
and Associated Instrument Quality Control
safety concerns, if any, associated with its use. It is the
BIPM-5Decay Data Evaluation Project (DDEP)
responsibility of the user of this standard to establish appro-
NUDAT2
priate safety, health, and environmental practices and deter-
mine the applicability of regulatory limitations prior to use.
3. Terminology
For specific hazard statements, see Section 9.
3.1 Definitions:
1.5 This international standard was developed in accor-
3.1.1 For definitions of terms used in this standard, refer to
dance with internationally recognized principles on standard-
Terminology D1129.
ization established in the Decision on Principles for the
3.1.2 For definitions of terms used in this standard relating
Development of International Standards, Guides and Recom-
to radiochemical analysis, refer to Terminology D7902.
mendations issued by the World Trade Organization Technical
Barriers to Trade (TBT) Committee.
4. Summary of Test Method
2. Referenced Documents
4.1 This test method is based on the utilization of solid
phase extraction of strontium from water samples with detec-
2.1 ASTM Standards:
tionoftheradioactivestrontiumbygrossbetagasproportional
D1129Terminology Relating to Water
counting.
D1193Specification for Reagent Water
D1890Test Method for Beta Particle Radioactivity ofWater
4.2 An aliquant of the sample is measured into a beaker,
D2777Practice for Determination of Precision and Bias of
strontium carrier added, digested with nitric acid, sorbed on an
Applicable Test Methods of Committee D19 on Water
ion exchange column, eluted, evaporated to dryness, dissolved
D3370Practices for Sampling Water from Flowing Process
in nitric acid (8 M), selectively sorbed on a solid phase
Streams
extraction column, eluted with dilute nitric acid, dried on a
planchet. The sample test source (STS) is counted for beta
This test method is under the jurisdiction ofASTM Committee D19 on Water
radiation.
andisthedirectresponsibilityofSubcommitteeD19.04onMethodsofRadiochemi-
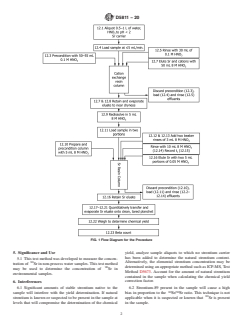
cal Analysis. 4.3 Fig. 1 shows a flow diagram for this test method.
Current edition approved May 1, 2020. Published June 2020. Originally
approved in 1995. Last previous edition approved in 2013 as D5811–08 (2013).
DOI: 10.1520/D5811-20. Available from Institute of Electrical and Electronics Engineers, Inc. (IEEE),
For referenced ASTM standards, visit the ASTM website, www.astm.org, or 445 Hoes Ln., Piscataway, NJ 08854-4141, http://www.ieee.org.
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Available from BIPM, Sèvres Cedex, France, https://www.bipm.org.
Standards volume information, refer to the standard’s Document Summary page on Available from National Nuclear Data Center at Brookhaven National
the ASTM website. Laboratory, W Princeton Ave, Yaphank, NY 11980, http://www.nndc.bnl.gov.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D5811 − 20
FIG. 1 Flow Diagram for the Procedure
5. Significance and Use yield, analyze sample aliquots to which no strontium carrier
has been added to determine the natural strontium content.
5.1 This test method was developed to measure the concen-
Alternatively, the elemental strontium concentration may be
tration of Sr in non-process water samples.This test method
90 determined using an appropriate method such as ICP-MS,Test
may be used to determine the concentration of Sr in
Method D5673. Account for the amount of natural strontium
environmental samples.
contained in the sample when calculating the chemical yield
correction factor.
6. Interferences
6.1 Significant amounts of stable strontium native to the 6.2 Strontium-89 present in the sample will cause a high
89 90
sample will interfere with the yield determination. If natural bias in proportion to the Sr/ Sr ratio. This technique is not
strontium is known or suspected to be present in the sample at applicable when it is suspected or known that Sr is present
levels that will compromise the determination of the chemical in the sample.
D5811 − 20
6.3 Strontium nitrate (Sr(NO ) ) is hygroscopic. This procedure to standardize the prepared strontium carrier: Care-
3 2
chemicalpropertymayadduncertaintyinthegravimetricyield fully pipet a 5.0 mL portion of the strontium carrier solution
determination of the chemical yield. onto a clean, dried, and tared planchet. Dry the planchet under
the same conditions used for the final evaporation in 12.20.
7. Apparatus
Allow the planchet to cool to room temperature and reweigh
the planchet to the nearest 0.0001 g. Divide the net weight by
7.1 Analytical Balance, 0.0001 g.
10. This result is the amount of strontium nitrate actually
7.2 Low Background Gas Proportional Beta Counting Sys-
added.Useanaverageofthreevaluesinthedenominatorofthe
tem.
chemical yield equation in 11.12 and 13.1. This value should
7.3 Ion Exchange Columns, 10 mL resin capacity, glass or
be within 3% of 12.08 mg/0.5 mL.
acid-resistantplastic.Anattachedreservoirofatleast50mLis
8.9 Strontium Extraction Chromatography Column,2mL
desirable.
bed volume consisting of an octanol solution of 4,4’(5’)-bis
7.4 Planchets, stainless steel to match calibration source.
(t-butyl-cyclohexano)-18-crown-6 sorbed on an inert poly-
meric support.
8. Reagents and Materials
8.10 Strontium-90 Standardizing Solution—Solution with a
8.1 Purity of Reagents—Reagent grade chemicals shall be
final concentration typically ranging from 85 to 125 Bq/mL
used in all tests. Unless otherwise indicated, it is intended that
and less than 0.1 mg of stable strontium, traceable to the SI
all reagents shall conform to specifications of the Committee
through a national metrology institute (NMI), such as National
on Analytical Reagents of the American Chemical Society.
Institute of Standards and Technology (NIST).
Other grades may be used, provided it is first ascertained that
the reagent is of sufficiently high purity to permit its use
9. Hazards
without lessening the accuracy of the determination. Some
9.1 Use extreme caution when handling all acids. They are
reagents, even those of high purity, may contain naturally-
extremely corrosive and skin contact could result in severe
occurring radioactivity, such as isotopes of uranium, radium,
burns.
actinium, thorium, rare earths, potassium compounds, or arti-
9.2 When diluting concentrated acids, always use safety
ficially produced radionuclides, or combinations thereof.
Consequently, when such reagents are used in the analysis of glasses and protective clothing, and add the acid to the water.
low-radioactivity samples, the activity of the reagents should
10. Sampling
be determined under analytical conditions that are as close as
practicable to those used for the test sample. The activity
10.1 Collect a sample in accordance with Practices D3370,
contributed by the reagents should be accounted for and
Guide D4448, and Guide D6001, or other documented proce-
applied as a correction when calculating the test sample result.
dure.
8.2 Purity of Water—Unless otherwise indicated, reference
11. Calibration
towatershallbeunderstoodtomeanreagentwaterconforming
to Specification D1193, Type III.
11.1 Calibrate the low background gas proportional beta
counting system in accordance with Practice D7282, Sections
8.3 Cation Exchange Resin, 100 to 200 mesh, hydrogen
14, 15, and 16. Prepare a set of three working calibration
form. 8 % cross linked, analytical grade.
standards (WCS) according to the procedure outlined in the
8.4 Nitric Acid (sp gr 1.42)—Concentrated HNO .
subsequent steps.
8.5 Nitric Acid (8 M HNO )—Add 500 mL of concentrated
11.2 Pipet 0.5 mL of strontium carrier into a small beaker.
HNO to 400 mL of water. Dilute to 1 L with water.
11.3 Add 1 mL of traceable Sr solution and evaporate to
8.6 Nitric Acid (0.1 HNO )—Add 6.4 mL of concentrated
near dryness on a hot plate.
HNO to 600 mL of water. Dilute to 1 L with water.
11.4 Redissolve the residual in 5 mL of 8 M nitric acid.
8.7 Nitric Acid (0.05 M HNO )—Add 3.2 mL of concen-
11.5 Follow the steps described in 12.10 through 12.23.
trated HNO to 600 mL of water. Dilute to 1 L with water.
11.6 Count to accumulate 10 000 net counts in the counting
8.8 Strontium Carrier (10 g/L)—Preferably use 10000
period. Counting should be completed within3hof column
µg/mL ICP standard. Alternatively, dissolve 24.16 g dry
elution. Record the time and date of the midpoint of this
strontium nitrate (Sr(NO ) ) in water, add 20 mLconcentrated
3 2
countingperiodast .CounteachWCStwice,onceforthisstep
nitric acid, and dilute with water to 1 L. Use the following 2
having a counting date designated as t and a second time as
specified below.
Stainless steel planchets available commercially have been found satisfactory.
11.7 Calculate the net count rate of the WCS at time t
ACS Reagent Chemicals, Specifications and Procedures for Reagents and (R ) by subtracting the instrument background count rate
n(2)
Standard-Grade Reference Materials, American Chemical Society, Washington,
from the gross count rate.
DC. For suggestions on the testing of reagents not listed by theAmerican Chemical
Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset,
U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharma-
copeial Convention, Inc. (USPC), Rockville, MD. Sr resin available commercially have been found satisfactory.
D5811 − 20
11.8 Store the WCS for at least 7 days to allow for Y 12.5 Rinse the column with 25 to 30 mL of 0.1 M HNO .
ingrowth.
12.6 Properly dispose of the feed and rinse.
11.9 RecounttheWCStoamass10000countsinacounting
12.7 Elute the strontium (and other cations) with 50 mL of
period. Record the time and date of the midpoint of this count
8 M HNO into a 150 mL beaker.
period as t .
12.8 Evaporate the eluate to near dryness on a hot plate in a
11.10 Calculate the net count rate of the second count at
fume hood. The residue will dissolve more easily in the next
time t (R ) by subtracting the instrument background count
stepiftheevaporationisstoppedjustasthesamplestartstogo
3 n(3)
rate from the gross count rate.
dry.
11.11 Calculate the Sr detection efficiency, ε , and 12.9 Dissolve the residues in 5 mL of8MHNO.If
Sr
the Y detection efficiency, ε , for each WCS using the necessary, cover with a watchglass and heat gently to facilitate
Y
equations presented below. Calculate the mean and standard complete dissolution.
deviation of the three ε and ε values. Use the relative
Sr Y
12.10 Prepare a strontium extraction chromatography col-
standard deviation of these parameters to estimate the relative
umn by removing the bottom plug and the cap. Press the top
uncertainty of the ingrowth efficiency factor, (defined in Eq 5),
frit down snugly to the resin surface using a glass rod (or
u(ε) and used in Eq 7.
r I
equivalent) and let the water drain out.Add 5 mLof HNO (8
11.12 Effıciency Calculations— Sr detection efficiency ε : M) and let the solution drain by gravity.
Sr
12.11 Carefully transfer the sample solution to the reservoir
R 3IF 2 R 3IF
~ ! ~ !
n~2! 3 n~3! 2
ε 5 (1)
Sr ofthecolumn.Addhalfandletthesolutiondrainbeforeadding
Y 3A 3 IF 2 IF
~ !
Sr C 2 3 2
~ !
the second half.
Y detection efficiency ε :
Y
12.12 Rinse the beaker with 3 mLof8MHNO and add to
R 2 R
the column after the feed has passed through.
n~3! n~2!
ε 5 (2)
Y
Y 3A 3 IF 2 IF
~ !
Sr C 2 3 2
~ !
12.13 Repeat 12.12.
where: 12.14 Rinse the column with 10 mL of8MHNO .
A = activity of Sr in becquerels (Bq) at the time of the
12.15 Record the end time of the last rinse as the time
C(2)
90 90
first count of the calibration mount,
of Y separation (start of Y ingrowth, t ).
IF = ingrowth factor for Y at the midpoint of the count
12.16 Elutethestrontiumwithtwo5mLportionsof0.05M
2λ t 2 t
~ !
Y 2 1
at time t , 12e ,
HNO into a suitable container (for example, a liquid scintil-
90 3
IF = ingrowth factor for Y at the midpoint of the count
lation counting vial or centrifuge tube).
2λ ~t 2 t !
Y 3 1
at time t , 12e ,
90 –1 9
12.17 Clean a planchet with a paper towel moistened with
λ = decay constant for Y (0.2599 d ),
Y
alcohol. Wipe the planchet and let it dry.
R = net count rate of the calibration test source at the
n(2)
midpoint of the first count, in counts per second,
12.18 Weightheplanchettothenearest0.0001gandrecord
R = net count rate of cali
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D5811 − 08 (Reapproved 2013) D5811 − 20
Standard Test Method for
Strontium-90 in Water
This standard is issued under the fixed designation D5811; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This test method covers the determination of radioactive Sr in environmental water samples (for example, non-process and
effluent waters) in the range of 0.037 Bq/L (1.0 pCi/L) or greater.
1.2 The values stated in SI units are to be regarded as the standard. The values given in parentheses are for information only.
1.2 This test method has been used successfully with tap water. It is the user’suser’s responsibility to ensure the validity of this
test method for samples larger than 1 L and for waters of untested matrices.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use. For specific hazard statements, see Section 9.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D1129 Terminology Relating to Water
D1193 Specification for Reagent Water
D1890 Test Method for Beta Particle Radioactivity of Water
D2777 Practice for Determination of Precision and Bias of Applicable Test Methods of Committee D19 on Water
D3370 Practices for Sampling Water from Flowing Process Streams
D3648 Practices for the Measurement of Radioactivity
D4448 Guide for Sampling Ground-Water Monitoring Wells
D5673 Test Method for Elements in Water by Inductively Coupled Plasma—Mass Spectrometry
D5847 Practice for Writing Quality Control Specifications for Standard Test Methods for Water Analysis
D6001 Guide for Direct-Push Groundwater Sampling for Environmental Site Characterization
D7282 Practice for Set-up, Calibration, and Quality Control of Instruments Used for Radioactivity Measurements
D7902 Terminology for Radiochemical Analyses
2.2 Other Documents:
ANSI N42.22 Traceability of Radioactive Sources to the National Institute of Standards and Technology (NIST) and Associated
Instrument Quality Control
BIPM-5 Decay Data Evaluation Project (DDEP)
NUDAT2
This test method is under the jurisdiction of ASTM Committee D19 on Water and is the direct responsibility of Subcommittee D19.04 on Methods of Radiochemical
Analysis.
Current edition approved June 15, 2013May 1, 2020. Published July 2013June 2020. Originally approved in 1995. Last previous edition approved in 20082013 as
D5811 – 08.D5811 – 08 (2013). DOI: 10.1520/D5811-08R13.10.1520/D5811-20.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’sstandard’s Document Summary page on the ASTM website.
Available from Institute of Electrical and Electronics Engineers, Inc. (IEEE), 445 Hoes Ln., Piscataway, NJ 08854-4141, http://www.ieee.org.
Available from BIPM, Sèvres Cedex, France, https://www.bipm.org.
Available from National Nuclear Data Center at Brookhaven National Laboratory, W Princeton Ave, Yaphank, NY 11980, http://www.nndc.bnl.gov.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D5811 − 20
3. Terminology
3.1 Definitions—Definitions: For definitions of terms used in this test method, refer to Terminology D1129.
3.1.1 For definitions of terms used in this standard, refer to Terminology D1129.
3.1.2 For definitions of terms used in this standard relating to radiochemical analysis, refer to Terminology D7902.
4. Summary of Test Method
4.1 This test method is based on the utilization of solid phase extraction of strontium from water samples with detection of the
radioactive strontium by gross beta gas proportional counting.
4.2 An aliquant of the sample is measured into a beaker, strontium carrier added, digested with nitric acid, sorbed on an ion
exchange column, eluted, evaporated to dryness, dissolved in nitric acid (8M), (8 M), selectively sorbed on a solid phase extraction
column, eluted with dilute nitric acid, dried on a planchet, and planchet. The sample test source (STS) is counted for beta radiation.
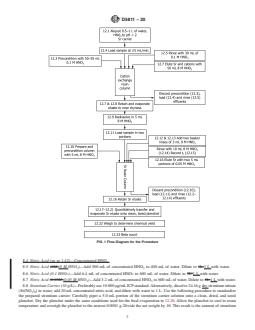
4.3 Fig. 1 shows a flow diagram for this test method.
5. Significance and Use
5.1 This test method was developed to measure the concentration of Sr in non-process water samples. This test method may
be used to determine the concentration of Sr in environmental samples.
6. Interferences
6.1 Significant amounts of stable strontium present innative to the sample will interfere with the yield determination. If it natural
strontium is known or suspected that natural strontium is to be present in the sample at levels that will compromise the
determination of the chemical yield, blankanalyze sample aliquots to which no strontium carrier is added shall be analyzed has
been added to determine the natural strontium content. The Alternatively, the elemental strontium concentration may be determined
using an appropriate method such as ICP-MS, Test Method D5673. Account for the amount of natural strontium contained in the
sample shall be reflected when calculating the chemical yield correction factor.
89 90
6.2 Strontium-89 present in the sample will cause a high bias in proportion to the Sr/ Sr ratio. This technique is not
applicable when it is suspected or known that Sr is present in the sample.
6.3 Strontium nitrate (Sr(NO ) ) is hygroscopic. This chemical property may add uncertainty in the gravimetric yield
3 2
determination.determination of the chemical yield.
7. Apparatus
7.1 Analytical Balance, 0.0001 g.
7.2 Low Background Gas Proportional Beta Counting System.
7.3 Ion Exchange Columns, 10 mL resin capacity, glass or acid-resistant plastic. An attached reservoir of at least 50 mL is
desirable.
7.4 Planchets, stainless steel to match calibration source.
8. Reagents and Materials
8.1 Purity of Reagents—Reagent grade chemicals shall be used in all tests. Unless otherwise indicated, it is intended that all
reagents shall conform to specifications of the Committee on Analytical Reagents of the American Chemical Society. Other grades
may be used, provided it is first ascertained that the reagent is of sufficiently high purity to permit its use without lessening the
accuracy of the determination. Reagent blanks shall be run with all determinations.Some reagents, even those of high purity, may
contain naturally-occurring radioactivity, such as isotopes of uranium, radium, actinium, thorium, rare earths, potassium
compounds, or artificially produced radionuclides, or combinations thereof. Consequently, when such reagents are used in the
analysis of low-radioactivity samples, the activity of the reagents should be determined under analytical conditions that are as close
as practicable to those used for the test sample. The activity contributed by the reagents should be accounted for and applied as
a correction when calculating the test sample result.
8.2 Purity of Water—Unless otherwise indicated, reference to water shall be understood to mean reagent water conforming to
Specification D1193, Type III.
8.3 Cation Exchange Resin, 100 to 200 mesh, hydrogen form. 8% 8 % cross linked, analytical grade.
Stainless steel planchets available commercially have been found satisfactory.
Reagent Chemicals, American Chemical Society Specifications,ACS Reagent Chemicals, Specifications and Procedures for Reagents and Standard-Grade Reference
Materials, American Chemical Society, Washington, DC. For Suggestionssuggestions on the testing of reagents not listed by the American Chemical Society, see
AnnualAnalar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharmacopeial
Convention, Inc. (USPC), Rockville, MD.
D5811 − 20
FIG. 1 Flow Diagram for the Procedure
8.4 Nitric Acid (sp gr 1.42)—Concentrated HNO .
8.5 Nitric Acid (8M (8 M HNO )—Add 500 mL of concentrated HNO to 400 mL of water. Dilute to 1L 1 L with water.
3 3
8.6 Nitric Acid (0.1 HNO )—Add 6.4 mL of concentrated HNO to 600 mL of water. Dilute to 1L 1 L with water.
3 3
8.7 Nitric Acid (0.05M (0.05 M HNO )—Add 3.2 mL of concentrated HNO to 600 mL of water. Dilute to 1L 1 L with water.
3 3
8.8 Strontium Carrier (10 g/L)—Preferably use 10 000 μg/mL ICP standard. Alternatively, dissolve 24.16 g dry strontium nitrate
(Sr(NO ) ) in water, add 20 mL concentrated nitric acid, and dilute with water to 1 L. Use the following procedure to standardize
3 2
the prepared strontium carrier: Carefully pipet a 5.0 mL portion of the strontium carrier solution onto a clean, dried, and tared
planchet. Dry the planchet under the same conditions used for the final evaporation in 12.20. Allow the planchet to cool to room
temperature and reweigh the planchet to the nearest 0.0001 g. Divide the net weight by 10. This result is the amount of strontium
D5811 − 20
nitrate actually added. Use an average of three values in the denominator of the recovery chemical yield equation in 11.12 and 13.1.
This value should be within 3 % of 12.08 mg/0.5 mL.
8.9 Strontium Extraction Chromatography Column, 2 mL bed volume consisting of an octanol solution of 4,4’(5’)-bis
(t-butyl-cyclohexano)-18-crown-6-sorbed 4,4’(5’)-bis (t-butyl-cyclohexano)-18-crown-6 sorbed on an inert polymeric support.
8.10 Strontium-90 Standardizing Solution—Traceable to a national standard body such as National Institute of Standards and
Technology or National Physical Laboratory solution with less than 0.1 mg of stable strontium per mL of final solution with a
typical concentration range from 85 to 125 Bq/mL.Solution with a final concentration typically ranging from 85 to 125 Bq/mL and
less than 0.1 mg of stable strontium, traceable to the SI through a national metrology institute (NMI), such as National Institute
of Standards and Technology (NIST).
9. Hazards
9.1 Use extreme caution when handling all acids. They are extremely corrosive and skin contact could result in severe burns.
9.2 When diluting concentrated acids, always use safety glasses and protective clothing, and add the acid to the water.
10. Sampling
10.1 Collect a sample in accordance with PracticePractices D3370, Guide D4448, and Guide D6001, or other documented
procedure.
11. Calibration
11.1 Calibrate the low background gas proportional beta counting system in accordance with Practice D7282. , Sections 14, 15,
and 16. Prepare a set of three working calibration samples standards (WCS) according to the calibration procedure outlined in the
subsequent steps.
11.2 Pipet 0.5 mL of strontium carrier into a small beaker.
11.3 Add 1 mL of traceable Sr solution and evaporate to near dryness on a hot plate.
11.4 Redissolve the residual in 5 mL of 8M 8 M nitric acid.
11.5 Follow the steps described in 12.10 through 12.23.
11.6 Count to accumulate 10 000 net counts in the counting period. Counting should be completed within 3 h of column elution.
Record the time and date of the midpoint of this counting period as t . Count each sample mount WCS twice, once for this step
having a counting date designated as t and a second time as specified below.
11.7 Calculate the net count rate of the countWCS at time t (R ) by subtracting the instrument background count rate from
2 n(2)
the gross count rate.
11.8 Store the calibration mount WCS for at least 7 days to allow for Y ingrowth.
11.9 Recount the calibration mount WCS to amass 10 000 counts in a counting period. Record the time and date of the midpoint
of this count period as t .
11.10 Calculate the net count rate of the second count at time t (R ) by subtracting the instrument background count rate from
3 n(3)
the gross count rate.
90 90
11.11 Calculate the Sr detection efficiency, ε , and the Y detection efficiency, ε , for each calibration mount WCS using the
Sr Y
equations presented below. Calculate the mean and standard deviation of the three ε and ε values. Use the relative standard
Sr Y
deviation of these parameters to estimate the relative uncertainty of the ingrowth efficiency factor, (defined in Eq 5), u (ε ) and used
r I
in Eq 7.
11.12 Effıciency Calculations— Sr detection efficiency ε :
Sr
R 3IF 2 R 3IF
~ ! ~ !
n~2! 3 n~3! 2
ε 5 (1)
Sr
Y 3A 3~IF 2 IF !
Sr C~2! 3 2
R 3IF 2 R 3IF
~ ! ~ !
n~2! 3 n~3! 2
ε 5 (1)
Sr
Y 3A 3 IF 2 IF
~ !
Sr C 2 3 2
~ !
Y detection efficiency ε :
Y
R 2 R
n~3! n~2!
ε 5 (2)
Y
Y 3A 3 IF 2 IF
~ !
Sr C 2 3 2
~ !
The sole source of supply of the apparatus known to the committee at this time is Sr Resin available from Eichrom Technologies, Inc. If you are aware of alternative
suppliers, please provide this information to ASTM International Headquarters. Your comments will receive careful consideration at a meeting of the responsible technical
committee,Sr resin available commercially have been found satisfactory. which you may attend.
D5811 − 20
R 2 R
n 3 n 2
~ ! ~ !
ε 5 (2)
Y
Y 3A 3~IF 2 IF !
Sr C 2 3 2
~ !
where:
A = activity of Sr in becquerels (Bq) at the time of the first count of the calibration mount,
C(2)
2 λ 3 t 2t
@ ~ !#
Y 2 1
IF = ingrowth factor for Y at the midpoint of the count at time t , e
2 2
2λ ~t 2 t !
Y 2 1
IF = ingrowth factor for Y at the midpoint of the count at time t , 12e ,
2 2
2@λ 3~t 2t !#
Y 3 1
IF = ingrowth factor for Y at the midpoint of the count at time t , e
3 3
2λ t 2 t
~ !
Y 3 1
IF = ingrowth factor for Y at the midpoint of the count at time t , 12e ,
3 3
90 –1 9
λ = decay constant for Y (0.2600 d ),
Y
90 –1 9
λ = decay constant for Y (0.2599 d ),
Y
R = net count rate of the calibration test source at the midpoint of the first count, in counts per second,
n(2)
R = net count rate of calibration test source at the midpoint of the second count, in counts per second,
n(3)
t = date and time of Y separation,
t = date and time of midpoint of first count,
t = date and time of midpoint of second count.
Y = fractional chemical yield of strontium carrier (see Eq 4).
Sr
NOTE 1—The time differences (t − t ) and (t − t ) are expressed in days.
2 1 3 1
12. Procedure
12.1 Add 0.5 mL of strontium carrier to a maximum of 1 L of sample. Add 1 mL of 8M 8 M HNO per 100 mL of sample and
mix. Bring sample to a boil for 30 min and then cool.
12.2 Prepare a cation exchange column containing 10 mL of cation exchange resin.
12.3 Precondition the column by passing 50 to 55 mL of 0.1M 0.1 M HNO through the column.
12.4 Pass the sample through the column at a rate of not more than 5 mL/min.
12.5 Rinse the column with 25 to 30 mL of 0.1M 0.1 M HNO .
12.6 Properly dispose of the feed and rinse.
12.7 Elute the strontium (and other cations) with 50 mL of 8M 8 M HNO into a 150 mL beaker.
12.8 Evaporate the eluate to near dryness on a hot plate in a fume hood. The residue will dissolve more easily in the next step
if the evaporation is stopped just as the sample starts to go dry.
12.9 Dissolve the saltsresidues in 5 mL of 8M 8 M HNO . If necessary, cover with a watchglass and heat gently to facilitate
complete dissolution.
12.10 Prepare a strontium extraction chromatography column by removing the bottom plug and the cap. Press the top frit down
snugly to the resin surface using a glass rod (or equivalent) and let the water drain out. Add 5 mL of HNO (8M) (8 M) and let
the solution drain by gravity.
12.11 Car
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...