ASTM D2892-23
(Test Method)Standard Test Method for Distillation of Crude Petroleum (15-Theoretical Plate Column)
Standard Test Method for Distillation of Crude Petroleum (15-Theoretical Plate Column)
SIGNIFICANCE AND USE
5.1 This test method is one of a number of tests conducted on a crude oil to determine its value. It provides an estimate of the yields of fractions of various boiling ranges and is therefore valuable in technical discussions of a commercial nature.
5.2 This test method corresponds to the standard laboratory distillation efficiency referred to as 15/5. The fractions produced can be analyzed as produced or combined to produce samples for analytical studies, engineering, and product quality evaluations. The preparation and evaluation of such blends is not part of this test method.
5.3 This test method can be used as an analytical tool for examination of other petroleum mixtures with the exception of LPG, very light naphthas, and mixtures with initial boiling points above 400 °C.
SCOPE
1.1 This test method covers the procedure for the distillation of stabilized crude petroleum (see Note 1) to a final cut temperature of 400 °C Atmospheric Equivalent Temperature (AET). This test method employs a fractionating column having an efficiency of 14 to 18 theoretical plates operated at a reflux ratio of 5:1. Performance criteria for the necessary equipment is specified. Some typical examples of acceptable apparatus are presented in schematic form. This test method offers a compromise between efficiency and time in order to facilitate the comparison of distillation data between laboratories.
Note 1: Defined as having a Reid vapor pressure less than 82.7 kPa (12 psi).
1.2 This test method details procedures for the production of a liquefied gas, distillate fractions, and residuum of standardized quality on which analytical data can be obtained, and the determination of yields of the above fractions by both mass and volume. From the preceding information, a graph of temperature versus mass % distilled can be produced. This distillation curve corresponds to a laboratory technique, which is defined at 15/5 (15 theoretical plate column, 5:1 reflux ratio) or TBP (true boiling point).
1.3 This test method can also be applied to any petroleum mixture except liquefied petroleum gases, very light naphthas, and fractions having initial boiling points above 400 °C.
1.4 This test method contains the following annexes and appendixes:
1.4.1 Annex A1—Test Method for the Determination of the Efficiency of a Distillation Column,
1.4.2 Annex A2—Test Method for the Determination of the Dynamic Holdup of a Distillation Column,
1.4.3 Annex A3—Test Method for the Determination of the Heat Loss in a Distillation Column (Static Conditions),
1.4.4 Annex A4—Test Method for the Verification of Temperature Sensor Location,
1.4.5 Annex A5—Test Method for Determination of the Temperature Response Time,
1.4.6 Annex A6—Practice for the Calibration of Sensors,
1.4.7 Annex A7—Test Method for the Verification of Reflux Dividing Valves,
1.4.8 Annex A8—Practice for Conversion of Observed Vapor Temperature to Atmospheric Equivalent Temperature (AET),
1.4.9 Appendix X1—Test Method for Dehydration of a Sample of Wet Crude Oil, and
1.4.10 Appendix X2—Practice for Performance Check.
1.5 The values stated in SI units are to be regarded as standard. The values given in parentheses after SI units are provided for information only and are not considered standard.
1.6 WARNING—Mercury has been designated by many regulatory agencies as a hazardous substance that can cause serious medical issues. Mercury, or its vapor, has been demonstrated to be hazardous to health and corrosive to materials. Use Caution when handling mercury and mercury-containing products. See the applicable product Safety Data Sheet (SDS) for additional information. The potential exists that selling mercury or mercury-containing products, or both, is prohibited by local or national law. Users must determine legality of sales in their location.
1.7 This standard does not purport to address all of the safety concerns, if any, asso...
General Information
- Status
- Published
- Publication Date
- 31-Oct-2023
- Technical Committee
- D02 - Petroleum Products, Liquid Fuels, and Lubricants
- Drafting Committee
- D02.08 - Volatility
Relations
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Mar-2024
- Effective Date
- 01-Dec-2023
- Effective Date
- 01-Jul-2023
- Effective Date
- 01-Jul-2023
- Effective Date
- 15-Jul-2017
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Nov-2023
Overview
ASTM D2892-23 is the Standard Test Method for Distillation of Crude Petroleum (15-Theoretical Plate Column), developed by ASTM International. This internationally recognized method details the laboratory-scale distillation procedure for stabilized crude petroleum using a fractionating column with 14 to 18 theoretical plates at a 5:1 reflux ratio. The technique measures the yields of different boiling range fractions up to 400 °C (Atmospheric Equivalent Temperature, AET), allowing laboratories to estimate the boiling point distribution and product fractions of crude oils with high precision and reproducibility.
Key Topics
Purpose and Significance:
- Provides a standardized method to determine the boiling range distribution of crude oil, which is crucial for technical evaluation and commercial negotiations in oil refining.
- Establishes laboratory distillation efficiency, commonly referred to as the 15/5 method (15 theoretical plate column, 5:1 reflux ratio).
Scope and Applicability:
- Designed for stabilized crude petroleum (Reid vapor pressure less than 82.7 kPa/12 psi).
- Excludes liquefied petroleum gases (LPG), very light naphthas, and mixtures with initial boiling points above 400 °C.
- Enables production of standardized liquefied gas, distillate fractions, and residue for further analysis.
Procedural Highlights:
- Fractionation yields are determined by both mass and volume.
- Production of temperature versus mass percent distilled curves (TBP curves – true boiling point) for analytical and engineering purposes.
- Equipment performance criteria and verification procedures ensure reliability of results.
Annexes and Appendices:
- Methods for verifying distillation column efficiency, dynamic holdup, heat loss, temperature sensor calibration and placement, reflux dividing valves, and dehydration of crude oil samples.
- Guidelines for performance checks and quality control are included.
Applications
Crude Oil Evaluation:
- Used by refineries, laboratories, and petroleum product traders to assess crude oil qualities, predict yields, and optimize refining processes.
- Facilitates technical discussions around crude oil value and product output.
Product Quality Control:
- Fractional distillation data supports further analytical studies, engineering assessments, and product quality evaluations.
Research and Development:
- Analytical tool for the study of boiling range distributions in various petroleum mixtures, excluding ultra-light and high-boiling-point components.
- Supports blend studies and aids in the calibration of analytical instrumentation.
Inter-Laboratory Data Comparison:
- Enables consistent comparison of distillation data across laboratories due to harmonized efficiency and procedural criteria.
Related Standards
- ASTM D2887 – Boiling Range Distribution of Petroleum Fractions by Gas Chromatography.
- ASTM D4006 – Water in Crude Oil by Distillation.
- ASTM D1298 – Density, Relative Density, or API Gravity of Crude Petroleum and Liquid Petroleum Products by Hydrometer Method.
- ASTM D4052 – Density, Relative Density, and API Gravity of Liquids by Digital Density Meter.
- ASTM D4057 – Manual Sampling of Petroleum and Petroleum Products.
- ASTM D4177 – Automatic Sampling of Petroleum and Petroleum Products.
- ASTM D6729, D6730, D6733 – Analysis of individual components in petroleum products via high-resolution gas chromatography.
Practical Value
Implementing ASTM D2892-23 ensures reliable, reproducible distillation data crucial for quality control, asset evaluation, and process optimization in the petroleum industry. Its standardized approach supports technical decision-making and commercial activities by providing consistent, high-quality data on boiling point distribution and product fraction yields. Adopting this distillation method is essential for any laboratory or refinery involved in crude oil analysis and refining.
Buy Documents
ASTM D2892-23 - Standard Test Method for Distillation of Crude Petroleum (15-Theoretical Plate Column)
REDLINE ASTM D2892-23 - Standard Test Method for Distillation of Crude Petroleum (15-Theoretical Plate Column)
Get Certified
Connect with accredited certification bodies for this standard

ABS Quality Evaluations Inc.
American Bureau of Shipping quality certification.

Element Materials Technology
Materials testing and product certification.

ABS Group Brazil
ABS Group certification services in Brazil.
Sponsored listings
Frequently Asked Questions
ASTM D2892-23 is a standard published by ASTM International. Its full title is "Standard Test Method for Distillation of Crude Petroleum (15-Theoretical Plate Column)". This standard covers: SIGNIFICANCE AND USE 5.1 This test method is one of a number of tests conducted on a crude oil to determine its value. It provides an estimate of the yields of fractions of various boiling ranges and is therefore valuable in technical discussions of a commercial nature. 5.2 This test method corresponds to the standard laboratory distillation efficiency referred to as 15/5. The fractions produced can be analyzed as produced or combined to produce samples for analytical studies, engineering, and product quality evaluations. The preparation and evaluation of such blends is not part of this test method. 5.3 This test method can be used as an analytical tool for examination of other petroleum mixtures with the exception of LPG, very light naphthas, and mixtures with initial boiling points above 400 °C. SCOPE 1.1 This test method covers the procedure for the distillation of stabilized crude petroleum (see Note 1) to a final cut temperature of 400 °C Atmospheric Equivalent Temperature (AET). This test method employs a fractionating column having an efficiency of 14 to 18 theoretical plates operated at a reflux ratio of 5:1. Performance criteria for the necessary equipment is specified. Some typical examples of acceptable apparatus are presented in schematic form. This test method offers a compromise between efficiency and time in order to facilitate the comparison of distillation data between laboratories. Note 1: Defined as having a Reid vapor pressure less than 82.7 kPa (12 psi). 1.2 This test method details procedures for the production of a liquefied gas, distillate fractions, and residuum of standardized quality on which analytical data can be obtained, and the determination of yields of the above fractions by both mass and volume. From the preceding information, a graph of temperature versus mass % distilled can be produced. This distillation curve corresponds to a laboratory technique, which is defined at 15/5 (15 theoretical plate column, 5:1 reflux ratio) or TBP (true boiling point). 1.3 This test method can also be applied to any petroleum mixture except liquefied petroleum gases, very light naphthas, and fractions having initial boiling points above 400 °C. 1.4 This test method contains the following annexes and appendixes: 1.4.1 Annex A1—Test Method for the Determination of the Efficiency of a Distillation Column, 1.4.2 Annex A2—Test Method for the Determination of the Dynamic Holdup of a Distillation Column, 1.4.3 Annex A3—Test Method for the Determination of the Heat Loss in a Distillation Column (Static Conditions), 1.4.4 Annex A4—Test Method for the Verification of Temperature Sensor Location, 1.4.5 Annex A5—Test Method for Determination of the Temperature Response Time, 1.4.6 Annex A6—Practice for the Calibration of Sensors, 1.4.7 Annex A7—Test Method for the Verification of Reflux Dividing Valves, 1.4.8 Annex A8—Practice for Conversion of Observed Vapor Temperature to Atmospheric Equivalent Temperature (AET), 1.4.9 Appendix X1—Test Method for Dehydration of a Sample of Wet Crude Oil, and 1.4.10 Appendix X2—Practice for Performance Check. 1.5 The values stated in SI units are to be regarded as standard. The values given in parentheses after SI units are provided for information only and are not considered standard. 1.6 WARNING—Mercury has been designated by many regulatory agencies as a hazardous substance that can cause serious medical issues. Mercury, or its vapor, has been demonstrated to be hazardous to health and corrosive to materials. Use Caution when handling mercury and mercury-containing products. See the applicable product Safety Data Sheet (SDS) for additional information. The potential exists that selling mercury or mercury-containing products, or both, is prohibited by local or national law. Users must determine legality of sales in their location. 1.7 This standard does not purport to address all of the safety concerns, if any, asso...
SIGNIFICANCE AND USE 5.1 This test method is one of a number of tests conducted on a crude oil to determine its value. It provides an estimate of the yields of fractions of various boiling ranges and is therefore valuable in technical discussions of a commercial nature. 5.2 This test method corresponds to the standard laboratory distillation efficiency referred to as 15/5. The fractions produced can be analyzed as produced or combined to produce samples for analytical studies, engineering, and product quality evaluations. The preparation and evaluation of such blends is not part of this test method. 5.3 This test method can be used as an analytical tool for examination of other petroleum mixtures with the exception of LPG, very light naphthas, and mixtures with initial boiling points above 400 °C. SCOPE 1.1 This test method covers the procedure for the distillation of stabilized crude petroleum (see Note 1) to a final cut temperature of 400 °C Atmospheric Equivalent Temperature (AET). This test method employs a fractionating column having an efficiency of 14 to 18 theoretical plates operated at a reflux ratio of 5:1. Performance criteria for the necessary equipment is specified. Some typical examples of acceptable apparatus are presented in schematic form. This test method offers a compromise between efficiency and time in order to facilitate the comparison of distillation data between laboratories. Note 1: Defined as having a Reid vapor pressure less than 82.7 kPa (12 psi). 1.2 This test method details procedures for the production of a liquefied gas, distillate fractions, and residuum of standardized quality on which analytical data can be obtained, and the determination of yields of the above fractions by both mass and volume. From the preceding information, a graph of temperature versus mass % distilled can be produced. This distillation curve corresponds to a laboratory technique, which is defined at 15/5 (15 theoretical plate column, 5:1 reflux ratio) or TBP (true boiling point). 1.3 This test method can also be applied to any petroleum mixture except liquefied petroleum gases, very light naphthas, and fractions having initial boiling points above 400 °C. 1.4 This test method contains the following annexes and appendixes: 1.4.1 Annex A1—Test Method for the Determination of the Efficiency of a Distillation Column, 1.4.2 Annex A2—Test Method for the Determination of the Dynamic Holdup of a Distillation Column, 1.4.3 Annex A3—Test Method for the Determination of the Heat Loss in a Distillation Column (Static Conditions), 1.4.4 Annex A4—Test Method for the Verification of Temperature Sensor Location, 1.4.5 Annex A5—Test Method for Determination of the Temperature Response Time, 1.4.6 Annex A6—Practice for the Calibration of Sensors, 1.4.7 Annex A7—Test Method for the Verification of Reflux Dividing Valves, 1.4.8 Annex A8—Practice for Conversion of Observed Vapor Temperature to Atmospheric Equivalent Temperature (AET), 1.4.9 Appendix X1—Test Method for Dehydration of a Sample of Wet Crude Oil, and 1.4.10 Appendix X2—Practice for Performance Check. 1.5 The values stated in SI units are to be regarded as standard. The values given in parentheses after SI units are provided for information only and are not considered standard. 1.6 WARNING—Mercury has been designated by many regulatory agencies as a hazardous substance that can cause serious medical issues. Mercury, or its vapor, has been demonstrated to be hazardous to health and corrosive to materials. Use Caution when handling mercury and mercury-containing products. See the applicable product Safety Data Sheet (SDS) for additional information. The potential exists that selling mercury or mercury-containing products, or both, is prohibited by local or national law. Users must determine legality of sales in their location. 1.7 This standard does not purport to address all of the safety concerns, if any, asso...
ASTM D2892-23 is classified under the following ICS (International Classification for Standards) categories: 75.040 - Crude petroleum. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D2892-23 has the following relationships with other standards: It is inter standard links to ASTM D2892-20, ASTM D6300-24, ASTM D6300-23a, ASTM D6300-23, ASTM D2887-23, ASTM D1298-12b(2017)e1, ASTM D5188-16, ASTM D5191-22, ASTM D5399-09(2023), ASTM D7798-20, ASTM D7213-15(2019), ASTM D6377-20, ASTM D6352-19e1, ASTM D7398-21, ASTM D6897-16. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D2892-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D2892 − 23
Standard Test Method for
Distillation of Crude Petroleum (15-Theoretical Plate
Column)
This standard is issued under the fixed designation D2892; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* 1.4.4 Annex A4—Test Method for the Verification of Tem-
perature Sensor Location,
1.1 This test method covers the procedure for the distillation
1.4.5 Annex A5—Test Method for Determination of the
of stabilized crude petroleum (see Note 1) to a final cut
Temperature Response Time,
temperature of 400 °C Atmospheric Equivalent Temperature
1.4.6 Annex A6—Practice for the Calibration of Sensors,
(AET). This test method employs a fractionating column
1.4.7 Annex A7—Test Method for the Verification of Reflux
having an efficiency of 14 to 18 theoretical plates operated at a
Dividing Valves,
reflux ratio of 5:1. Performance criteria for the necessary
1.4.8 Annex A8—Practice for Conversion of Observed Va-
equipment is specified. Some typical examples of acceptable
por Temperature to Atmospheric Equivalent Temperature
apparatus are presented in schematic form. This test method
(AET),
offers a compromise between efficiency and time in order to
1.4.9 Appendix X1—Test Method for Dehydration of a
facilitate the comparison of distillation data between laborato-
Sample of Wet Crude Oil, and
ries.
1.4.10 Appendix X2—Practice for Performance Check.
NOTE 1—Defined as having a Reid vapor pressure less than 82.7 kPa
(12 psi).
1.5 The values stated in SI units are to be regarded as
standard. The values given in parentheses after SI units are
1.2 This test method details procedures for the production of
provided for information only and are not considered standard.
a liquefied gas, distillate fractions, and residuum of standard-
ized quality on which analytical data can be obtained, and the
1.6 WARNING—Mercury has been designated by many
determination of yields of the above fractions by both mass and
regulatory agencies as a hazardous substance that can cause
volume. From the preceding information, a graph of tempera-
serious medical issues. Mercury, or its vapor, has been dem-
ture versus mass % distilled can be produced. This distillation
onstrated to be hazardous to health and corrosive to materials.
curve corresponds to a laboratory technique, which is defined
Use Caution when handling mercury and mercury-containing
at 15/5 (15 theoretical plate column, 5:1 reflux ratio) or TBP
products. See the applicable product Safety Data Sheet (SDS)
(true boiling point).
for additional information. The potential exists that selling
mercury or mercury-containing products, or both, is prohibited
1.3 This test method can also be applied to any petroleum
by local or national law. Users must determine legality of sales
mixture except liquefied petroleum gases, very light naphthas,
in their location.
and fractions having initial boiling points above 400 °C.
1.7 This standard does not purport to address all of the
1.4 This test method contains the following annexes and
safety concerns, if any, associated with its use. It is the
appendixes:
responsibility of the user of this standard to establish appro-
1.4.1 Annex A1—Test Method for the Determination of the
priate safety, health, and environmental practices and deter-
Efficiency of a Distillation Column,
mine the applicability of regulatory limitations prior to use.
1.4.2 Annex A2—Test Method for the Determination of the
For specific warning statements, see Section 10.
Dynamic Holdup of a Distillation Column,
1.8 This international standard was developed in accor-
1.4.3 Annex A3—Test Method for the Determination of the
dance with internationally recognized principles on standard-
Heat Loss in a Distillation Column (Static Conditions),
ization established in the Decision on Principles for the
Development of International Standards, Guides and Recom-
mendations issued by the World Trade Organization Technical
This test method is under the jurisdiction of ASTM Committee D02 on Barriers to Trade (TBT) Committee.
Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of
Subcommittee D02.08 on Volatility.
Current edition approved Nov. 1, 2023. Published November 2023. Originally
approved in 1970. Last previous edition approved in 2020 as D2892 – 20. DOI:
10.1520/D2892-23.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D2892 − 23
2. Referenced Documents 3.1.2 atmospheric equivalent temperature (AET), n—the
2 temperature converted from the measured vapor temperature
2.1 ASTM Standards:
using equations in Annex A8.
D941 Test Method for Density and Relative Density (Spe-
3.1.2.1 Discussion—The AET is the expected vapor tem-
cific Gravity) of Liquids by Lipkin Bicapillary Pycnom-
perature if the distillation was performed at atmospheric
eter
pressure and there was no thermal decomposition.
D1217 Test Method for Density and Relative Density (Spe-
cific Gravity) of Liquids by Bingham Pycnometer
3.1.3 boilup rate, n—in distillation, the quantity of vapor
D1298 Test Method for Density, Relative Density, or API
entering the column per unit of time.
Gravity of Crude Petroleum and Liquid Petroleum Prod-
3.1.4 debutanization of crude petroleum, n—the removal of
ucts by Hydrometer Method
the light hydrocarbons up to and including n-butane, and
D2887 Test Method for Boiling Range Distribution of Pe-
retention of the heavier hydrocarbons.
troleum Fractions by Gas Chromatography
3.1.4.1 Discussion—In practice, a crude petroleum is re-
D3710 Test Method for Boiling Range Distribution of Gaso-
garded as debutanized if the light hydrocarbon cut collected in
line and Gasoline Fractions by Gas Chromatography
the cold trap contains more than 95 % of the C to C
2 4
(Withdrawn 2014)
hydrocarbons and less than 5 % of the C hydrocarbons
D4006 Test Method for Water in Crude Oil by Distillation
initially present in the sample.
D4052 Test Method for Density, Relative Density, and API
3.1.5 distillation pressure, n—the pressure measured as
Gravity of Liquids by Digital Density Meter
close as possible to the point where the vapor temperature is
D4057 Practice for Manual Sampling of Petroleum and
taken, normally at the top of the condenser.
Petroleum Products
D4177 Practice for Automatic Sampling of Petroleum and
3.1.6 distillation temperature, n—the temperature of the
Petroleum Products
saturated vapor measured in the head just above the fraction-
D5134 Test Method for Detailed Analysis of Petroleum
ating column.
Naphthas through n-Nonane by Capillary Gas Chroma-
3.1.6.1 Discussion—It is also known as the head tempera-
tography
ture or the vapor temperature.
D6300 Practice for Determination of Precision and Bias
3.1.7 dynamic hold-up, n—in column distillation, the quan-
Data for Use in Test Methods for Petroleum Products,
tity of liquid held up in the column under normal operating
Liquid Fuels, and Lubricants
conditions.
D6729 Test Method for Determination of Individual Com-
ponents in Spark Ignition Engine Fuels by 100 Metre
3.1.8 flood point, n—in distillation, the point at which the
Capillary High Resolution Gas Chromatography
velocity of the upflowing vapors obstructs the down-coming
D6730 Test Method for Determination of Individual Com-
reflux and the column suddenly fills with liquid.
ponents in Spark Ignition Engine Fuels by 100-Metre
3.1.9 internal reflux, n—in distillation, the liquid normally
Capillary (with Precolumn) High-Resolution Gas Chro-
running down inside the column.
matography
3.1.10 pressure drop, n—the difference between the pres-
D6733 Test Method for Determination of Individual Com-
sure measured in the condenser and the pressure measured in
ponents in Spark Ignition Engine Fuels by 50-Metre
the distillation flask.
Capillary High Resolution Gas Chromatography
3.1.10.1 Discussion—It is expressed in kilopascals (mm Hg)
3. Terminology
per metre of packed height for packed columns, or kilopascals
(mm Hg) overall for real plate columns. It is higher for
3.1 Definitions:
aromatics than for paraffins, and for higher molecular weights
3.1.1 adiabaticity, n—the condition in which there is no
than for lighter molecules, at a given boilup rate.
significant gain or loss of heat throughout the length of the
column.
3.1.11 reflux ratio, R, n—in distillation, the ratio of the
3.1.1.1 Discussion—When distilling a mixture of com-
condensate at the head of the column that is returned to the
pounds as is the case of crude petroleum, there will be a normal
column (reflux) to that withdrawn as product.
increase in reflux ratio down the column. In the case where
3.1.12 static hold-up or wettage, n—the quantity of liquid
heat losses occur in the column, the internal reflux is abnor-
retained in the column after draining at the end of a distillation.
mally greater than the reflux in the head. The opposite is true
3.1.12.1 Discussion—It is characteristic of the packing or
when the column gains heat, as with an overheated mantle.
the design of the plates, and depends on the composition of the
material in the column at the final cut point and on the final
temperature.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
3.1.13 takeoff rate, n— in distillation, the volume of product
Standards volume information, refer to the standard’s Document Summary page on
withdrawn from the reflux divider over a specified period.
the ASTM website.
Withdrawn. The last approved version of this historical standard is referenced
3.1.14 theoretical plate, n—the section of a column required
on www.astm.org.
to achieve thermodynamic equilibrium between a liquid and its
The last approved version of this historical standard is referenced on
www.astm.org. vapor.
D2892 − 23
3.1.14.1 Discussion—The height equivalent to one theoreti- 6.1.1.1 The sidearm is used as a thermowell. It shall
cal plate (HETP) for packed columns is expressed in millime- terminate about 5 mm from the bottom of the flask to ensure its
tres. In the case of real plate columns, the efficiency is immersion at the end of the distillation. When a second sidearm
expressed as the percentage of one theoretical plate that is is present, it can be used for pressure drop detection with a
achieved on one real plate.
nitrogen bleed or for mechanical stirring, or both.
6.1.1.2 If a magnetic stirrer is used with a spherical flask,
4. Summary of Test Method
the flask shall have a slightly flattened or concave area at the
bottom on which the magnetic stirrer can rotate without
4.1 A weighed sample of 1 L to 30 L of stabilized crude
petroleum is distilled to a maximum temperature of 400 °C grinding the glass. In this case, termination of the thermowell
AET in a fractionating column having an efficiency at total shall be off center 40 mm 6 5 mm to avoid the magnetic
reflux of at least 14, but not greater than 18, theoretical plates. stirring bar. Boiling chips can be used as an alternative to a
stirrer.
4.2 A reflux ratio of 5:1 is maintained at all operating
6.1.1.3 (Warning—While the advantage of visibility in
pressures, except that at the lowest operating pressures be-
glass distillation flasks is desirable, flasks of glass may become
tween 0.674 kPa and 0.27 kPa (5 mm and 2 mm Hg), a reflux
hazardous the larger the charge they contain. For this reason,
ratio of 2:1 is optional. In cooperative testing or in cases of
glass flasks of a volume greater than 10 L are not recom-
dispute, the stages of low pressure, the reflux ratios, and the
mended.)
temperatures of cut points must be mutually agreed upon by the
interested parties prior to beginning the distillation.
6.1.2 Heating System—Heating of the flask shall be pro-
vided in such a way that full boilup can be maintained at a
4.3 Observations of temperature, pressure, and other vari-
steady rate at all pressure levels. An electric heating mantle
ables are recorded at intervals and at the end of each cut or
covering the lower half of the flask and having one third of the
fraction.
heat in an element located in the bottom central area and the
4.4 The mass and density of each cut or fraction are
remaining two thirds in the rest of the hemisphere is recom-
obtained. Distillation yields by mass are calculated from the
mended. While proportioning controllers are preferred, heat
mass of all fractions, including liquefied gas cut and the
input can be manually adjusted by use of a variable auto
residue. Distillation yields by volume of all fractions and the
transformer on each circuit, the smaller heater being automati-
residue at 15 °C are calculated from mass and density.
cally controlled by an instrument sensing the pressure drop of
the column as registered in a differential pressure instrument or
4.5 From these data the TBP curves in mass or volume
percent, or both, versus AET are drawn. alternatively by direct measurement of distillation rate.
6.1.2.1 Minimum wattage required to provide full boilup of
5. Significance and Use
crude petroleum is approximately 0.125 W ⁄mL of charge.
Twice this amount is recommended for quick heat-up.
5.1 This test method is one of a number of tests conducted
on a crude oil to determine its value. It provides an estimate of
6.1.2.2 The heat density in the flask heaters is approxi-
2 2
the yields of fractions of various boiling ranges and is therefore
mately equal to 0.5 W ⁄cm to 0.6 W ⁄cm . This requires the use
valuable in technical discussions of a commercial nature.
of nickel-reinforced quartz fabric to ensure a reasonable
service life.
5.2 This test method corresponds to the standard laboratory
6.1.2.3 Immersion heaters can be employed in a similar way
distillation efficiency referred to as 15/5. The fractions pro-
and have the advantage of faster response, but they are more
duced can be analyzed as produced or combined to produce
fragile and require a specially designed flask to ensure that the
samples for analytical studies, engineering, and product quality
heating elements remain immersed at the end of the run. When
evaluations. The preparation and evaluation of such blends is
used, their heat density should be approximately equal to
not part of this test method.
4 W ⁄cm .
5.3 This test method can be used as an analytical tool for
6.1.2.4 The upper half of the flask shall be covered with a
examination of other petroleum mixtures with the exception of
mantle to avoid unnecessary heat losses from the upper surface
LPG, very light naphthas, and mixtures with initial boiling
and shall have an electric heater supplying about 0.25 W ⁄cm
points above 400 °C.
at full-rated voltage.
6. Apparatus 6.1.3 Fractionating Column—The fractionating column
must contain either particulate packing or real plates similar to
6.1 Distillation at Atmospheric Pressure—All components
those whose performance characteristics are summarized in
must conform to the requirements specified as follows. Auto-
Table 1 and meet the specifications stated in 6.1.3.1 through
matic devices can be employed provided they meet the same
6.1.3.4. Table 2 lists current North American suppliers of
requirements. A typical apparatus is illustrated in Fig. 1.
suitable packings.
6.1.1 Distillation Flask—The distillation flask shall be of a
6.1.3.1 The internal diameter shall be between 25 mm and
size that is at least 50 % larger than the volume of the charge.
70 mm.
The size of the charge, between 1.0 L and 30 L, is determined
by the holdup characteristics of the fractionating column, as 6.1.3.2 The efficiency shall be between 14 and 18 theoreti-
shown in Table 1 and described in Annex A2. The distillation cal plates at total reflux when measured by the procedure
flask shall have at least one sidearm. described in Annex A1.
D2892 − 23
FIG. 1 Apparatus
at the bottom of the column.
6.1.3.3 The fractionating column shall be comprised of a
integral glass column and reflux divider totally enclosed in a
6.1.3.4 The column shall be enclosed in a heat-insulating
highly reflective vacuum jacket having a permanent vacuum of
system, such as a glass-fabric mantle, capable of maintaining
−6
less than 0.1 mPa (;10 mm Hg). It shall be essentially
the temperature of the outer wall of the glass vacuum jacket
adiabatic when tested in accordance with Annex A3.
equal to that of the internal vapor temperature. To verify this,
NOTE 2—In the case of an adiabatic column when distilling a pure
the vacuum jacket shall have a temperature sensor, such as a
compound, the internal reflux is constant from top to bottom and is equal 2
thermocouple, soldered to about 6 cm of thin copper or brass
to the reflux at the reflux divider. When distilling crude petroleum, the
sheet and fastened to the outer wall of the glass jacket at a level
fractionation occurring in the dynamic holdup will cause a temperature
gradient to be established with attendant greater amount of internal reflux just below the reflux divider.
D2892 − 23
TABLE 1 Data for n-Heptane-Methylcyclohexane Test Mixture at 75 % of Maximum Boilup and 101.3 kPa (760 mm Hg)
A,B,C,D,E F,G,H E,I,J E,K
Propak Helipak Perforated Plates Wire Mesh
Column diameter, mm 25 50 70 25 50 25 50 25 50
L L L L
Packing size, mm 4 6 6 No. 2917 No. 2918 NA NA NA NA
Boilup, mL/h × cm 650 670 675 300 350 640 660 810 1050
Dynamic holdup
L L
% of packed volume 17 15.3 17.0 15 14.3 NA NA 8.0 10.0
mL/theoretical plate 3.2 16 39 1.6 8.7 2.8 12.3 2.0 12.9
Pressure drop
L L
kPa/m 1.2 1.05 0.94 1.53 1.41 NA NA 0.97 0.75
L L
mm Hg/m 9.0 7.9 7.1 11.5 10.6 NA NA 7.3 5.6
kPa/theoretical plate 0.045 0.056 0.06 0.03 0.045 0.15 0.16 0.05 0.05
mm Hg/theoretical plate 0.34 0.42 0.43 0.24 0.34 1.1 1.2 0.35 0.37
HETP, mm (% of real plates) 38 53 61 21 32 (60 %) (65 %) 48 66
For 15-plate Towers
Packed height, cm (plates) 57 80 91 31.5 48 (25) (23) 72 99
L L
Packed volume, mL 280 1570 3460 155 917 NA NA 353 1940
Dynamic holdup, mL 47 240 590 23 131 42 184 28 194
Pressure drop
kPa 0.68 0.84 0.86 0.48 0.68 2.2 2.4 0.70 0.73
mm Hg 5.1 6.3 6.5 3.6 5.1 16.5 18.0 5.3 5.5
Charge volume, L
Min (4 % Holdup) 1.2 6.0 15 0.575 3.3 1.0 4.6 0.7 4.9
Max (1 % Holdup) 4.8 24.0 60 2.3 13.0 4.2 10.4 2.8 19.4
A
Cooke, G. M. and Jameson, B. G. Analytical Chemistry, Vol 27, 1955, p. 1798.
B
Struck, R. T. and Kinner, C. R. Industrial and Engineering Chemistry, Vol 42, 1950, p. 77.
C
Cannon, M. R. Industrial and Engineering Chemistry, Vol 41, No. 9, 1949, p. 1953.
D
Cannon Instrument Company, 2139 High Tech Rd., State College, PA 16803.
E
Cooke, G. M. Analytical Chemistry, Vol 39, 1967, p. 286.
F
Bulletin of Podbielniak Div. of Reliance Glass Works, P.O. Box 825, Bensenville, IL 60106.
G
Feldman, J., et al, Industrial and Engineering Chemistry, Vol 45, January 1953, p. 214.
H
Helipak Performance Characteristics, Begemean, C. R. and Turkal, P. J. (Laboratory Report of Podbielniak Inc.), 1950.
I
Umholtz, C. L. and Van Winkle, M. Petroleum Refiner, Vol 34, 1955, p. 114 for NH:MCH. Pressure Drop Calculated from data obtained on o- and m-xylene binary.
J
Oldershaw, C. F. Industrial and Engineering Chemistry, Vol 13, 1941, p. 265.
K
Bragg, L. B. Industrial and Engineering Chemistry, Vol 49, 1957, p. 1062.
L
NA = not applicable.
TABLE 2 North American Sources of Commercially Available
takeoff line over an approximate range of rates from 10 % to
Packing Materials
90 % of the maximum boil up rate of the column when
Name Size Source
determined in accordance with Annex A7.
Propak 6 mm by 6 mm Cannon Instrument Company
6.1.4 Condenser—The condenser shall have sufficient ca-
2139 High Tech Rd.
State College, PA 16803
pacity to condense essentially all the C and C vapors from the
4 5
Helipak 2.5 mm by 4 mm Reliance Glass Works Inc.
crude at the specified rate, using a coolant temperature
P.O. Box 825
Bensenville, IL 60106 of −20 °C.
Perforated plates 25 mm and 50 mm Reliance Glass Works Inc.
6.1.5 Cold Traps—Two efficient traps of adequate capacity
P.O. Box 825
Bensenville, IL 60106 cooled by dry ice and alcohol mixture shall be connected in
W.A. Sales Inc.
series to the vent line of the condenser when light hydrocar-
419 Harvester Ct.
bons are present, as at the beginning of the distillation. For
Wheeling, IL 60090
Knitted wire mesh- Pegasus Industrial Specialties Ltd.
vacuum distillation, a Dewar-style trap also cooled by dry ice
Goodloe multiknit P.O. Box 319
is used to protect the vacuum gauge from vapors.
Agincourt, Ontario MIS 3B9 Canada
Packed Column Co.
6.1.6 Gas Collector—If uncondensed gas is to be measured,
970 New Durham Rd.
a gas meter can be connected to the outlet of the cold trap but
Edison, NJ 08817
with a calcium chloride drying tube between them to keep
moisture from collecting in the traps. When analysis of the gas
NOTE 3—For certain types of columns there is no significant difference
sample is required, the gas can be collected in an empty plastic
in yields and fraction qualities between an uncompensated and a heat-
balloon of suitable size either in place of the meter or following
compensated column. In such a case, by mutual agreement between
it. The volume of its contents can be determined by calculation
parties concerned, the application of a heated insulating system can be
from the rise in pressure after expanding the sample into an
omitted.
evacuated vessel of known volume.
6.1.3.5 The adjustable reflux divider shall be located about
6.1.7 Fraction Collector—This part of the apparatus permits
one column diameter above the top of the packing or topmost
plate. It must be capable of dividing the condensate with an the collection of the distillate without interruption during
accuracy of better than 90 % between the column and the withdrawal of product from the receiver under atmospheric or
D2892 − 23
reduced pressure. It also permits removal of product from the 6.3.1.1 The vapor temperature sensor can be a platinum
vacuum system without disturbing conditions in the column. resistance thermometer, a Type J thermocouple with the
6.1.8 Product Receivers—The receivers shall be of suitable junction head fused to the lower tip of the thermowell, or any
size for the quantity of crude petroleum being distilled. The other device that meets the requirements in this paragraph and
recommended capacity is from 100 mL to 500 mL. They shall
6.3.1.2. The tip of the sensor shall be located above the top of
be calibrated and graduated to permit reading to the nearest the packing or the topmost glass plate and in close proximity to
1 %.
the reflux divider but not in contact with the liquid reflux. The
location of the vapor temperature sensor shall be proved by the
6.2 Distillation Under Reduced Pressure—In addition to the
test method described in Annex A4. The sensor shall have a
apparatus listed in 6.1, the apparatus for distillation under
cooling time of not more than 175 s, as described in Annex A5.
reduced pressure shall include the following:
6.3.1.2 The vapor temperature measuring device shall have
6.2.1 Vacuum Pump—The vacuum system shall be capable
an accuracy of 0.5 °C or better and be measured with a
of maintaining smooth pressure operation at all pressure levels.
resolution of 0.1 °C or better. The liquid temperature measur-
It shall have the capacity to draw down the pressure in the
ing device shall have an accuracy of 1.0 °C or better and be
receiver(s) from atmospheric to 0.25 kPa (2 mm Hg) in less
measured with a resolution of 0.5 °C or better. Temperatures
than 30 s so as to avoid disturbance of the system during
emptying of receivers under vacuum. Alternatively, a separate are recorded either manually or automatically.
pump can be employed for this purpose.
6.3.1.3 Temperature sensors shall be calibrated as described
6.2.2 Vacuum Gauge—The point of connection of the
in Annex A6. Alternatively certified sensors may be used,
vacuum gauge to the system shall be as close as practical to the
provided the calibration of the sensor and its associated
reflux dividing head. The connecting tubing shall be of
recording instrument can be traced back to a primary tempera-
sufficient diameter to ensure that no measurable pressure drop
ture standard. Temperature sensors are calibrated over the full
occurs in the line. In no case shall the vacuum gauge
range of temperature (from 0 °C to 327.4 °C) at the time of first
connection be near the vacuum pump.
use of the sensor in combination with its associated instrument.
6.2.2.1 All gauges shall be carefully protected from con-
Recalibrate when either the sensor or the instrument is repaired
densable vapors, especially water vapor, by a cold trap main-
or serviced. Verification of the calibration of the temperature
tained at the temperature of dry ice.
sensors is to be made on a regular basis. For vapor temperature
6.2.3 Pressure Regulator—The regulator shall maintain the
sensors, verification at least once a month is recommended and
pressure in the system essentially constant at all operating
for liquid temperature sensors once every six months. Verifi-
pressures. Automatic regulation can be achieved by a device
cation of the calibration of the sensors can be accomplished
that regulates the demand on the vacuum source. A satisfactory
potentiometrically by the use of standard precision resistance
device is a solenoid valve positioned between the vacuum
or by distilling a pure compound with accurately known
source and a surge tank of at least 10 L capacity. Alternatively,
boiling point.
a manual bleed valve can be maintained by a trained operator
6.3.2 Vacuum Gauge—Primary standards, such as the non-
with a minimum of attention.
tilting McLeod gauge, mercury manometer, or other analogous
6.3 Sensing and Recording Apparatus:
primary standard pressure devices can be used without calibra-
6.3.1 Temperature Sensors—Only temperature measure- tion when properly used and maintained. A mercury
ment systems meeting the requirements of 6.3.1.1 and 6.3.1.2 manometer, however, will only be of satisfactory accuracy
shall be used. down to a pressure of about 1 kPa and then only when read
FIG. 2 Approximate Pressure Drop-Fractionators Using Propak
D2892 − 23
with a good cathetometer (an instrument based on a telescope 7.4 The correctness of the AET is mainly, but not only,
mounted on a vernier scale to determine levels very accu- dependent on the accuracy of the (vapor) temperature and
rately). Alternatively, a tensimeter or certified electronic sen-
(operating) pressure sensors (Annex A6). Other factors affect-
sors may be used, provided the calibration of the sensor and its ing the accuracy and precision of the boiling point curve are:
associated recording instrument can be traced back to a
7.4.1 The location of the temperature and pressure sensor
primary pressure standard. Sensors of the diaphragm type have
(Annex A4).
been found satisfactory. Vacuum gauges based on hot wires,
7.4.2 The dynamic response of the sensors (Annex A5).
radiation, or electrical conductivity detectors are not recom-
7.4.3 The correct operation of the reflux divider (Annex
mended.
A7).
6.3.2.1 The gauge for measuring subatmospheric pressures
7.4.4 The heat loss from the column (Annex A3).
shall have an accuracy at least equal to that stated as follows:
7.4.5 The efficiency of the column (Annex A1).
Distillation Pressure Accuracy
7.4.6 These factors are basically covered through the appro-
kpa mm Hg kPa mm Hg
priate annexes. However, it should be realized that this takes
100-13.3 760 to 100 0.13 1.0
care only of individual components and does not cover the
13.3-1.33 99 to 10 0.013 0.1
1.33-0.266 9 to 2 0.006 0.06
combined effect of small deviations. Moreover, the aforemen-
6.3.2.2 Noncertified gauges shall be calibrated from a pri- tioned tests are all done under more or less static conditions,
mary standard or a secondary electronic standard traceable to a
not necessarily representative for the behavior of the system
primary standard. A basic calibration procedure is described in
under actual dynamic conditions.
Annex A6. Recalibrate when either the sensor or the instrument
7.5 Cut quality is mainly defined by the efficiency of the
is repaired or serviced. Verification of the calibration of the
column (Annex A1), but is also affected by:
electronic pressure sensors is to be made on a regular basis. A
7.5.1 The correct operation of the reflux divider (reflux
frequency of at least once a month is recommended. Verifica-
ratio) (Annex A7).
tion of the calibration of the sensors can be accomplished using
the procedures described in Annex A6 or against a certified 7.5.2 The heat loss from the column, that is, internal reflux
reference system.
(Annex A3).
6.3.3 Boilup Rate—The boilup rate is normally controlled
7.5.3 The dynamic hold up of the column (Annex A2).
by sensing the pressure drop in the column. The pressure drop
7.5.4 Column efficiency is covered in this test method
during operation is measured by means of a manometer or
through Table 1 and Annex A1. However, Table 1 only
pressure transducer connected between the flask and the
provides an assumption on efficiency and is not a guarantee.
condenser. Prevention of condensation in the connecting tube
Annex A1 only provides a check under static conditions,
can be accomplished by injecting a very small flow of nitrogen
infinite reflux ratio, rather low actual temperatures and a binary
(8 cm /s) between the pressure drop sensor manometer and the
component system. Hence, although there is some safeguard on
flask (see Fig. 1) or by placing a small water-cooled condenser
standard performance, through conformance to Annex A1,
between the flask and the pressure drop sensor. Alternatively,
Annex A2, Annex A3, and Annex A7, again it remains
the boilup rate can be controlled from the measurement of take
questionable whether this is truly representative for columns
off rate.
under actual operating conditions.
7. Verification of Apparatus Performance
7.6 Theoretically, an overall performance check, like the
7.1 Test Method D2892 provides a standard framework for
one described in Appendix X2, is capable of verifying the
the laboratory distillation of crude oils in order to produce cuts
performance of a column and the correctness of the AET under
of defined quality (for further testing) and the concurrent
actual operating conditions. Appendix X2, in principle, mea-
production of a boiling point curve. As the quantity require-
sures the combined effect of all factors affecting the results of
ments and cut points might be widely different between
Test Method D2892.
companies and application areas, this test method does not
7.6.1 The minimum tray number as defined in Appendix X2
standardize on equipment design but on equipment perfor-
is a measure of overall cut quality, and the difference between
mance.
nominal cut point (AET) and effective cut point (ECP as
7.2 The nature of the test method (the use of large sample
defined in Appendix X2) provides a measure for the correct-
quantities and very time consuming) and the nature of the
ness of the AET. However, insufficient data are available right
product being tested (highly volatile and unstable material),
now to define the allowable tolerances in a rigid statistical way.
precludes the use of standard statistical control techniques.
Moreover, the test method described is very labor intensive and
Moreover, this test method does not produce a single result, nor
precludes its use on a regular, short time interval basis and,
is the series of results (the boiling point curve) derived under
therefore, its use as a mandatory statistical control technique.
rigidly defined conditions (see 4.2).
7.6.2 Appendix X2, therefore, provides only recommended
guidelines for statistical control on column performance, in-
7.3 Equipment performance in the context of Test Method
D2892 consists of two elements; the efficiency of the column, cluding both correctness of AET and column efficiency. It is the
responsibility of the laboratory to provide for sufficient quality
defining cut quality, and the correctness of the cut point (AET),
defining the boiling point curve. controls to guarantee conformance to the test method.
D2892 − 23
8. Sampling 10.2.2 Begin circulation of refrigerant at a temperature no
higher than −20 °C in the condenser, distillate cooler, and
8.1 Obtain a sample for distillation in accordance with
receiver, if so equipped.
instructions given in Practice D4057 or D4177. The sample
10.2.3 Record the barometric pressure at the beginning and
must be received in a sealed container and show no evidence of
periodically throughout the distillation.
leakage.
10.2.4 Apply heat to the flask at such a rate that vapors
8.2 Cool the sample to between 0 °C and 5 °C by placing it
reach the top of the column between 20 min and 50 min after
in a refrigerator for several hours (preferably overnight) before
startup. Adjust heat input so as to achieve a pressure drop of
opening.
less than 0.13 kPa ⁄m (1.0 mm Hg/m) in packed columns or less
than 0.065 kPa (0.5 mm Hg) in real plate columns. Program
8.3 If the sample appears waxy or too viscous, raise the
automated equipment in accordance with the preceding direc-
temperature to 5 °C above its pour point.
tions. Turn on the stirring device if used.
8.4 Agitate the sample by whatever means are appropriate
10.2.5 Allow the column to operate at total reflux until the
to its size to ensure that it is well mixed.
vapor temperature reaches equilibrium but not longer than
15 min after the first drop of condensate appears in the reflux
8.5 Determine the water content of the sample by Test
divider.
Method D4006 or any other suitable method. If the water
content exceeds 0.3 % volume, the sample shall be dehydrated 10.2.6 Record the vapor temperature as the initial vapor
temperature.
prior to fractional distillation. A suitable practice for dehydra-
tion of wet crude oil samples is described in Appendix X1. 10.2.7 Stop the circulation of the refrigerant and observe the
vapor temperature. When the vapor temperature reaches 15 °C,
NOTE 4—Attempts to distill wet crude oil samples in glass columns
start the circulation of refrigerant again.
might result in breakage of the glassware, which poses a potential fire
10.2.8 If the vapor temperature drops below 15 °C, continue
hazard. Moreover, the presence of water will affect the accuracy of
distillation yield in the naphtha region. These effects are more pronounced
refluxing for at least 15 min. Repeat 10.2.7. If the vapor
for heavy crude oils, containing low amounts of hydrocarbons boiling
temperature remains at 15 °C or rises, continue with the
below 100 °C, than for light crudes where there is usually sufficient
atmospheric distillation. (Warning—The following three steps
hydrocarbon vapor generated to form an azeotrope and drive the water
should not be done until after the first naphtha cut has been
vapors through the column without problems.
removed to ensure that all the light gases have been recovered.)
10.2.9 Remove and weigh the dry ice traps containing light
9. Preparation of Apparatus
hydrocarbon liquid after carefully wiping them dry.
9.1 Clean and dry the distillation column and all the
10.2.10 Sample the contents of the first dry ice trap using a
ancillary glass apparatus before the distillation begins.
10 mL to 50 mL pressure vessel evacuated to no lower than
9.2 Ensure that the system is leak-free and all heaters, 26.6 kPa (200 mm Hg). Keep all containers at the temperature
control devices, and instruments are on and in working order. of dry ice to ensure no loss of volatiles. The first trap next to
A clock or other timing device should be ready for use. the condenser should contain all of the sample. If condensate is
found in the second trap, sample both traps or combine the
10. Procedure contents before sampling.
10.2.11 Submit the trap sample and gas balloon, if used, for
10.1 Charging:
analysis by a suitable gas chromatographic test method to be
10.1.1 The charge size shall be such that the dynamic hold
reported on a fixed-gas free basis. Test Methods D6729,
up as determined in accordance with Annex A2 is between 1 %
D6730, and D6733, equipped with liquid or gas sampling
and 4 % of the charge when operating at 75 % of maximum
valves, or both, for sample introduction equipment have been
boilup (see Table 1). Chill the flask to a temperature not lower
used successfully for this analysis.
then 0 °C.
10.3 Distillation at Atmospheric Pressure:
10.1.2 Insert the stirring device or place some pieces of
10.3.1 Maintain a temperature below −20 °C in the lines of
glass or porcelain into the flask to control bumping.
the distillate cooler and receiver as well as in the condenser.
10.1.3 Determine the density of the sample by Test Method
Turn on the column mantle heat controller and maintain the
D941, D1217, or D1298.
column jacket temperature 0 °C to 5 °C below the vapor
10.1.4 Calculate to within 65 % the mass of crude petro-
temperature.
leum corresponding to the desired volume of the charge. Weigh
10.3.2 Regulate the heat input as necessary to establish and
to the nearest 1 % this quantity of sample into the flask.
maintain a boilup rate approximately 75 % of maximum. Fig.
10.1.5 Attach the flask to the column and connect the
3 can be used as a guide for Propak. Rates for other sizes can
pressure drop measuring device. Install the heating system,
be estimated by multiplying the boilup rate in Table 1 by the
stirrer, and support device. (Warning—Poisonous H S gas is
cross-sectional area of the column and dividing by the sum of
frequently evolved from crude oil and precautions must be
the reflux ratio + 1.
taken either to absorb the gas that passes through the cold trap
or to vent it to a safe place.)
NOTE 5—Boilup rate is expressed in millilitres of liquid per hour for a
given column or in millilitres per hour per square centimetre of cross-
10.2 Debutanization:
sectional area for comparative purposes. In the latter case, it refers to the
10.2.1 For necessary apparatus refer to 6.1.5 and 6.1.6. test mixture of n-heptane and methylcyclohexane in the efficiency
D2892 − 23
reflux, not to exceed 15 min, to restore operating conditions
before continuing takeoff. Do not make a cut within 5 °C of
startup.
10.3.7 Continue taking cuts until the desired maximum
vapor temperature is reached or until the charge shows signs of
cracking. Pronounced cracking is evidenced by a fog appearing
in the flask and later at the reflux divider. Do not allow the
vapor temperature to exceed 210 °C nor the temperature of the
boiling liquid to exceed 310 °C.
10.3.8 Shut off the reflux valve and the heating system.
Allow the contents to cool to such a temperature that the
distillation can be commenced at 13.3 kPa (100 mm Hg)
without flooding. This temperature can be estimated by adding
the ΔT between the liquid and vapor temperatures found for the
column during atmospheric operation to the expected initial
vapor temperature at the reduced pressure, or by subtracting
the ΔT from the last recorded liquid temperature.
NOTE 7—Cooling of the liquid in the flask can be accelerated by
blowing a gentle stream of compressed air onto the flask after its heating
mantle has been removed. Avoid strong jets of cold air. Alternately, turn
on coolant in the quench coil of the flask, if used.
FIG. 3 Expected Takeoff Rates at 5:1 Reflux Ratio for Fraction-
10.3.9 Weigh all fractions and determine their densities.
ators Using Propak
10.3.10 Submit the first distillate fraction for analysis by gas
chromatography.
10.4 Distillation at 13.3 kPa (100 mm Hg):
10.4.1 If further cuts at higher temperatures are required,
evaluation (see Annex A1) and is measured at the bottom of the column.
distillation can be continued at reduced pressures, subject to
The maximum boilup of the n-heptane-methylcyclohexane test mixture is
that which the column can handle under stable conditions without the maximum temperature that the boiling liquid will stand
flooding. In routine adiabatic operation, the boilup rate can be estimated
without significant cracking. This is about 310 °C in most
roughly from the takeoff rate multiplied by the reflux ratio plus one.
cases. Notable exceptions are crude oils containing heat-
10.3.3 Commence takeoff at a reflux ratio of 5:1 and total
sensitive sulfur compounds. In any case, do not make a cut
cycle time of not over 36 s nor less than 24 s.
within 5 °C of the temperature at startup because the column
will not be at equilibrium.
NOTE 6—The vapor reaching the top of the column is totally condensed
10.4.2 Connect a vacuum pumping and control system to
and the resulting liquid is divided into two parts. One part L (reflux), is
returned to the column and the other part, D (distillate), is withdrawn as the apparatus as shown in Fig. 1.
product. The reflux ratio (R = L ⁄D) can vary from zero at total takeoff
10.4.3 Start the vacuum pump and adjust the pressure
(L = 0) to infinity at total reflux (D = 0).
downward gradually to the value of 13.3 kPa (100 mm Hg) or
10.3.4 Take off distillate in separate and consecutive frac-
set the pressure regulator at this value. The temperature of the
tions of suitable size. The recommended size of fraction is that
liquid in the flask must be below that at which it will boil at
corresponding to 5 °C or 10 °C in vapor temperature. Collect
13.3 kPa (100 mm Hg). If the liquid boils before this pressure
fractions boiling below 65 °C in receivers cooled to 0 °C or is reached, increase the pressure and cool further until the
below. When the vapor temperature reaches 65 °C, refrigerant
desired pressure can be achieved without boiling.
in the condenser and related coolers can be discontinued and
10.4.4 Apply heat to the boiler and reestablish reflux at any
water at ambient temperature substituted.
moderate rate in the reflux divider for about 15 min to reheat
10.3.5 At the end of each fraction and at each cut point,
the column to operating temperature. Momentarily stop heat
record the following observations:
input and raise the pressure with N for 1 min to drop the
10.3.5.1 Time in hours and minutes,
holdup into the distillation flask.
10.3.5.2 Volume in millilitres, 10.4.5 Reapply heat to the distillation flask and adjust the
10.3.5.3 Vapor temperature in °C to the nearest 0.5 °C, rate of heating to maintain a constant pressure drop equivalent
10.3.5.4 Temperature of the boiling liquid in °C to the to the boilup rate of approximately 75 % of the maximum rate
nearest 1 °C, for this pressure and begin takeoff without delay. The approxi-
mate pressure drops required for this purpose are indicated in
10.3.5.5 Atmospheric pressure in kPa (mm Hg), and
10.3.5.6 Pressure drop in the column in kPa (mm Hg). Fig. 3. Maintain a column insulation temperature 0 °C to 5 °C
below the vapor temperature throughout the operation.
10.3.6 If signs of flooding are observed, reduce the heating
10.4.6 Remove separately, cuts of suitable size as in 10.3.4.
rate while continuing takeoff until steady conditions are
restored. If a cut point is encountered during this period, stop 10.4.7 At the end of each distillate fraction and at each cut
point, record the following observations:
the distillation, cool the charge, and recombine the off-
condition cuts. Restart the distillation with a period at total 10.4.7.1 Time in hours and minutes,
D2892 − 23
10.4.7.2 Volume in millilitres observed at ambient 10.5.7 When the temperature of the residue in the flask has
temperature, fallen below 230 °C, shut off the vacuum pump. Vent the
fractionating unit with nitrogen or other inert gas. Do not use
10.4.7.3 Vapor temperature in °C to the nearest 0.5 °C with
correction, if any, air. (Warning—Air is suspected of initiating explosions in
fractionating units that are vented while too hot, such as at the
10.4.7.4 Temperature of the boiling liquid in °C to the
nearest 1 °C, end of a run.)
10.4.7.5 Pressure drop in the column in kPa (mm Hg),
10.5.8 Stop circulation of coolant in the co
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D2892 − 20 D2892 − 23
Standard Test Method for
Distillation of Crude Petroleum (15-Theoretical Plate
Column)
This standard is issued under the fixed designation D2892; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This test method covers the procedure for the distillation of stabilized crude petroleum (see Note 1) to a final cut temperature
of 400 °C Atmospheric Equivalent Temperature (AET). This test method employs a fractionating column having an efficiency of
14 to 18 theoretical plates operated at a reflux ratio of 5:1. Performance criteria for the necessary equipment is specified. Some
typical examples of acceptable apparatus are presented in schematic form. This test method offers a compromise between efficiency
and time in order to facilitate the comparison of distillation data between laboratories.
NOTE 1—Defined as having a Reid vapor pressure less than 82.7 kPa (12 psi).
1.2 This test method details procedures for the production of a liquefied gas, distillate fractions, and residuum of standardized
quality on which analytical data can be obtained, and the determination of yields of the above fractions by both mass and volume.
From the preceding information, a graph of temperature versus mass % distilled can be produced. This distillation curve
corresponds to a laboratory technique, which is defined at 15/5 (15 theoretical plate column, 5:1 reflux ratio) or TBP (true boiling
point).
1.3 This test method can also be applied to any petroleum mixture except liquefied petroleum gases, very light naphthas, and
fractions having initial boiling points above 400 °C.
1.4 This test method contains the following annexes and appendixes:
1.4.1 Annex A1—Test Method for the Determination of the Efficiency of a Distillation Column,
1.4.2 Annex A2—Test Method for the Determination of the Dynamic Holdup of a Distillation Column,
1.4.3 Annex A3—Test Method for the Determination of the Heat Loss in a Distillation Column (Static Conditions),
1.4.4 Annex A4—Test Method for the Verification of Temperature Sensor Location,
1.4.5 Annex A5—Test Method for Determination of the Temperature Response Time,
1.4.6 Annex A6—Practice for the Calibration of Sensors,
This test method is under the jurisdiction of ASTM Committee D02 on Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of Subcommittee
D02.08 on Volatility.
Current edition approved June 1, 2020Nov. 1, 2023. Published June 2020November 2023. Originally approved in 1970. Last previous edition approved in 20192020 as
D2892 – 19.D2892 – 20. DOI: 10.1520/D2892-20.10.1520/D2892-23.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D2892 − 23
1.4.7 Annex A7—Test Method for the Verification of Reflux Dividing Valves,
1.4.8 Annex A8—Practice for Conversion of Observed Vapor Temperature to Atmospheric Equivalent Temperature (AET),
1.4.9 Appendix X1—Test Method for Dehydration of a Sample of Wet Crude Oil, and
1.4.10 Appendix X2—Practice for Performance Check.
1.5 The values stated in SI units are to be regarded as standard. The values given in parentheses after SI units are provided for
information only and are not considered standard.
1.6 WARNING—Mercury has been designated by many regulatory agencies as a hazardous substance that can cause serious
medical issues. Mercury, or its vapor, has been demonstrated to be hazardous to health and corrosive to materials. Use Caution
when handling mercury and mercury-containing products. See the applicable product Safety Data Sheet (SDS) for additional
information. The potential exists that selling mercury or mercury-containing products, or both, is prohibited by local or national
law. Users must determine legality of sales in their location.
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use. For specific warning statements, see Section 10.
1.8 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
D2892 − 23
2. Referenced Documents
2.1 ASTM Standards:
D941 Test Method for Density and Relative Density (Specific Gravity) of Liquids by Lipkin Bicapillary Pycnometer
D1217 Test Method for Density and Relative Density (Specific Gravity) of Liquids by Bingham Pycnometer
D1298 Test Method for Density, Relative Density, or API Gravity of Crude Petroleum and Liquid Petroleum Products by
Hydrometer Method
D2887 Test Method for Boiling Range Distribution of Petroleum Fractions by Gas Chromatography
D3710 Test Method for Boiling Range Distribution of Gasoline and Gasoline Fractions by Gas Chromatography (Withdrawn
2014)
D4006 Test Method for Water in Crude Oil by Distillation
D4052 Test Method for Density, Relative Density, and API Gravity of Liquids by Digital Density Meter
D4057 Practice for Manual Sampling of Petroleum and Petroleum Products
D4177 Practice for Automatic Sampling of Petroleum and Petroleum Products
D5134 Test Method for Detailed Analysis of Petroleum Naphthas through n-Nonane by Capillary Gas Chromatography
D6300 Practice for Determination of Precision and Bias Data for Use in Test Methods for Petroleum Products, Liquid Fuels, and
Lubricants
D6729 Test Method for Determination of Individual Components in Spark Ignition Engine Fuels by 100 Metre Capillary High
Resolution Gas Chromatography
D6730 Test Method for Determination of Individual Components in Spark Ignition Engine Fuels by 100-Metre Capillary (with
Precolumn) High-Resolution Gas Chromatography
D6733 Test Method for Determination of Individual Components in Spark Ignition Engine Fuels by 50-Metre Capillary High
Resolution Gas Chromatography
3. Terminology
3.1 Definitions:
3.1.1 adiabaticity, n—the condition in which there is no significant gain or loss of heat throughout the length of the column.
3.1.1.1 Discussion—
When distilling a mixture of compounds as is the case of crude petroleum, there will be a normal increase in reflux ratio down
the column. In the case where heat losses occur in the column, the internal reflux is abnormally greater than the reflux in the head.
The opposite is true when the column gains heat, as with an overheated mantle.
3.1.2 atmospheric equivalent temperature (AET), n—the temperature converted from the measured vapor temperature using
equations in Annex A8.
3.1.2.1 Discussion—
The AET is the expected vapor temperature if the distillation was performed at atmospheric pressure and there was no thermal
decomposition.
3.1.3 boilup rate, n—in distillation, the quantity of vapor entering the column per unit of time.
3.1.4 debutanization of crude petroleum, n—the removal of the light hydrocarbons up to and including n-butane, and retention of
the heavier hydrocarbons.
3.1.4.1 Discussion—
In practice, a crude petroleum is regarded as debutanized if the light hydrocarbon cut collected in the cold trap contains more than
95 % of the C to C hydrocarbons and less than 5 % of the C hydrocarbons initially present in the sample.
2 4 5
3.1.5 distillation pressure, n—the pressure measured as close as possible to the point where the vapor temperature is taken,
normally at the top of the condenser.
3.1.6 distillation temperature, n—the temperature of the saturated vapor measured in the head just above the fractionating column.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Withdrawn. The last approved version of this historical standard is referenced on www.astm.org.
The last approved version of this historical standard is referenced on www.astm.org.
D2892 − 23
3.1.6.1 Discussion—
It is also known as the head temperature or the vapor temperature.
3.1.7 dynamic hold-up, n—in column distillation, the quantity of liquid held up in the column under normal operating conditions.
3.1.8 flood point, n—in distillation, the point at which the velocity of the upflowing vapors obstructs the down-coming reflux and
the column suddenly fills with liquid.
3.1.9 internal reflux, n—in distillation, the liquid normally running down inside the column.
3.1.10 pressure drop, n—the difference between the pressure measured in the condenser and the pressure measured in the
distillation flask.
3.1.10.1 Discussion—
It is expressed in kilopascals (mm Hg) per metre of packed height for packed columns, or kilopascals (mm Hg) overall for real
plate columns. It is higher for aromatics than for paraffins, and for higher molecular weights than for lighter molecules, at a given
boilup rate.
3.1.11 reflux ratio, R, n—in distillation, the ratio of the condensate at the head of the column that is returned to the column (reflux)
to that withdrawn as product.
3.1.12 static hold-up or wettage, n—the quantity of liquid retained in the column after draining at the end of a distillation.
3.1.12.1 Discussion—
It is characteristic of the packing or the design of the plates, and depends on the composition of the material in the column at the
final cut point and on the final temperature.
3.1.13 takeoff rate, n— in distillation, the volume of product withdrawn from the reflux divider over a specified period.
3.1.14 theoretical plate, n—the section of a column required to achieve thermodynamic equilibrium between a liquid and its vapor.
3.1.14.1 Discussion—
The height equivalent to one theoretical plate (HETP) for packed columns is expressed in millimetres. In the case of real plate
columns, the efficiency is expressed as the percentage of one theoretical plate that is achieved on one real plate.
4. Summary of Test Method
4.1 A weighed sample of 1 L to 30 L of stabilized crude petroleum is distilled to a maximum temperature of 400 °C AET in a
fractionating column having an efficiency at total reflux of at least 14, but not greater than 18, theoretical plates.
4.2 A reflux ratio of 5:1 is maintained at all operating pressures, except that at the lowest operating pressures between 0.674 kPa
and 0.27 kPa (5 mm and 2 mm Hg), a reflux ratio of 2:1 is optional. In cooperative testing or in cases of dispute, the stages of low
pressure, the reflux ratios, and the temperatures of cut points must be mutually agreed upon by the interested parties prior to
beginning the distillation.
4.3 Observations of temperature, pressure, and other variables are recorded at intervals and at the end of each cut or fraction.
4.4 The mass and density of each cut or fraction are obtained. Distillation yields by mass are calculated from the mass of all
fractions, including liquefied gas cut and the residue. Distillation yields by volume of all fractions and the residue at 15 °C are
calculated from mass and density.
4.5 From these data the TBP curves in mass or volume percent, or both, versus AET are drawn.
5. Significance and Use
5.1 This test method is one of a number of tests conducted on a crude oil to determine its value. It provides an estimate of the
yields of fractions of various boiling ranges and is therefore valuable in technical discussions of a commercial nature.
D2892 − 23
5.2 This test method corresponds to the standard laboratory distillation efficiency referred to as 15/5. The fractions produced can
be analyzed as produced or combined to produce samples for analytical studies, engineering, and product quality evaluations. The
preparation and evaluation of such blends is not part of this test method.
5.3 This test method can be used as an analytical tool for examination of other petroleum mixtures with the exception of LPG,
very light naphthas, and mixtures with initial boiling points above 400 °C.
6. Apparatus
6.1 Distillation at Atmospheric Pressure—All components must conform to the requirements specified as follows. Automatic
devices can be employed provided they meet the same requirements. A typical apparatus is illustrated in Fig. 1.
6.1.1 Distillation Flask—The distillation flask shall be of a size that is at least 50 % larger than the volume of the charge. The size
of the charge, between 1.0 L and 30 L, is determined by the holdup characteristics of the fractionating column, as shown in Table
1 and described in Annex A2. The distillation flask shall have at least one sidearm.
6.1.1.1 The sidearm is used as a thermowell. It shall terminate about 5 mm from the bottom of the flask to ensure its immersion
at the end of the distillation. When a second sidearm is present, it can be used for pressure drop detection with a nitrogen bleed
or for mechanical stirring, or both.
6.1.1.2 If a magnetic stirrer is used with a spherical flask, the flask shall have a slightly flattened or concave area at the bottom
on which the magnetic stirrer can rotate without grinding the glass. In this case, termination of the thermowell shall be off center
40 mm 6 5 mm to avoid the magnetic stirring bar. Boiling chips can be used as an alternative to a stirrer.
6.1.1.3 (Warning—While the advantage of visibility in glass distillation flasks is desirable, flasks of glass may become hazardous
the larger the charge they contain. For this reason, glass flasks of a volume greater than 10 L are not recommended.)
6.1.2 Heating System—Heating of the flask shall be provided in such a way that full boilup can be maintained at a steady rate at
all pressure levels. An electric heating mantle covering the lower half of the flask and having one third of the heat in an element
located in the bottom central area and the remaining two thirds in the rest of the hemisphere is recommended. While proportioning
controllers are preferred, heat input can be manually adjusted by use of a variable auto transformer on each circuit, the smaller
heater being automatically controlled by an instrument sensing the pressure drop of the column as registered in a differential
pressure instrument or alternatively by direct measurement of distillation rate.
6.1.2.1 Minimum wattage required to provide full boilup of crude petroleum is approximately 0.125 W ⁄mL of charge. Twice this
amount is recommended for quick heat-up.
2 2
6.1.2.2 The heat density in the flask heaters is approximately equal to 0.5 W ⁄cm to 0.6 W ⁄cm . This requires the use of
nickel-reinforced quartz fabric to ensure a reasonable service life.
6.1.2.3 Immersion heaters can be employed in a similar way and have the advantage of faster response, but they are more fragile
and require a specially designed flask to ensure that the heating elements remain immersed at the end of the run. When used, their
heat density should be approximately equal to 4 W ⁄cm .
6.1.2.4 The upper half of the flask shall be covered with a mantle to avoid unnecessary heat losses from the upper surface and
shall have an electric heater supplying about 0.25 W ⁄cm at full-rated voltage.
6.1.3 Fractionating Column—The fractionating column must contain either particulate packing or real plates similar to those
whose performance characteristics are summarized in Table 1 and meet the specifications stated in 6.1.3.1 through 6.1.3.4. Table
2 lists current North American suppliers of suitable packings.
6.1.3.1 The internal diameter shall be between 25 mm and 70 mm.
6.1.3.2 The efficiency shall be between 14 and 18 theoretical plates at total reflux when measured by the procedure described in
Annex A1.
D2892 − 23
FIG. 1 Apparatus
6.1.3.3 The fractionating column shall be comprised of a integral glass column and reflux divider totally enclosed in a highly
−6
reflective vacuum jacket having a permanent vacuum of less than 0.1 mPa (;10 mm Hg). It shall be essentially adiabatic when
tested in accordance with Annex A3.
NOTE 2—In the case of an adiabatic column when distilling a pure compound, the internal reflux is constant from top to bottom and is equal to the reflux
at the reflux divider. When distilling crude petroleum, the fractionation occurring in the dynamic holdup will cause a temperature gradient to be established
with attendant greater amount of internal reflux at the bottom of the column.
6.1.3.4 The column shall be enclosed in a heat-insulating system, such as a glass-fabric mantle, capable of maintaining the
temperature of the outer wall of the glass vacuum jacket equal to that of the internal vapor temperature. To verify this, the vacuum
D2892 − 23
TABLE 1 Data for n-Heptane-Methylcyclohexane Test Mixture at 75 % of Maximum Boilup and 101.3 kPa (760 mm Hg)
A,B,C,D,E F,G,H E,I,J E,K
Propak Helipak Perforated Plates Wire Mesh
Column diameter, mm 25 50 70 25 50 25 50 25 50
L L L L
Packing size, mm 4 6 6 No. 2917 No. 2918 NA NA NA NA
Boilup, mL/h × cm 650 670 675 300 350 640 660 810 1050
Dynamic holdup
L L
% of packed volume 17 15.3 17.0 15 14.3 NA NA 8.0 10.0
mL/theoretical plate 3.2 16 39 1.6 8.7 2.8 12.3 2.0 12.9
Pressure drop
L L
kPa/m 1.2 1.05 0.94 1.53 1.41 NA NA 0.97 0.75
L L
mm Hg/m 9.0 7.9 7.1 11.5 10.6 NA NA 7.3 5.6
kPa/theoretical plate 0.045 0.056 0.06 0.03 0.045 0.15 0.16 0.05 0.05
mm Hg/theoretical plate 0.34 0.42 0.43 0.24 0.34 1.1 1.2 0.35 0.37
HETP, mm (% of real plates) 38 53 61 21 32 (60 %) (65 %) 48 66
For 15-plate Towers
Packed height, cm (plates) 57 80 91 31.5 48 (25) (23) 72 99
L L
Packed volume, mL 280 1570 3460 155 917 NA NA 353 1940
Dynamic holdup, mL 47 240 590 23 131 42 184 28 194
Pressure drop
kPa 0.68 0.84 0.86 0.48 0.68 2.2 2.4 0.70 0.73
mm Hg 5.1 6.3 6.5 3.6 5.1 16.5 18.0 5.3 5.5
Charge volume, L
Min (4 % Holdup) 1.2 6.0 15 0.575 3.3 1.0 4.6 0.7 4.9
Max (1 % Holdup) 4.8 24.0 60 2.3 13.0 4.2 10.4 2.8 19.4
A
Cooke, G. M. and Jameson, B. G. Analytical Chemistry, Vol 27, 1955, p. 1798.
B
Struck, R. T. and Kinner, C. R. Industrial and Engineering Chemistry, Vol 42, 1950, p. 77.
C
Cannon, M. R. Industrial and Engineering Chemistry, Vol 41, No. 9, 1949, p. 1953.
D
Cannon Instrument Company, 2139 High Tech Rd., State College, PA 16803.
E
Cooke, G. M. Analytical Chemistry, Vol 39, 1967, p. 286.
F
Bulletin of Podbielniak Div. of Reliance Glass Works, P.O. Box 825, Bensenville, IL 60106.
G
Feldman, J., et al, Industrial and Engineering Chemistry, Vol 45, January 1953, p. 214.
H
Helipak Performance Characteristics, Begemean, C. R. and Turkal, P. J. (Laboratory Report of Podbielniak Inc.), 1950.
I
Umholtz, C. L. and Van Winkle, M. Petroleum Refiner, Vol 34, 1955, p. 114 for NH:MCH. Pressure Drop Calculated from data obtained on o- and m-xylene binary.
J
Oldershaw, C. F. Industrial and Engineering Chemistry, Vol 13, 1941, p. 265.
K
Bragg, L. B. Industrial and Engineering Chemistry, Vol 49, 1957, p. 1062.
L
NA = not applicable.
TABLE 2 North American Sources of Commercially Available
Packing Materials
Name Size Source
Propak 6 mm by 6 mm Cannon Instrument Company
2139 High Tech Rd.
State College, PA 16803
Helipak 2.5 mm by 4 mm Reliance Glass Works Inc.
P.O. Box 825
Bensenville, IL 60106
Perforated plates 25 mm and 50 mm Reliance Glass Works Inc.
P.O. Box 825
Bensenville, IL 60106
W.A. Sales Inc.
419 Harvester Ct.
Wheeling, IL 60090
Knitted wire mesh- Pegasus Industrial Specialties Ltd.
Goodloe multiknit P.O. Box 319
Agincourt, Ontario MIS 3B9 Canada
Packed Column Co.
970 New Durham Rd.
Edison, NJ 08817
jacket shall have a temperature sensor, such as a thermocouple, soldered to about 6 cm of thin copper or brass sheet and fastened
to the outer wall of the glass jacket at a level just below the reflux divider.
NOTE 3—For certain types of columns there is no significant difference in yields and fraction qualities between an uncompensated and a heat-compensated
column. In such a case, by mutual agreement between parties concerned, the application of a heated insulating system can be omitted.
6.1.3.5 The adjustable reflux divider shall be located about one column diameter above the top of the packing or topmost plate.
It must be capable of dividing the condensate with an accuracy of better than 90 % between the column and the takeoff line over
an approximate range of rates from 10 % to 90 % of the maximum boil up rate of the column when determined in accordance with
Annex A7.
D2892 − 23
6.1.4 Condenser—The condenser shall have sufficient capacity to condense essentially all the C and C vapors from the crude
4 5
at the specified rate, using a coolant temperature of −20 °C.
6.1.5 Cold Traps—Two efficient traps of adequate capacity cooled by dry ice and alcohol mixture shall be connected in series to
the vent line of the condenser when light hydrocarbons are present, as at the beginning of the distillation. For vacuum distillation,
a Dewar-style trap also cooled by dry ice is used to protect the vacuum gauge from vapors.
6.1.6 Gas Collector—If uncondensed gas is to be measured, a gas meter can be connected to the outlet of the cold trap but with
a calcium chloride drying tube between them to keep moisture from collecting in the traps. When analysis of the gas sample is
required, the gas can be collected in an empty plastic balloon of suitable size either in place of the meter or following it. The
volume of its contents can be determined by calculation from the rise in pressure after expanding the sample into an evacuated
vessel of known volume.
6.1.7 Fraction Collector—This part of the apparatus permits the collection of the distillate without interruption during withdrawal
of product from the receiver under atmospheric or reduced pressure. It also permits removal of product from the vacuum system
without disturbing conditions in the column.
6.1.8 Product Receivers—The receivers shall be of suitable size for the quantity of crude petroleum being distilled. The
recommended capacity is from 100 mL to 500 mL. They shall be calibrated and graduated to permit reading to the nearest 1 %.
6.2 Distillation Under Reduced Pressure—In addition to the apparatus listed in 6.1, the apparatus for distillation under reduced
pressure shall include the following:
6.2.1 Vacuum Pump—The vacuum system shall be capable of maintaining smooth pressure operation at all pressure levels. It shall
have the capacity to draw down the pressure in the receiver(s) from atmospheric to 0.25 kPa (2 mm Hg) in less than 30 s so as
to avoid disturbance of the system during emptying of receivers under vacuum. Alternatively, a separate pump can be employed
for this purpose.
6.2.2 Vacuum Gauge—The point of connection of the vacuum gauge to the system shall be as close as practical to the reflux
dividing head. The connecting tubing shall be of sufficient diameter to ensure that no measurable pressure drop occurs in the line.
In no case shall the vacuum gauge connection be near the vacuum pump.
6.2.2.1 All gauges shall be carefully protected from condensable vapors, especially water vapor, by a cold trap maintained at the
temperature of dry ice.
6.2.3 Pressure Regulator—The regulator shall maintain the pressure in the system essentially constant at all operating pressures.
Automatic regulation can be achieved by a device that regulates the demand on the vacuum source. A satisfactory device is a
solenoid valve positioned between the vacuum source and a surge tank of at least 10 L capacity. Alternatively, a manual bleed valve
can be maintained by a trained operator with a minimum of attention.
6.3 Sensing and Recording Apparatus:
6.3.1 Temperature Sensors—Only temperature measurement systems meeting the requirements of 6.3.1.1 and 6.3.1.2 shall be
used.
6.3.1.1 The vapor temperature sensor can be a platinum resistance thermometer, a Type J thermocouple with the junction head
fused to the lower tip of the thermowell, or any other device that meets the requirements in this paragraph and 6.3.1.2. The tip of
the sensor shall be located above the top of the packing or the topmost glass plate and in close proximity to the reflux divider but
not in contact with the liquid reflux. The location of the vapor temperature sensor shall be proved by the test method described
in Annex A4. The sensor shall have a cooling time of not more than 175 s, as described in Annex A5.
6.3.1.2 The vapor temperature measuring device shall have an accuracy of 0.5 °C or better and be measured with a resolution of
0.1 °C or better. The liquid temperature measuring device shall have an accuracy of 1.0 °C or better and be measured with a
resolution of 0.5 °C or better. Temperatures are recorded either manually or automatically.
6.3.1.3 Temperature sensors shall be calibrated as described in Annex A6. Alternatively certified sensors may be used, provided
the calibration of the sensor and its associated recording instrument can be traced back to a primary temperature standard.
D2892 − 23
FIG. 2 Approximate Pressure Drop-Fractionators Using Propak
Temperature sensors are calibrated over the full range of temperature (from 0 °C to 327.4 °C) at the time of first use of the sensor
in combination with its associated instrument. Recalibrate when either the sensor or the instrument is repaired or serviced.
Verification of the calibration of the temperature sensors is to be made on a regular basis. For vapor temperature sensors,
verification at least once a month is recommended and for liquid temperature sensors once every six months. Verification of the
calibration of the sensors can be accomplished potentiometrically by the use of standard precision resistance or by distilling a pure
compound with accurately known boiling point.
6.3.2 Vacuum Gauge—Primary standards, such as the non-tilting McLeod gauge, mercury manometer, or other analogous primary
standard pressure devices can be used without calibration when properly used and maintained. A mercury manometer, however,
will only be of satisfactory accuracy down to a pressure of about 1 kPa and then only when read with a good cathetometer (an
instrument based on a telescope mounted on a vernier scale to determine levels very accurately). Alternatively, a tensimeter or
certified electronic sensors may be used, provided the calibration of the sensor and its associated recording instrument can be traced
back to a primary pressure standard. Sensors of the diaphragm type have been found satisfactory. Vacuum gauges based on hot
wires, radiation, or electrical conductivity detectors are not recommended.
6.3.2.1 The gauge for measuring subatmospheric pressures shall have an accuracy at least equal to that stated as follows:
Distillation Pressure Accuracy
kpa mm Hg kPa mm Hg
100-13.3 760 to 100 0.13 1.0
13.3-1.33 99 to 10 0.013 0.1
1.33-0.266 9 to 2 0.006 0.06
6.3.2.2 Noncertified gauges shall be calibrated from a primary standard or a secondary electronic standard traceable to a primary
standard. A basic calibration procedure is described in Annex A6. Recalibrate when either the sensor or the instrument is repaired
or serviced. Verification of the calibration of the electronic pressure sensors is to be made on a regular basis. A frequency of at
least once a month is recommended. Verification of the calibration of the sensors can be accomplished using the procedures
described in Annex A6 or against a certified reference system.
6.3.3 Boilup Rate—The boilup rate is normally controlled by sensing the pressure drop in the column. The pressure drop during
operation is measured by means of a manometer or pressure transducer connected between the flask and the condenser. Prevention
of condensation in the connecting tube can be accomplished by injecting a very small flow of nitrogen (8 cm /s) between the
pressure drop sensor manometer and the flask (see Fig. 1) or by placing a small water-cooled condenser between the flask and the
pressure drop sensor. Alternatively, the boilup rate can be controlled from the measurement of take off rate.
7. Verification of Apparatus Performance
7.1 Test Method D2892 provides a standard framework for the laboratory distillation of crude oils in order to produce cuts of
defined quality (for further testing) and the concurrent production of a boiling point curve. As the quantity requirements and cut
points might be widely different between companies and application areas, this test method does not standardize on equipment
design but on equipment performance.
D2892 − 23
7.2 The nature of the test method (the use of large sample quantities and very time consuming) and the nature of the product being
tested (highly volatile and unstable material), precludes the use of standard statistical control techniques. Moreover, this test
method does not produce a single result, nor is the series of results (the boiling point curve) derived under rigidly defined
conditions (see 4.2).
7.3 Equipment performance in the context of Test Method D2892 consists of two elements; the efficiency of the column, defining
cut quality, and the correctness of the cut point (AET), defining the boiling point curve.
7.4 The correctness of the AET is mainly, but not only, dependent on the accuracy of the (vapor) temperature and (operating)
pressure sensors (Annex A6). Other factors affecting the accuracy and precision of the boiling point curve are:
7.4.1 The location of the temperature and pressure sensor (Annex A4).
7.4.2 The dynamic response of the sensors (Annex A5).
7.4.3 The correct operation of the reflux divider (Annex A7).
7.4.4 The heat loss from the column (Annex A3).
7.4.5 The efficiency of the column (Annex A1).
7.4.6 These factors are basically covered through the appropriate annexes. However, it should be realized that this takes care only
of individual components and does not cover the combined effect of small deviations. Moreover, the aforementioned tests are all
done under more or less static conditions, not necessarily representative for the behavior of the system under actual dynamic
conditions.
7.5 Cut quality is mainly defined by the efficiency of the column (Annex A1), but is also affected by:
7.5.1 The correct operation of the reflux divider (reflux ratio) (Annex A7).
7.5.2 The heat loss from the column, that is, internal reflux (Annex A3).
7.5.3 The dynamic hold up of the column (Annex A2).
7.5.4 Column efficiency is covered in this test method through Table 1 and Annex A1. However, Table 1 only provides an
assumption on efficiency and is not a guarantee. Annex A1 only provides a check under static conditions, infinite reflux ratio, rather
low actual temperatures and a binary component system. Hence, although there is some safeguard on standard performance,
through conformance to Annex A1, Annex A2, Annex A3, and Annex A7, again it remains questionable whether this is truly
representative for columns under actual operating conditions.
7.6 Theoretically, an overall performance check, like the one described in Appendix X2, is capable of verifying the performance
of a column and the correctness of the AET under actual operating conditions. Appendix X2, in principle, measures the combined
effect of all factors affecting the results of Test Method D2892.
7.6.1 The minimum tray number as defined in Appendix X2 is a measure of overall cut quality, and the difference between nominal
cut point (AET) and effective cut point (ECP as defined in Appendix X2) provides a measure for the correctness of the AET.
However, insufficient data are available right now to define the allowable tolerances in a rigid statistical way. Moreover, the test
method described is very labor intensive and precludes its use on a regular, short time interval basis and, therefore, its use as a
mandatory statistical control technique.
7.6.2 Appendix X2, therefore, provides only recommended guidelines for statistical control on column performance, including
both correctness of AET and column efficiency. It is the responsibility of the laboratory to provide for sufficient quality controls
to guarantee conformance to the test method.
D2892 − 23
8. Sampling
8.1 Obtain a sample for distillation in accordance with instructions given in Practice D4057 or D4177. The sample must be
received in a sealed container and show no evidence of leakage.
8.2 Cool the sample to between 0 °C and 5 °C by placing it in a refrigerator for several hours (preferably overnight) before
opening.
8.3 If the sample appears waxy or too viscous, raise the temperature to 5 °C above its pour point.
8.4 Agitate the sample by whatever means are appropriate to its size to ensure that it is well mixed.
8.5 Determine the water content of the sample by Test Method D4006 or any other suitable method. If the water content exceeds
0.3 % volume, the sample shall be dehydrated prior to fractional distillation. A suitable practice for dehydration of wet crude oil
samples is described in Appendix X1.
NOTE 4—Attempts to distill wet crude oil samples in glass columns might result in breakage of the glassware, which poses a potential fire hazard.
Moreover, the presence of water will affect the accuracy of distillation yield in the naphtha region. These effects are more pronounced for heavy crude
oils, containing low amounts of hydrocarbons boiling below 100 °C, than for light crudes where there is usually sufficient hydrocarbon vapor generated
to form an azeotrope and drive the water vapors through the column without problems.
9. Preparation of Apparatus
9.1 Clean and dry the distillation column and all the ancillary glass apparatus before the distillation begins.
9.2 Ensure that the system is leak-free and all heaters, control devices, and instruments are on and in working order. A clock or
other timing device should be ready for use.
10. Procedure
10.1 Charging:
10.1.1 The charge size shall be such that the dynamic hold up as determined in accordance with Annex A2 is between 1 % and
4 % of the charge when operating at 75 % of maximum boilup (see Table 1). Chill the flask to a temperature not lower then 0 °C.
10.1.2 Insert the stirring device or place some pieces of glass or porcelain into the flask to control bumping.
10.1.3 Determine the density of the sample by Test Method D941, D1217, or D1298.
10.1.4 Calculate to within 65 % the mass of crude petroleum corresponding to the desired volume of the charge. Weigh to the
nearest 1 % this quantity of sample into the flask.
10.1.5 Attach the flask to the column and connect the pressure drop measuring device. Install the heating system, stirrer, and
support device. (Warning—Poisonous H S gas is frequently evolved from crude oil and precautions must be taken either to absorb
the gas that passes through the cold trap or to vent it to a safe place.)
10.2 Debutanization:
10.2.1 For necessary apparatus refer to 6.1.5 and 6.1.6.
10.2.2 Begin circulation of refrigerant at a temperature no higher than −20 °C in the condenser, distillate cooler, and receiver, if
so equipped.
10.2.3 Record the barometric pressure at the beginning and periodically throughout the distillation.
10.2.4 Apply heat to the flask at such a rate that vapors reach the top of the column between 20 min and 50 min after startup.
D2892 − 23
Adjust heat input so as to achieve a pressure drop of less than 0.13 kPa ⁄m (1.0 mm Hg/m) in packed columns or less than 0.065 kPa
(0.5 mm Hg) in real plate columns. Program automated equipment in accordance with the preceding directions. Turn on the stirring
device if used.
10.2.5 Allow the column to operate at total reflux until the vapor temperature reaches equilibrium but not longer than 15 min after
the first drop of condensate appears in the reflux divider.
10.2.6 Record the vapor temperature as the initial vapor temperature.
10.2.7 Stop the circulation of the refrigerant and observe the vapor temperature. When the vapor temperature reaches 15 °C, start
the circulation of refrigerant again.
10.2.8 If the vapor temperature drops below 15 °C, continue refluxing for at least 15 min. Repeat 10.2.7. If the vapor temperature
remains at 15 °C or rises, continue with the atmospheric distillation. (Warning—The following three steps should not be done until
after the first naphtha cut has been removed to ensure that all the light gases have been recovered.)
10.2.9 Remove and weigh the dry ice traps containing light hydrocarbon liquid after carefully wiping them dry.
10.2.10 Sample the contents of the first dry ice trap using a 10 mL to 50 mL pressure vessel evacuated to no lower than 26.6 kPa
(200 mm Hg). Keep all containers at the temperature of dry ice to ensure no loss of volatiles. The first trap next to the condenser
should contain all of the sample. If condensate is found in the second trap, sample both traps or combine the contents before
sampling.
10.2.11 Submit the trap sample and gas balloon, if used, for analysis by a suitable gas chromatographic test method to be reported
on a fixed-gas free basis. Test Methods D6729, D6730, and D6733, equipped with liquid or gas sampling valves, or both, for
sample introduction equipment have been used successfully for this analysis.
10.3 Distillation at Atmospheric Pressure:
10.3.1 Maintain a temperature below −20 °C in the lines of the distillate cooler and receiver as well as in the condenser. Turn on
the column mantle heat controller and maintain the column jacket temperature 0 °C to 5 °C below the vapor temperature.
10.3.2 Regulate the heat input as necessary to establish and maintain a boilup rate approximately 75 % of maximum. Fig. 3 can
be used as a guide for Propak. Rates for other sizes can be estimated by multiplying the boilup rate in Table 1 by the cross-sectional
area of the column and dividing by the sum of the reflux ratio + 1.
FIG. 3 Expected Takeoff Rates at 5:1 Reflux Ratio for Fractionators Using Propak
D2892 − 23
NOTE 5—Boilup rate is expressed in millilitres of liquid per hour for a given column or in millilitres per hour per square centimetre of cross-sectional
area for comparative purposes. In the latter case, it refers to the test mixture of n-heptane and methylcyclohexane in the efficiency evaluation (see Annex
A1) and is measured at the bottom of the column. The maximum boilup of the n-heptane-methylcyclohexane test mixture is that which the column can
handle under stable conditions without flooding. In routine adiabatic operation, the boilup rate can be estimated roughly from the takeoff rate multiplied
by the reflux ratio plus one.
10.3.3 Commence takeoff at a reflux ratio of 5:1 and total cycle time of not over 36 s nor less than 24 s.
NOTE 6—The vapor reaching the top of the column is totally condensed and the resulting liquid is divided into two parts. One part L (reflux), is returned
to the column and the other part, D (distillate), is withdrawn as product. The reflux ratio (R = L ⁄D) can vary from zero at total takeoff (L = 0) to infinity
at total reflux (D = 0).
10.3.4 Take off distillate in separate and consecutive fractions of suitable size. The recommended size of fraction is that
corresponding to 5 °C or 10 °C in vapor temperature. Collect fractions boiling below 65 °C in receivers cooled to 0 °C or below.
When the vapor temperature reaches 65 °C, refrigerant in the condenser and related coolers can be discontinued and water at
ambient temperature substituted.
10.3.5 At the end of each fraction and at each cut point, record the following observations:
10.3.5.1 Time in hours and minutes,
10.3.5.2 Volume in millilitres,
10.3.5.3 Vapor temperature in °C to the nearest 0.5 °C,
10.3.5.4 Temperature of the boiling liquid in °C to the nearest 1 °C,
10.3.5.5 Atmospheric pressure in kPa (mm Hg), and
10.3.5.6 Pressure drop in the column in kPa (mm Hg).
10.3.6 If signs of flooding are observed, reduce the heating rate while continuing takeoff until steady conditions are restored. If
a cut point is encountered during this period, stop the distillation, cool the charge, and recombine the off-condition cuts. Restart
the distillation with a period at total reflux, not to exceed 15 min, to restore operating conditions before continuing takeoff. Do not
make a cut within 5 °C of startup.
10.3.7 Continue taking cuts until the desired maximum vapor temperature is reached or until the charge shows signs of cracking.
Pronounced cracking is evidenced by a fog appearing in the flask and later at the reflux divider. Do not allow the vapor temperature
to exceed 210 °C nor the temperature of the boiling liquid to exceed 310 °C.
10.3.8 Shut off the reflux valve and the heating system. Allow the contents to cool to such a temperature that the distillation can
be commenced at 13.3 kPa (100 mm Hg) without flooding. This temperature can be estimated by adding the ΔT between the liquid
and vapor temperatures found for the column during atmospheric operation to the expected initial vapor temperature at the reduced
pressure, or by subtracting the ΔT from the last recorded liquid temperature.
NOTE 7—Cooling of the liquid in the flask can be accelerated by blowing a gentle stream of compressed air onto the flask after its heating mantle has
been removed. Avoid strong jets of cold air. Alternately, turn on coolant in the quench coil of the flask, if used.
10.3.9 Weigh all fractions and determine their densities.
10.3.10 Submit the first distillate fraction for analysis by gas chromatography.
10.4 Distillation at 13.3 kPa (100 mm Hg):
10.4.1 If further cuts at higher temperatures are required, distillation can be continued at reduced pressures, subject to the
maximum temperature that the boiling liquid will stand without significant cracking. This is about 310 °C in most cases. Notable
D2892 − 23
exceptions are crude oils containing heat-sensitive sulfur compounds. In any case, do not make a cut within 5 °C of the temperature
at startup because the column will not be at equilibrium.
10.4.2 Connect a vacuum pumping and control system to the apparatus as shown in Fig. 1.
10.4.3 Start the vacuum pump and adjust the pressure downward gradually to the value of 13.3 kPa (100 mm Hg) or set the
pressure regulator at this value. The temperature of the liquid in the flask must be below that at which it will boil at 13.3 kPa
(100 mm Hg). If the liquid boils before this pressure is reached, increase the pressure and cool further until the desired pressure
can be achieved without boiling.
10.4.4 Apply heat to the boiler and reestablish reflux at any moderate rate in the reflux divider for about 15 min to reheat the
column to operating temperature. Momentarily stop heat input and raise the pressure with N for 1 min to drop the holdup into
the distillation flask.
10.4.5 Reapply heat to the distillation flask and adjust the rate of heating to maintain a constant pressure drop equivalent to the
boilup rate of approximately 75 % of the maximum rate for this pressure and begin takeoff without delay. The approximate pressure
drops required for this purpose are indicated in Fig. 3. Maintain a column insulation temperature 0 °C to 5 °C below the vapor
temperature throughout the operation.
10.4.6 Remove separately, cuts of suitable size as in 10.3.4.
10.4.7 At the end of each distillate fraction and at each cut point, record the following observations:
10.4.7.1 Time in hours and minutes,
10.4.7.2 Volume in millilitres observed at ambient temperature,
10.4.7.3 Vapor temperature in °C to the nearest 0.5 °C with correction, if any,
10.4.7.4 Temperature of the boiling liquid in °C to the nearest 1 °C,
10.4.7.5 Pressure drop in the column in kPa (mm Hg),
10.4.7.6 Operating pres
...



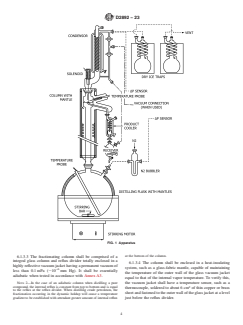




Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...