ASTM F2077-22
(Test Method)Standard Test Methods for Intervertebral Body Fusion Devices
Standard Test Methods for Intervertebral Body Fusion Devices
SIGNIFICANCE AND USE
5.1 Intervertebral body fusion device assemblies are generally simple geometric-shaped devices which are often porous or hollow in nature. Their function is to support the anterior column of the spine to facilitate arthrodesis of the motion segment. This test method outlines materials and methods for the characterization and evaluation of the mechanical performance of different intervertebral body fusion device assemblies so that comparisons can be made between different designs.
5.2 This test method is designed to quantify the static and dynamic characteristics of different designs of intervertebral body fusion device assemblies. These tests are conducted in vitro to allow for analysis and comparison of the mechanical performance of intervertebral body fusion device assemblies to specific force modalities.
5.3 The forces applied to the intervertebral body fusion assemblies may differ from the complex loading seen in vivo, and therefore, the results from these tests may not directly predict in vivo performance. The results, however, can be used to compare mechanical performance of different intervertebral body fusion device assemblies.
5.4 Since the environment may affect the dynamic performance of intervertebral body fusion device assemblies, dynamic testing in a saline environment may be considered. Fatigue tests should first be conducted in air (at ambient temperature) for comparison purposes since the environmental effects could be significant. If a simulated in vivo environment is desired, the investigator should consider testing in a saline environmental bath at 37 °C (for example, 0.9 g NaCl per 100 mL water) at a rate of 1 Hz or less. A simulated body fluid, a saline drip or mist, distilled water, or other type of lubrication at 37 °C could also be used with adequate justification.
5.5 If the devices are known to be temperature and environment dependent, testing should be conducted in physiologic solution as described in 5.4. Devices that require...
SCOPE
1.1 This test method covers the materials and methods for the static and dynamic testing of intervertebral body fusion device assemblies, spinal implants designed to promote arthrodesis at a given spinal motion segment.
1.2 This test method is intended to provide a basis for the mechanical comparison among past, present, and future nonbiologic intervertebral body fusion device assemblies. This test method allows comparison of intervertebral body fusion device assemblies with different intended spinal locations and methods of application to the intradiscal spaces. This test method is intended to enable the user to compare intervertebral body fusion device assemblies mechanically and does not purport to provide performance standards for intervertebral body fusion device assemblies.
1.3 The test method describes static and dynamic tests by specifying force types and specific methods of applying these forces. These tests are designed to allow for the comparative evaluation of intervertebral body fusion device assemblies.
1.4 These tests are designed to characterize the structural integrity of the device and are not intended to test the bone-implant interface.
1.5 This test method does not address expulsion testing of intervertebral body fusion device assemblies (see 1.4).
1.6 Guidelines are established for measuring displacements, determining the yield force or moment, and evaluating the stiffness and strength of the intervertebral body fusion device assemblies.
1.7 Some intervertebral body fusion device assemblies may not be testable in all test configurations.
1.8 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard, with the exception of angular measurements, which may be reported in terms of either degrees or radians.
1.9 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is ...
General Information
- Status
- Published
- Publication Date
- 30-Jun-2022
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.25 - Spinal Devices
Relations
- Effective Date
- 15-Feb-2024
- Effective Date
- 01-Feb-2024
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Oct-2016
- Effective Date
- 01-Jun-2014
- Effective Date
- 01-May-2014
- Effective Date
- 01-May-2013
- Effective Date
- 01-May-2013
- Effective Date
- 15-Dec-2012
- Effective Date
- 15-Nov-2012
- Effective Date
- 01-Sep-2012
- Effective Date
- 01-Aug-2012
- Effective Date
- 15-May-2012
- Effective Date
- 15-Mar-2012
- Effective Date
- 01-Dec-2011
Overview
ASTM F2077-22: Standard Test Methods for Intervertebral Body Fusion Devices specifies standardized procedures for mechanical testing of intervertebral body fusion device assemblies, which are spinal implants designed to promote arthrodesis (bone fusion) at a particular spinal motion segment. These devices, typically placed in the disc space between adjacent vertebral bodies, are essential components in spinal surgery, supporting the anterior column and enhancing patient outcomes.
This ASTM standard provides both static and dynamic mechanical test methods, enabling the comparative evaluation of various intervertebral body fusion device designs. By defining test environments, apparatus, and reporting requirements, ASTM F2077-22 ensures consistency and reliability in mechanical assessment, supporting innovation and safety in spinal implant development.
Key Topics
Scope of Testing:
- Covers static and dynamic testing of nonbiologic intervertebral body fusion devices.
- Provides procedures to compare devices across different spinal regions (lumbar, thoracic, cervical).
- Does not establish minimum performance standards, but enables mechanical comparison.
Types of Mechanical Testing:
- Static Tests: Assess ultimate strength, stiffness, and yield force/moment.
- Dynamic (Fatigue) Tests: Determine endurance limits through repeated cycling to simulate in vitro use.
- Test Setups: Include axial compression, compression-shear, and torsion configurations.
Test Apparatus and Conditions:
- Use of polyacetal or stainless steel blocks to mimic vertebral end plates, with geometry matched to implant design.
- Environmental testing in air (ambient) and, when justified, in physiologic solutions (e.g., saline at 37°C).
Reporting Requirements:
- Comprehensive documentation of test setup, sample details, loads/frequencies, apparatus configuration, and observed failure modes.
- Provision of force-displacement plots, tabulated results, and photographic evidence.
Applications
Device Comparison and Benchmarking:
- Enables manufacturers and researchers to benchmark new intervertebral body fusion device designs against current and previous models.
- Facilitates evaluation of mechanical properties (stiffness, strength, fatigue resistance) across devices intended for various spinal segments.
Regulatory and Quality Assurance:
- Provides consistent protocols that support regulatory submissions and product safety claims for spinal implant devices.
- Assists in demonstrating compliance with stringent mechanical performance expectations for spine surgery implants.
Product Development and Innovation:
- Supports R&D teams in optimizing designs through repeatable, comparative test data.
- Encourages innovation by offering well-defined methodologies to assess novel materials and configurations.
Clinical Relevance:
- While testing is in vitro and may not fully replicate in vivo conditions, results are valuable for informed selection of devices, understanding loading behavior, and anticipating performance under expected spinal loads.
Related Standards
- ASTM E4 - Practices for Force Calibration and Verification of Testing Machines
- ASTM E6 - Terminology Relating to Methods of Mechanical Testing
- ASTM E177 - Practice for Use of Terms Precision and Bias in ASTM Test Methods
- ASTM E691 - Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
- ASTM E1823 - Terminology Relating to Fatigue and Fracture Testing
- ASTM E2309 - Practices for Verification of Displacement Measuring Systems and Devices Used in Material Testing Machines
- ASTM F1582 - Terminology Relating to Spinal Implants
By following ASTM F2077-22, industry professionals benefit from tested and trusted methodologies, promoting safer and more effective intervertebral body fusion device assemblies in spinal care.
Buy Documents
ASTM F2077-22 - Standard Test Methods for Intervertebral Body Fusion Devices
REDLINE ASTM F2077-22 - Standard Test Methods for Intervertebral Body Fusion Devices
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F2077-22 is a standard published by ASTM International. Its full title is "Standard Test Methods for Intervertebral Body Fusion Devices". This standard covers: SIGNIFICANCE AND USE 5.1 Intervertebral body fusion device assemblies are generally simple geometric-shaped devices which are often porous or hollow in nature. Their function is to support the anterior column of the spine to facilitate arthrodesis of the motion segment. This test method outlines materials and methods for the characterization and evaluation of the mechanical performance of different intervertebral body fusion device assemblies so that comparisons can be made between different designs. 5.2 This test method is designed to quantify the static and dynamic characteristics of different designs of intervertebral body fusion device assemblies. These tests are conducted in vitro to allow for analysis and comparison of the mechanical performance of intervertebral body fusion device assemblies to specific force modalities. 5.3 The forces applied to the intervertebral body fusion assemblies may differ from the complex loading seen in vivo, and therefore, the results from these tests may not directly predict in vivo performance. The results, however, can be used to compare mechanical performance of different intervertebral body fusion device assemblies. 5.4 Since the environment may affect the dynamic performance of intervertebral body fusion device assemblies, dynamic testing in a saline environment may be considered. Fatigue tests should first be conducted in air (at ambient temperature) for comparison purposes since the environmental effects could be significant. If a simulated in vivo environment is desired, the investigator should consider testing in a saline environmental bath at 37 °C (for example, 0.9 g NaCl per 100 mL water) at a rate of 1 Hz or less. A simulated body fluid, a saline drip or mist, distilled water, or other type of lubrication at 37 °C could also be used with adequate justification. 5.5 If the devices are known to be temperature and environment dependent, testing should be conducted in physiologic solution as described in 5.4. Devices that require... SCOPE 1.1 This test method covers the materials and methods for the static and dynamic testing of intervertebral body fusion device assemblies, spinal implants designed to promote arthrodesis at a given spinal motion segment. 1.2 This test method is intended to provide a basis for the mechanical comparison among past, present, and future nonbiologic intervertebral body fusion device assemblies. This test method allows comparison of intervertebral body fusion device assemblies with different intended spinal locations and methods of application to the intradiscal spaces. This test method is intended to enable the user to compare intervertebral body fusion device assemblies mechanically and does not purport to provide performance standards for intervertebral body fusion device assemblies. 1.3 The test method describes static and dynamic tests by specifying force types and specific methods of applying these forces. These tests are designed to allow for the comparative evaluation of intervertebral body fusion device assemblies. 1.4 These tests are designed to characterize the structural integrity of the device and are not intended to test the bone-implant interface. 1.5 This test method does not address expulsion testing of intervertebral body fusion device assemblies (see 1.4). 1.6 Guidelines are established for measuring displacements, determining the yield force or moment, and evaluating the stiffness and strength of the intervertebral body fusion device assemblies. 1.7 Some intervertebral body fusion device assemblies may not be testable in all test configurations. 1.8 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard, with the exception of angular measurements, which may be reported in terms of either degrees or radians. 1.9 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is ...
SIGNIFICANCE AND USE 5.1 Intervertebral body fusion device assemblies are generally simple geometric-shaped devices which are often porous or hollow in nature. Their function is to support the anterior column of the spine to facilitate arthrodesis of the motion segment. This test method outlines materials and methods for the characterization and evaluation of the mechanical performance of different intervertebral body fusion device assemblies so that comparisons can be made between different designs. 5.2 This test method is designed to quantify the static and dynamic characteristics of different designs of intervertebral body fusion device assemblies. These tests are conducted in vitro to allow for analysis and comparison of the mechanical performance of intervertebral body fusion device assemblies to specific force modalities. 5.3 The forces applied to the intervertebral body fusion assemblies may differ from the complex loading seen in vivo, and therefore, the results from these tests may not directly predict in vivo performance. The results, however, can be used to compare mechanical performance of different intervertebral body fusion device assemblies. 5.4 Since the environment may affect the dynamic performance of intervertebral body fusion device assemblies, dynamic testing in a saline environment may be considered. Fatigue tests should first be conducted in air (at ambient temperature) for comparison purposes since the environmental effects could be significant. If a simulated in vivo environment is desired, the investigator should consider testing in a saline environmental bath at 37 °C (for example, 0.9 g NaCl per 100 mL water) at a rate of 1 Hz or less. A simulated body fluid, a saline drip or mist, distilled water, or other type of lubrication at 37 °C could also be used with adequate justification. 5.5 If the devices are known to be temperature and environment dependent, testing should be conducted in physiologic solution as described in 5.4. Devices that require... SCOPE 1.1 This test method covers the materials and methods for the static and dynamic testing of intervertebral body fusion device assemblies, spinal implants designed to promote arthrodesis at a given spinal motion segment. 1.2 This test method is intended to provide a basis for the mechanical comparison among past, present, and future nonbiologic intervertebral body fusion device assemblies. This test method allows comparison of intervertebral body fusion device assemblies with different intended spinal locations and methods of application to the intradiscal spaces. This test method is intended to enable the user to compare intervertebral body fusion device assemblies mechanically and does not purport to provide performance standards for intervertebral body fusion device assemblies. 1.3 The test method describes static and dynamic tests by specifying force types and specific methods of applying these forces. These tests are designed to allow for the comparative evaluation of intervertebral body fusion device assemblies. 1.4 These tests are designed to characterize the structural integrity of the device and are not intended to test the bone-implant interface. 1.5 This test method does not address expulsion testing of intervertebral body fusion device assemblies (see 1.4). 1.6 Guidelines are established for measuring displacements, determining the yield force or moment, and evaluating the stiffness and strength of the intervertebral body fusion device assemblies. 1.7 Some intervertebral body fusion device assemblies may not be testable in all test configurations. 1.8 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard, with the exception of angular measurements, which may be reported in terms of either degrees or radians. 1.9 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is ...
ASTM F2077-22 is classified under the following ICS (International Classification for Standards) categories: 11.040.20 - Transfusion, infusion and injection equipment. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F2077-22 has the following relationships with other standards: It is inter standard links to ASTM E1823-24a, ASTM E1823-24, ASTM E1823-20, ASTM F1582-98(2016), ASTM E4-14, ASTM E177-14, ASTM E177-13, ASTM E691-13, ASTM E1823-12e, ASTM E1823-12d, ASTM E1823-12c, ASTM E1823-12b, ASTM E1823-12a, ASTM E1823-12, ASTM F1582-98(2011). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F2077-22 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F2077 − 22
Standard Test Methods for
Intervertebral Body Fusion Devices
This standard is issued under the fixed designation F2077; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 1.9 This standard does not purport to address all of the
safety concerns, if any, associated with its use. It is the
1.1 This test method covers the materials and methods for
responsibility of the user of this standard to establish appro-
the static and dynamic testing of intervertebral body fusion
priate safety, health, and environmental practices and deter-
deviceassemblies,spinalimplantsdesignedtopromotearthro-
mine the applicability of regulatory limitations prior to use.
desis at a given spinal motion segment.
1.10 This international standard was developed in accor-
1.2 This test method is intended to provide a basis for the
dance with internationally recognized principles on standard-
mechanical comparison among past, present, and future non-
ization established in the Decision on Principles for the
biologicintervertebralbodyfusiondeviceassemblies.Thistest
Development of International Standards, Guides and Recom-
methodallowscomparisonofintervertebralbodyfusiondevice
mendations issued by the World Trade Organization Technical
assemblies with different intended spinal locations and meth-
Barriers to Trade (TBT) Committee.
ods of application to the intradiscal spaces.This test method is
intended to enable the user to compare intervertebral body 2. Referenced Documents
fusion device assemblies mechanically and does not purport to 2
2.1 ASTM Standards:
provide performance standards for intervertebral body fusion
E4Practices for Force Calibration and Verification of Test-
device assemblies.
ing Machines
1.3 The test method describes static and dynamic tests by E6Terminology Relating to Methods of MechanicalTesting
E177Practice for Use of the Terms Precision and Bias in
specifying force types and specific methods of applying these
ASTM Test Methods
forces. These tests are designed to allow for the comparative
evaluation of intervertebral body fusion device assemblies. E691Practice for Conducting an Interlaboratory Study to
Determine the Precision of a Test Method
1.4 These tests are designed to characterize the structural
E1823TerminologyRelatingtoFatigueandFractureTesting
integrity of the device and are not intended to test the
E2309Practices forVerification of Displacement Measuring
bone-implant interface.
Systems and Devices Used in Material Testing Machines
1.5 This test method does not address expulsion testing of
F1582Terminology Relating to Spinal Implants
intervertebral body fusion device assemblies (see 1.4).
3. Terminology
1.6 Guidelinesareestablishedformeasuringdisplacements,
3.1 For definition of terms refer to Terminologies E6,
determining the yield force or moment, and evaluating the
E1823, and F1582.
stiffness and strength of the intervertebral body fusion device
assemblies.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 coordinate system/axes, n—three orthogonal axes are
1.7 Some intervertebral body fusion device assemblies may
defined as follows. The center of the coordinate system is
not be testable in all test configurations.
locatedatthegeometriccenteroftheintervertebralbodyfusion
1.8 The values stated in SI units are to be regarded as
device assembly. The XY plane is to bisect the sagittal plane
standard. No other units of measurement are included in this
angle between superior and inferior lines (surfaces) that are
standard, with the exception of angular measurements, which
intended to simulate the adjacent vertebral end plates. The
may be reported in terms of either degrees or radians.
positive Z axis is to be directed superiorly. Force components
parallel to the XY plane are shear components of loading. The
compressive axial force is defined to be the component in the
These test methods are under the jurisdiction of ASTM Committee F04 on
Medical and Surgical Materials and Devices and are the direct responsibility of
Subcommittee F04.25 on Spinal Devices. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
CurrenteditionapprovedJuly1,2022.PublishedJuly2022.Originallyapproved contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
in 2000. Last previous edition approved in 2018 as F2077–18. DOI: 10.1520/ Standards volume information, refer to the standard’s Document Summary page on
F2077-22. theASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2077 − 22
negative Z direction. Torsional force is defined to be the intervertebral body fusion device assembly in which all of the
component of moment parallel to the Z axis. tested constructs have withstood 5000000 cycles without
functional or mechanical failure.
3.2.2 crack, n—an externally visible physical discontinuity
in the form of a narrow opening that arises from mechanical 3.2.12 mechanical failure, n—that associated with the onset
forces.
of a new defect in the material (that is, initiation of fatigue
crack).
3.2.3 fatigue life, n—the number of cycles, N, that the
intervertebral body fusion device assembly can sustain at a
3.2.13 offset angular displacement, n—(Distance OB—Fig.
particular force or moment before mechanical or functional 6)—offset on the angular displacement axis equal to 10% of
failure occurs.
the intradiscal height, H, divided by the outside diameter or
height of the implant (maximum dimension of implant in XZ
3.2.4 functional failure, n—permanentdeformationthatren-
plane if not cylindrical) (for example, for a 10-mm intradiscal
ders the intervertebral body fusion device assembly ineffective
height and 16-mm intervertebral body fusion device assembly,
orunabletoresistforceand/ormaintainattachmentadequately.
distance OB = 10 mm/16 mm (0.10)(180°)/π = 3.6°).
3.2.5 ideal insertion location, n—the implant location with
3.2.14 offset displacement, n—(Distance OB—Fig. 6)—
respect to the simulated inferior and superior vertebral bodies
offset on the displacement axis equal to 2% of the intradiscal
(polyacetal or metal blocks) dictated by the type, design, and
height (that is, 0.2 mm for a 10-mm intradiscal height).
manufacturer’s surgical installation instructions.
3.2.15 permanent deformation, n—the displacement (mm)
3.2.6 intended method of application, n—intervertebral
or angular displacement (degree of the subassembly relative to
body fusion device assemblies may contain different types of
the unloaded condition) remaining after the applied load
stabilizing anchors such as threads, spikes, and knurled sur-
moment or torque has been removed. Care must be taken to
faces. Each type of anchor has an intended method of appli-
ensure that the loading fixtures are rigid and do not signifi-
cation or attachment to the spine.
cantly contribute to the measurement of deflection.
3.2.7 intended spinal location, n—the anatomic region of
3.2.16 stiffness (N/mm or N*mm/Degree (Radian)) (The
the spine intended for the intervertebral body fusion device
Slope of Line OG—Fig. 6), n—the slope of the initial linear
assembly.Intervertebralbodyfusiondeviceassembliesmaybe
portion of the force-displacement curve or the slope of the
designed and developed for specific regions of the spine such
initial linear portion of the moment—angular displacement
as the lumbar, thoracic, and cervical spine. Also, there exists
curve.
different anatomical potential surgical approaches, which will
result in different implant orientation at different levels of the
3.2.17 test block, n—thecomponentofthetestapparatusfor
spine.
mounting the intervertebral body fusion device assembly for
the intended test configuration.
3.2.8 intervertebral body fusion device, n—a structure (bio-
logic or synthetic) that is placed in the disc space between two
3.2.18 ultimate displacement (mm or degrees or radians)
adjacent vertebral bodies to provide support for eventual
(Displacement OF—Fig. 6), n—the displacement associated
arthrodesis of the two adjacent vertebral bodies.
with the ultimate force or ultimate moment.
3.2.9 intradiscal height, n—the straight-line distance along
3.2.19 ultimate force or moment (N or N*mm) (Point
the Z axis between the unaltered simulated vertebral bodies.
E—Fig. 6), n—the maximum applied force, F, transmitted by
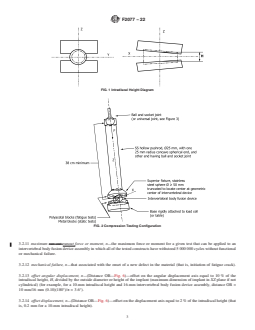
See Fig. 1.
thepushrod(assumedequaltoforcecomponentparalleltoand
3.2.10 force point, n—the point through which the resultant indicated by load cell), or the applied moment about the Z axis
force on the intervertebral device passes (that is, the geometric that can be applied to an intervertebral body fusion device
center of the superior fixture’s sphere) (Figs. 2-5). assembly.
3.2.11 maximum runout force or moment, n—the maximum 3.2.20 yield displacement (Distance OA—Fig. 6), n—the
force or moment for a given test that can be applied to an displacement (mm) or angular displacement (deg) when an
FIG. 1 Intradiscal Height Diagram
F2077 − 22
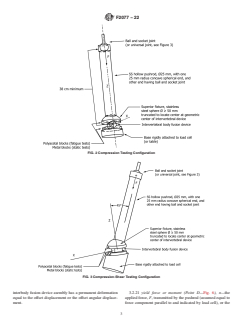
FIG. 2 Compression Testing Configuration
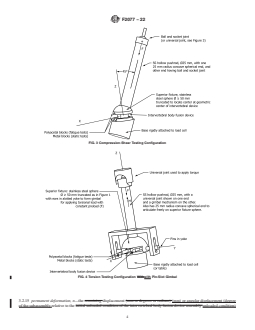
FIG. 3 Compression-Shear Testing Configuration
interbody fusion device asembly has a permanent deformation 3.2.21 yield force or moment (Point D—Fig. 6), n—the
equal to the offset displacement or the offset angular displace- applied force, F, transmitted by the pushrod (assumed equal to
ment. force component parallel to and indicated by load cell), or the
F2077 − 22
FIG. 4 Torsion Testing Configuration with Pin-Slot Gimbal
FIG. 5 Spherical Gimbal (Cross Section) for Torsion Testing Apparatus
applied moment about the Z axis required to produce a 4.2 Fatigue testing of the intervertebral body fusion device
permanent deformation equal to the offset displacement or the assemblies will simulate a motion segment via a gap between
offset angular displacement.
two polyacetal test blocks. The polyacetal will eliminate the
effectsofthevariabilityofbonepropertiesandmorphologyfor
4. Summary of Test Method
the fatigue tests. The minimum ultimate tensile strength of the
4.1 These test methods are proposed for the mechanical
polyacetal blocks shall be no less than 61 MPa.
testingofintervertebralbodyfusiondeviceassembliesspecific
to the lumbar, thoracic, and cervical spine.
F2077 − 22
FIG. 6 Typical Force Displacement Curve
4.3 Static testing of the intervertebral body fusion device body fusion device assemblies. These tests are conducted in
assemblies will simulate a motion segment via a gap between vitro to allow for analysis and comparison of the mechanical
two stainless steel blocks. The minimum ultimate tensile performanceofintervertebralbodyfusiondeviceassembliesto
strength of the blocks shall be no less than 1310 MPa. specific force modalities.
4.4 The pushrod shall also be manufactured from stainless
5.3 The forces applied to the intervertebral body fusion
steel, which shall also have a minimum ultimate tensile assemblies may differ from the complex loading seen in vivo,
strength no less than 1310 MPa. and therefore, the results from these tests may not directly
predict in vivo performance.The results, however, can be used
4.5 Static and dynamic tests will evaluate the intervertebral
to compare mechanical performance of different intervertebral
bodyfusiondeviceassembly.Theuserofthistestmethodmust
body fusion device assemblies.
decide which series of tests are applicable to the intervertebral
body fusion device assembly in question. The user of this test
5.4 Since the environment may affect the dynamic perfor-
method may choose to use all or a selection of the tests mance of intervertebral body fusion device assemblies, dy-
described in this test method for testing a particular interver-
namic testing in a saline environment may be considered.
tebral body fusion device assembly.
Fatigue tests should first be conducted in air (at ambient
temperature) for comparison purposes since the environmental
5. Significance and Use
effects could be significant. If a simulated in vivo environment
is desired, the investigator should consider testing in a saline
5.1 Intervertebral body fusion device assemblies are gener-
ally simple geometric-shaped devices which are often porous environmental bath at 37°C (for example, 0.9 g NaCl per
100mLwater)atarateof1Hzorless.Asimulatedbodyfluid,
or hollow in nature. Their function is to support the anterior
column of the spine to facilitate arthrodesis of the motion asalinedripormist,distilledwater,orothertypeoflubrication
at 37°C could also be used with adequate justification.
segment. This test method outlines materials and methods for
the characterization and evaluation of the mechanical perfor-
5.5 If the devices are known to be temperature and envi-
manceofdifferentintervertebralbodyfusiondeviceassemblies
ronmentdependent,testingshouldbeconductedinphysiologic
so that comparisons can be made between different designs.
solution as described in 5.4. Devices that require physiologic
5.2 This test method is designed to quantify the static and solution for testing should be tested in the same type solution
dynamic characteristics of different designs of intervertebral for comparison purposes.
F2077 − 22
5.6 The location within the simulated vertebral bodies and 6.4 Compression-Shear Testing Apparatus—The
positionoftheintervertebralbodyfusiondeviceassemblywith compression-sheartestapparatus(Fig.3),withexceptionofthe
respect to the loading axis will be dependent upon the design, inferior fixture, is identical to the axial compression apparatus
themanufacturer’srecommendation,orthesurgeon’spreferred (Fig. 2). The inferior fixture is to be designed to orient the
method for implant placement. initialpositionoftheintervertebraldevice’s Zaxisateither45°
or 27° flexion relative to the pushrod’s axis. The resultant
5.7 It is well known that the failure of materials is depen-
force, F, being applied to the intervertebral body fusion device
dent upon stress, test frequency, surface treatments, and envi-
assembly passes through the center of the superior fixture’s
ronmental factors. Therefore, when determining the effect of
spherical surface and is coincident with the pushrod’s axis.
changing one of these parameters (for example, frequency,
Thus, a combined compressive force F and an anterior shear
z
material, or environment), all others must be kept constant to
force F is created, which initially are either equal in magni-
x
facilitate interpretation of the results.
tude or F is twice that of F and passes through the geometric
z x
center of the intervertebral body fusion device assembly.
6. Apparatus
NOTE 1—Benfanti and colleagues measured the L5-S1 angle in 14
healthy volunteers in a standing position and obtained an average of 16.1
6.1 Test machines will conform to the requirements of
6 3.3°. Assuming a normal distribution, an angle of greater than the
Practices E4.
average +3σ would represent greater than 99.7% of the population. 16.1
6 9.9° = 26°; however, using an angle of 26.6°, rounded to 27°, is
6.2 The intradiscal height, H, shall be determined from
convenient as the normal force is twice that of the shear force.
vertebral body and disc morphometric data at the intended
6.5 Torsion Testing Apparatus—The torsion test apparatus
level of application. Suggested heights are as follows: 10 mm
(Fig. 4) is similar to the axial compression test apparatus (Fig.
forthelumbarspine,6mmforthethoracicspine,and4mmfor
2)withexceptionofthepushrodinterconnections.Theactuator
the cervical spine.The intradiscal height should not reach zero
of the testing machine must be connected to the pushrod by a
before the onset of functional or mechanical failure. If this
minimal friction (that is, unconstrained in bending) universal
occurs, the test is considered a failure. The user of the test
joint to be able to transmit torsional moment in addition to
method should select the intradiscal height that is appropriate
axialforce.Thepushrodisconnectedtothesuperiorfixtureby
for the device being tested.
a spherical gimbal mechanism to apply combined compressive
6.3 Axial Compression Test Apparatus—The actuator of the
force, F, and moment, M, with negligible bending moment to
testing machine is connected to the pushrod by a minimal
the intervertebral body fusion device assembly. Two examples
friction ball-and-socket joint or universal joint (that is, uncon-
of a gimbal mechanism are: (1) a sphere with pegs engaged in
strained in bending). The pushrod is connected to the superior
a slotted yoke attached to the pushrod (Fig. 4), and (2) a pair
fixturebyaminimalfrictionspherejoint(thatis,unconstrained
of spherical surfaces with interdigitating tongue and grooves
in bending and torsion). The hollow pushrod should be of
located 90° to each other (Fig. 5). The test apparatus is to be
minimal weight so as to be considered a “two-force” member.
assembled so that the Z axis of the intervertebral body fusion
It thus applies to the intervertebral body fusion device assem-
device assembly is initially coincident with the pushrod’s axis
bly a resultant force directed along the pushrod’s axis and
andcollinearwiththeaxisofthetestingmachine’sactuatorand
located at the center of the superior fixture’s sphere joint (the
load cell. This setup is designed so that the initially applied
geometric center of the device being tested). For the fatigue
force, F, and moment, M, are equal to the compression force,
tests, the device is placed between two polyacetal blocks,
F, and torsional moment, M, on the intervertebral body fusion
which are rigidly attached to the metal blocks (Fig. 2). For the
device assembly.
static tests, metal blocks are to be used, which could be
6.6 The geometry of the polyacetal or metal block shall be
incorporated as an integral part of the superior and inferior
determined and justified by the user of these test methods. It
fixtures. The blocks are to have surfaces that mate geometri-
may be necessary to machine geometry of the blocks to match
callywiththeintervertebraldevicesimilartohowthedeviceis
that of the implants to maintain stability during testing. In this
intended to mate with vertebral end plates. The test apparatus
situation,itisrecommendedthatthemachinedpocketdepthin
will be assembled such that the Z axis of the intervertebral
theblockshallbenomorethan3mmatthedeepestpoint,and
device is initially coincident with the pushrod’s axis and
the intradiscal height shall leave no less than 50% of the
collinear with the axis of the testing machine’s actuator and
device exposed (see X1.11). Any deviations from this recom-
load cell. The length of the pushrod between the center of the
mendation should be justified, that is, extremely tall or ex-
ball-and-socket joint to the center of the spherical surface is to
tremely short devices.
beaminimumof38cm.Thisisrequiredtominimizedeviation
of the pushrod’s axis (direction of applied force, F) from that
7. Sampling
of the test machine’s load cell axis. In other words, this is to
7.1 All components in the intervertebral body fusion device
minimize the error in using and reporting that the force
assembly shall be previously unused parts only; no implants
indicated by the load cell “F ” is the applied force, F, and is
ind
shall be retested.
equal to the compressive force, F , on the intervertebral body
z
fusion device assembly. For example, a 1-mm displacement of
Benfanti P. L., Geissele, A. E., “The effect of intraoperative hip position on
the spherical surfaces center in the XY plane would produce an
maintenance of lumbar lordosis: a radiographic study of anesthetized patients and
anglebetweenaxesof0.15°,(10mmproducing1.5°).Fig.2is
unanesthetized volunteers on the Wilson frame,” Spine, Vol 22, No. 19, 1997, pp.
a schematic of this test setup. 2299–2303.
F2077 − 22
7.2 Each pair of polyacetal blocks shall be used for one test 8.2 The force, F, and moment, M , are to be applied as
z
only. Metal blocks may be reused if undamaged. describedinSection6ofthistestmethodinpositioncontrolat
a rate no greater than 25mm/min or 60°/min (radians/minute)
7.3 The test assemblies (that is, intervertebral body fusion
untilfunctionalormechanicalfailureoftheintervertebralbody
device assembly and polyacetal blocks) shall be labeled and
fusion device assembly is obtained.
shall be maintained according to good laboratory practice.The
test assembly can be disassembled to facilitate examination of 8.3 Physiological compressive preloads of 100, 300, and
surface conditions. 500 N for cervical, thoracic, and lumbar intervertebral body
fusion device assemblies, respectively, are required for the
7.4 All static tests should have a minimum of five test
static torsion test to avoid separation of the blocks during
samples.
testing. Other loads may be used with adequate justification.
7.5 The user of this test method should select the necessary
8.4 The force displacement curve shall be recorded. The
fatigue loads to establish the endurance limit of the interver-
yield displacement (mm or degrees or radians), stiffness
tebral body fusion device assembly and develop a curve of
(N/mm or N*mm/degree (radian)), yield force or moment (N
cyclical compression force or torsional moment versus the
or N*mm), ultimate displacement (mm or degrees or radians),
numberofcyclestofailure.Thistrendshallbecomprisedofat
and ultimate force or moment (N or N*mm) are to be
least six data points. Initial fatigue loading conditions may be
established. The user may reference Practices E2309 for
determined primarily by the user’s experience. In the absence
assistance in static test yield determination.
of such experience, initial fatigue loads corresponding to 75,
50,and/or25%oftheyieldloadasdeterminedinthestatictest
9. Procedure for Dynamic Tests
may serve as a starting point for establishing the fatigue
characteristics. The precision of the endurance limit shall be 9.1 An intervertebral body fusion device assembly is to be
inserted between two prepared polyacetal blocks having the
established by ensuring that the lowest load that results in a
failed construct is not greater than 1.5× the highest established appropriate matching geometry of the intervertebral body
fusiondeviceassembly(Fig.7).Theintradiscalheight, H,shall
runoutload.Forexample,ifthehighestestablishedrunoutload
is100.0Nthenthelowestloadthatresultsinafailedconstruct beconstantf
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F2077 − 18 F2077 − 22
Standard Test Methods Forfor
Intervertebral Body Fusion Devices
This standard is issued under the fixed designation F2077; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This test method covers the materials and methods for the static and dynamic testing of intervertebral body fusion device
assemblies, spinal implants designed to promote arthrodesis at a given spinal motion segment.
1.2 This test method is intended to provide a basis for the mechanical comparison among past, present, and future nonbiologic
intervertebral body fusion device assemblies. This test method allows comparison of intervertebral body fusion device assemblies
with different intended spinal locations and methods of application to the intradiscal spaces. This test method is intended to enable
the user to compare intervertebral body fusion device assemblies mechanically and does not purport to provide performance
standards for intervertebral body fusion device assemblies.
1.3 The test method describes static and dynamic tests by specifying force types and specific methods of applying these forces.
These tests are designed to allow for the comparative evaluation of intervertebral body fusion device assemblies.
1.4 These tests are designed to characterize the structural integrity of the device and are not intended to test the bone-implant
interface.
1.5 This test method does not address expulsion testing of intervertebral body fusion device assemblies (see 1.4).
1.6 Guidelines are established for measuring displacements, determining the yield force or moment, and evaluating the
stiffness,stiffness and strength of the intervertebral body fusion device assemblies.
1.7 Some intervertebral body fusion device assemblies may not be testable in all test configurations.
1.8 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard, with
the exception of angular measurements, which may be reported in terms of either degrees or radians.
1.9 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.10 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ThisThese test method ismethods are under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and isare the direct responsibility
of Subcommittee F04.25 on Spinal Devices.
Current edition approved June 1, 2018July 1, 2022. Published August 2018July 2022. Originally publishedapproved in 2000. Last previous edition approved in 20172018
as F2077 – 17.F2077 – 18. DOI: 10.1520/F2077-18.10.1520/F2077-22.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2077 − 22
2. Referenced Documents
2.1 ASTM Standards:
E4 Practices for Force Calibration and Verification of Testing Machines
E6 Terminology Relating to Methods of Mechanical Testing
E177 Practice for Use of the Terms Precision and Bias in ASTM Test Methods
E691 Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
E1823 Terminology Relating to Fatigue and Fracture Testing
E2309 Practices for Verification of Displacement Measuring Systems and Devices Used in Material Testing Machines
F1582 Terminology Relating to Spinal Implants
3. Terminology
3.1 For definition of terms refer to TerminologyTerminologies E6, E1823, and F1582.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 coordinate system/axes, n—Threethree orthogonal axes are defined by Terminologyas follows. F1582. The center of the
coordinate system is located at the geometric center of the intervertebral body fusion device assembly. The XY plane is to bisect
the sagittal plane angle between superior and inferior lines (surfaces) that are intended to simulate the adjacent vertebral end plates.
The positive Z axis is to be directed superiorly. Force components parallel to the XY plane are shear components of loading. The
compressive axial force is defined to be the component in the negative Z direction. Torsional force is defined to be the component
of moment parallel to the Z axis.
3.2.2 crack, n—an externally visible physical discontinuity in the form of a narrow opening that arises from mechanical forces.
3.2.3 fatigue life, n—the number of cycles, N, that the intervertebral body fusion device assembly can sustain at a particular force
or moment before mechanical or functional failure occurs.
3.2.4 functional failure, n—permanent deformation that renders the intervertebral body fusion device assembly ineffective or
unable to resist force and/or maintain attachment adequately.
3.2.5 ideal insertion location, n—the implant location with respect to the simulated inferior and superior vertebral bodies
(polyacetal or metal blocks) dictated by the type, design, and manufacturer’s surgical installation instructions.
3.2.6 intended method of application, n—intervertebral body fusion device assemblies may contain different types of stabilizing
anchors such as threads, spikes, and knurled surfaces. Each type of anchor has an intended method of application or attachment
to the spine.
3.2.7 intended spinal location, n—the anatomic region of the spine intended for the intervertebral body fusion device assembly.
Intervertebral body fusion device assemblies may be designed and developed for specific regions of the spine such as the lumbar,
thoracic, and cervical spine. Also, there exists different anatomical potential surgical approaches, which will result in different
implant orientation at different levels of the spine.
3.2.8 intervertebral body fusion device, n—a structure (biologic or synthetic) that is placed in the disc space between two adjacent
vertebral bodies to provide support for eventual arthrodesis of the two adjacent vertebral bodies.
3.2.9 intradiscal height, n—the straight-line distance along the Z axis between the unaltered simulated vertebral bodies—
minimum height of 4 mm and a maximum height of 18 mm. bodies. See Fig. 1.
3.2.10 force point, n—the point through which the resultant force on the intervertebral device passes (that is, the geometric center
of the superior fixture’s sphere) (Figs. 2-5).
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
F2077 − 22
FIG. 1 Intradiscal Height Diagram
FIG. 2 Compression Testing Configuration
3.2.11 maximum run out runout force or moment, n—the maximum force or moment for a given test that can be applied to an
intervertebral body fusion device assembly in which all of the tested constructs have withstood 5 000 000 cycles without functional
or mechanical failure.
3.2.12 mechanical failure, n—that associated with the onset of a new defect in the material (that is, initiation of fatigue crack).
3.2.13 offset angular displacement, n—(Distance OB—Fig. 6)—offset on the angular displacement axis equal to 10 % of the
intradiscal height, H, divided by the outside diameter or height of the implant (maximum dimension of implant in XZ plane if not
cylindrical) (for example, for a 10-mm intradiscal height and 16-mm intervertebral body fusion device assembly, distance OB =
10 mm/16 mm (0.10)(180°)/π = 3.6°).
3.2.14 offset displacement, n—(Distance OB—Fig. 6)—offset on the displacement axis equal to 2 % of the intradiscal height (that
is, 0.2 mm for a 10-mm intradiscal height).
F2077 − 22
FIG. 3 Compression-Shear Testing Configuration
FIG. 4 Torsion Testing Configuration Withwith Pin-Slot Gimbal
3.2.15 permanent deformation, n—the remaining displacement (mm or degrees or radians) (mm) or angular displacement (degree
of the subassembly relative to the initial unloaded condition of the intervertebral body fusion device assembly unloaded condition)
F2077 − 22
FIG. 5 Spherical Gimbal (Cross Section) for Torsion Testing Apparatus
FIG. 6 Typical Force Displacement Curve
remaining after the applied force load moment or torque has been removed. Care must be taken to ensure that the loading fixtures
are rigid and do not significantly contribute to the measurement of deflection.
F2077 − 22
3.2.16 stiffness (N/mm or N*mm/Degree (Radian)) (The Slope of Line OG—Fig. 6),n—the slope of the initial linear portion of the
force-displacement curve or the slope of the initial linear portion of the moment—angular displacement curve.
3.2.17 test block, n—the component of the test apparatus for mounting the intervertebral body fusion device assembly for the
intended test configuration.
3.2.18 ultimate displacement (mm or degrees or radians) (Displacement OF—Fig. 6),n—the displacement associated with the
ultimate force or ultimate moment.
3.2.19 ultimate force or moment (N or N*mm) (Point E—Fig. 6),n—the maximum applied force, F, transmitted by the pushrod
(assumed equal to force component parallel to and indicated by load cell), or the applied moment about the Z axis that can be
applied to an intervertebral body fusion device assembly.
3.2.20 yield displacement (Distance OA—Fig. 6),n—the displacement (mm) or angular displacement (deg) when an interbody
fusion device asembly has a permanent deformation equal to the offset displacement or the offset angular displacement.
3.2.21 yield force or moment (Point D—Fig. 6),n—the applied force, F, transmitted by the pushrod (assumed equal to force
component parallel to and indicated by load cell), or the applied moment about the Z axis required to produce a permanent
deformation equal to the offset displacement or the offset angular displacement.
4. Summary of Test Method
4.1 These test methods are proposed for the mechanical testing of intervertebral body fusion device assemblies specific to the
lumbar, thoracic, and cervical spine.
4.2 Fatigue testing of the intervertebral body fusion device assemblies will simulate a motion segment via a gap between two
polyacetal test blocks. The polyacetal will eliminate the effects of the variability of bone properties and morphology for the fatigue
tests. The minimum ultimate tensile strength of the polyacetal blocks shall be no less than 61 MPa.
4.3 Static testing of the intervertebral body fusion device assemblies will simulate a motion segment via a gap between two
stainless steel blocks. The minimum ultimate tensile strength of the blocks shall be no less than 1310 MPa.
4.4 The pushrod shall also be manufactured from stainless steel, which shall also have a minimum ultimate tensile strength no less
than 1310 MPa.
4.5 Static and dynamic tests will evaluate the intervertebral body fusion device assembly. The user of this test method must decide
which series of tests are applicable to the intervertebral body fusion device assembly in question. The user of this test method may
choose to use all or a selection of the tests described in this test method for testing a particular intervertebral body fusion device
assembly.
5. Significance and Use
5.1 Intervertebral body fusion device assemblies are generally simple geometric-shaped devices which are often porous or hollow
in nature. Their function is to support the anterior column of the spine to facilitate arthrodesis of the motion segment. This test
method outlines materials and methods for the characterization and evaluation of the mechanical performance of different
intervertebral body fusion device assemblies so that comparisons can be made between different designs.
5.2 This test method is designed to quantify the static and dynamic characteristics of different designs of intervertebral body fusion
device assemblies. These tests are conducted in vitro to allow for analysis and comparison of the mechanical performance of
intervertebral body fusion device assemblies to specific force modalities.
5.3 The forces applied to the intervertebral body fusion assemblies may differ from the complex loading seen in vivo, and
therefore, the results from these tests may not directly predict in vivo performance. The results, however, can be used to compare
mechanical performance of different intervertebral body fusion device assemblies.
F2077 − 22
5.4 Since the environment may affect the dynamic performance of intervertebral body fusion device assemblies, dynamic testing
in a saline environment may be considered. Fatigue tests should first be conducted in air (at ambient temperature) for comparison
purposes since the environmental effects could be significant. If a simulated in vivo environment is desired, the investigator should
consider testing in a saline environmental bath at 37°C37 °C (for example, 0.9-g 0.9 g NaCl per 100-mL100 mL water) at a rate
of 1 Hz or less. A simulated body fluid, a saline drip or mist, distilled water, or other type of lubrication at 37°C37 °C could also
be used with adequate justification.
5.5 If the devices are known to be temperature and environment dependent, testing should be conducted in physiologic solution
as described in 5.4. Devices that require physiologic solution for testing should be tested in the same type solution for comparison
purposes.
5.6 The location within the simulated vertebral bodies and position of the intervertebral body fusion device assembly with respect
to the loading axis will be dependent upon the design, the manufacturer’s recommendation, or the surgeon’s preferred method for
implant placement.
5.7 It is well known that the failure of materials is dependent upon stress, test frequency, surface treatments, and environmental
factors. Therefore, when determining the effect of changing one of these parameters (for example, frequency, material, or
environment), all others must be kept constant to facilitate interpretation of the results.
6. Apparatus
6.1 Test machines will conform to the requirements of Practices E4.
6.2 The intradiscal height, H, shall be determined from vertebral body and disc morphometric data at the intended level of
application. Suggested heights are as follows: 10 mm for the lumbar spine, 6 mm for the thoracic spine, and 4 mm for the cervical
spine. The intradiscal height should not reach zero before the onset of functional or mechanical failure. If this occurs, the test is
considered a failure. The user of the test method should select the intradiscal height that is appropriate for the device being tested.
6.3 Axial Compression Test Apparatus—The actuator of the testing machine is connected to the pushrod by a minimal friction ball
and socket ball-and-socket joint or universal joint (that is, unconstrained in bending). The pushrod is connected to the superior
fixture by a minimal friction sphere joint (that is, unconstrained in bending and torsion). The hollow pushrod should be of minimal
weight so as to be considered a “two-force” member. It thus applies to the intervertebral body fusion device assembly a resultant
force directed along the pushrod’s axis and located at the center of the superior fixture’s sphere joint (the geometric center of the
device being tested). For the fatigue tests, the device is placed between two polyacetal blocks, which are rigidly attached to the
metal blocks (Fig. 2). For the static tests, metal blocks are to be used, which could be incorporated as an integral part of the superior
and inferior fixtures. The blocks are to have surfaces that mate geometrically with the intervertebral device similar to how the
device is intended to mate with vertebral end plates. The test apparatus will be assembled such that the Z axis of the intervertebral
device is initially coincident with the pushrod’s axis and collinear with the axis of the testing machine’s actuator and load cell. The
length of the pushrod between the center of the ball-and-socket joint to the center of the spherical surface is to be a minimum of
38 cm. This is required to minimize deviation of the pushrod’s axis (direction of applied force, F) from that of the test machine’s
load cell axis. In other words, this is to minimize the error in using and reporting that the force indicated by the load cell “F ”
ind
is the applied force, F, and is equal to the compressive force, F , on the intervertebral body fusion device assembly. For example,
z
a 1-mm displacement of the spherical surfaces center in the XY plane would produce an angle between axes of 0.15°, (10 mm
producing 1.5°). Fig. 2 is a schematic of this test set-up.setup.
6.4 Compression-Shear Testing Apparatus—The compression-shear test apparatus (Fig. 3), with exception of the inferior fixture,
is identical to the axial compression apparatus (Fig. 2). The inferior fixture is to be designed to orient the initial position of the
intervertebral device’s Z axis at either 45° or 27° flexion relative to the pushrod’s axis. The resultant force, F, being applied to
the intervertebral body fusion device assembly passes through the center of the superior fixture’s spherical surface and is coincident
with the pushrod’s axis. Thus, a combined compressive force F and an anterior shear force F is created, which initially are either
z x
equal in magnitude or F is twice that of F and passes through the geometric center of the intervertebral body fusion device
z x
assembly.
Benfanti P. L., Geissele, A. E., “The effect of intraoperative hip position on maintenance of lumbar lordosis: a radiographic study of anesthetized patients and
unanesthetized volunteers on the Wilson frame,” Spine, Vol 22, No. 19, 1997, pp. 2299–2303.
F2077 − 22
NOTE 1—Benfanti and colleagues measured the L5-S1 angle in 14 healthy volunteers in a standing position and obtained an average of 16.1 6 3.3°.
Assuming a normal distribution, an angle of greater than the average +3σ would represent greater than 99.7 % of the population. 16.1 6 9.9° = 26°;
however, using an angle of 26.6°, rounded to 27°, is convenient as the normal force is twice that of the shear force.
6.5 Torsion Testing Apparatus—The torsion test apparatus (Fig. 4) is similar to the axial compression test apparatus (Fig. 2) with
exception of the pushrod interconnections. The actuator of the testing machine must be connected to the pushrod by a minimal
friction (that is, unconstrained in bending) universal joint to be able to transmit torsional moment in addition to axial force. The
pushrod is connected to the superior fixture by a spherical gimbal mechanism to apply combined compressive force, F, and
moment, M, with negligible bending moment to the intervertebral body fusion device assembly. Two examples of a gimbal
mechanism are: (1) a sphere with pegs engaged in a slotted yoke attached to the pushrod (Fig. 4)), and (2) a pair of spherical
surfaces with interdigitating tongue and grooves located 90° to each other (Fig. 5). The test apparatus is to be assembled so that
the Z axis of the intervertebral body fusion device assembly is initially coincident with the pushrod’s axis and collinear with the
axis of the testing machine’s actuator and load cell. This setup is designed so that the initially applied force, F, and moment, M,
are equal to the compression force, F, and torsional moment, M, on the intervertebral body fusion device assembly.
6.6 The geometry of the polyacetal or metal block shall be determined and justified by the user of these test methods. It may be
necessary to machine geometry of the blocks to match that of the implants to maintain stability during testing. In this situation,
it is recommended that the machined pocket depth in the block shall be no more than 3 mm at the deepest point, and the intradiscal
height shall leave no less than 50 % of the device exposed. (Seeexposed (see X1.12X1.11). Any deviations from this
recommendation should be justified, that is, extremely tall or extremely short devices.
7. Sampling
7.1 All components in the intervertebral body fusion device assembly shall be previously unused parts only; no implants shall be
retested.
7.2 Each pair of polyacetal blocks shall be used for one test only. Metal blocks may be reused if undamaged.
7.3 The test assemblies (that is, intervertebral body fusion device assembly and polyacetal blocks) shall be labeled and shall be
maintained according to good laboratory practice. The test assembly can be disassembled to facilitate examination of surface
conditions.
7.4 All static tests should have a minimum of five test samples.
7.5 The user of this test method should select the necessary fatigue loads to establish the endurance limit of the intervertebral body
fusion device assembly and develop a curve of cyclical compression force or torsional moment versus the number of cycles to
failure. This trend shall be comprised of at least six data points. Initial fatigue loading conditions may be determined primarily by
the user’s experience. In the absence of such experience, initial fatigue loads corresponding to 75, 50, and/or 25 % of the yield
load as determined in the static test may serve as a starting point for establishing the fatigue characteristics. The precision of the
endurance limit shall be established by ensuring that the lowest load that results in a failed construct is not greater than 1.5× the
highest established run-outrunout load. For example, if the highest established run-outrunout load is 100.0 N then the lowest load
that results in a failed construct shall not be greater than 150.0 N. A regression analysis shall be conducted on the force or moment
versus number of cycles to failure data.
8. Procedure for Static Tests
8.1 The intervertebral body fusion device assembly is to be inserted between two prepared metal blocks having the appropriate
matching geometry of the intervertebral body fusion device assembly (Fig. 7). The intradiscal height, H, shall be constant for all
tests for an intervertebral body fusion device assembly of a given size.
8.2 The force, F, and moment, M , are to be applied as described in Section 6 of this test method in position control at a rate no
z
greater than 25mm/min or 60°/min (radians/minute) until functional or mechanical failure of the intervertebral body fusion device
assembly is obtained.
F2077 − 22
FIG. 7 Polyacetal or Metal Test Block
8.3 Physiological compressive preloads of 100, 300, and 500 N for cervical, thoracic, and lumbar intervertebral body fusion device
assemblies, respectively, are required for the static torsion test to avoid separation of the blocks during testing. Other loads may
be used with adequate justification.
8.4 The force displacement curve shall be recorded. The yield displacement (mm or degrees or radians), stiffness (N/mm or
N*mm/degree (radian)), yield force or moment (N or N*mm), ultimate displacement (mm or degrees or radians), and ultimate
force or moment (N or N*mm) are to be established. The user may reference Practices E2309 for assistance in static test yi
...





Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...