ASTM C1702-23
(Test Method)Standard Test Method for Measurement of Heat of Hydration of Hydraulic Cementitious Materials Using Isothermal Conduction Calorimetry
Standard Test Method for Measurement of Heat of Hydration of Hydraulic Cementitious Materials Using Isothermal Conduction Calorimetry
SIGNIFICANCE AND USE
5.1 This method is suitable for determining the total heat of hydration of hydraulic cement at constant temperature at ages up to 7 days to confirm specification compliance.
5.2 This method compliments Practice C1679 by providing details of calorimeter equipment, calibration, and operation. Practice C1679 emphasizes interpretation significant events in cement hydration by analysis of time dependent patterns of heat flow, but does not provide the level of detail necessary to give precision test results at specific test ages required for specification compliance.
SCOPE
1.1 This test method specifies the apparatus and procedure for determining total heat of hydration of hydraulic cementitious materials at test ages up to 7 days by isothermal conduction calorimetry.
1.2 This test method also outputs data on rate of heat of hydration versus time that is useful for other analytical purposes, as covered in Practice C1679.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Jul-2023
- Technical Committee
- C01 - Cement
- Drafting Committee
- C01.26 - Heat of Hydration
Relations
- Replaces
ASTM C186-17 - Standard Test Method for Heat of Hydration of Hydraulic Cement (Withdrawn 2019) - Effective Date
- 15-Oct-2019
- Effective Date
- 01-Jun-2014
- Effective Date
- 15-Dec-2013
- Effective Date
- 01-May-2013
- Effective Date
- 01-Nov-2011
- Effective Date
- 15-Jun-2009
- Effective Date
- 01-Oct-2008
- Effective Date
- 01-Jun-2008
- Effective Date
- 01-Nov-2007
- Effective Date
- 01-Nov-2005
- Effective Date
- 10-May-1999
Overview
ASTM C1702-23, titled Standard Test Method for Measurement of Heat of Hydration of Hydraulic Cementitious Materials Using Isothermal Conduction Calorimetry, establishes procedures for measuring the total heat of hydration released by hydraulic cementitious materials. This standard is developed by ASTM International and is widely referenced in the construction and cement industries for ensuring material compliance and performance validation.
This method is suitable for quantifying the heat of hydration at constant temperature for test periods up to 7 days. The results help assess the compliance of cementitious materials with technical specifications and provide valuable insight into the hydration process. ASTM C1702-23 supplements ASTM Practice C1679 by providing detailed guidance on equipment, calibration, and operation of isothermal conduction calorimeters.
Key Topics
- Isothermal Conduction Calorimetry: Provides requirements for the use of isothermal calorimeters that measure heat flow at a constant temperature, facilitating accurate determination of heat of hydration in cement-based materials.
- Test Age: Specifies test ages up to 7 days, allowing evaluation of short- and medium-term hydration characteristics.
- Calibration and Operation: Includes detailed procedures for instrument calibration, equipment verification, and operation protocols to ensure reliable and repeatable results.
- Data Output: Generates both total heat of hydration values and heat flow rate data versus time, supporting a variety of analytical and research needs.
- Result Reporting: Outlines the required information for comprehensive test reporting, emphasizing accuracy and traceability.
- Precision and Bias: Summarizes interlaboratory studies, defining single-operator and multilaboratory precision, and provides guidance for consistent application across different testing environments.
- SI Units: Mandates that all measurements and calculations be reported in SI units.
Applications
ASTM C1702-23 is critical for professionals in cement manufacturing, concrete technology, and construction quality assurance. The test method is used to:
- Confirm Specification Compliance: Verify that hydraulic cements meet contractual and regulatory requirements for total heat of hydration at specific ages.
- Quality Control: Monitor production consistency in cement plants and construction sites, reducing variability in concrete performance.
- Evaluate New Materials: Assess the behavior of alternative binders, blended cements, or supplementary cementitious materials under standardized conditions.
- Optimize Mix Design: Inform the development of mixes for reduced heat evolution in mass concrete applications, minimizing thermal cracking risks.
- Research and Development: Support scientific studies on hydration kinetics by providing detailed heat flow data for advanced modelling and interpretation.
Related Standards
Several ASTM standards are closely linked to the implementation and context of ASTM C1702-23:
- ASTM C1679: Practice for Measuring Hydration Kinetics of Hydraulic Cementitious Mixtures Using Isothermal Calorimetry (focuses on interpretation of heat flow patterns, complements C1702’s focus on precise test method).
- ASTM C186: Test Method for Heat of Hydration of Hydraulic Cement (withdrawn in 2019, formerly used for similar purposes).
- ASTM E691: Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method (referenced for evaluating method precision and repeatability).
Keywords: ASTM C1702-23, heat of hydration, hydraulic cement, isothermal conduction calorimetry, cement testing, cement specification compliance, calorimeter calibration, cementitious materials, portland cement, ASTM standards.
Buy Documents
ASTM C1702-23 - Standard Test Method for Measurement of Heat of Hydration of Hydraulic Cementitious Materials Using Isothermal Conduction Calorimetry
REDLINE ASTM C1702-23 - Standard Test Method for Measurement of Heat of Hydration of Hydraulic Cementitious Materials Using Isothermal Conduction Calorimetry
Get Certified
Connect with accredited certification bodies for this standard

BSMI (Bureau of Standards, Metrology and Inspection)
Taiwan's standards and inspection authority.
Sponsored listings
Frequently Asked Questions
ASTM C1702-23 is a standard published by ASTM International. Its full title is "Standard Test Method for Measurement of Heat of Hydration of Hydraulic Cementitious Materials Using Isothermal Conduction Calorimetry". This standard covers: SIGNIFICANCE AND USE 5.1 This method is suitable for determining the total heat of hydration of hydraulic cement at constant temperature at ages up to 7 days to confirm specification compliance. 5.2 This method compliments Practice C1679 by providing details of calorimeter equipment, calibration, and operation. Practice C1679 emphasizes interpretation significant events in cement hydration by analysis of time dependent patterns of heat flow, but does not provide the level of detail necessary to give precision test results at specific test ages required for specification compliance. SCOPE 1.1 This test method specifies the apparatus and procedure for determining total heat of hydration of hydraulic cementitious materials at test ages up to 7 days by isothermal conduction calorimetry. 1.2 This test method also outputs data on rate of heat of hydration versus time that is useful for other analytical purposes, as covered in Practice C1679. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 This method is suitable for determining the total heat of hydration of hydraulic cement at constant temperature at ages up to 7 days to confirm specification compliance. 5.2 This method compliments Practice C1679 by providing details of calorimeter equipment, calibration, and operation. Practice C1679 emphasizes interpretation significant events in cement hydration by analysis of time dependent patterns of heat flow, but does not provide the level of detail necessary to give precision test results at specific test ages required for specification compliance. SCOPE 1.1 This test method specifies the apparatus and procedure for determining total heat of hydration of hydraulic cementitious materials at test ages up to 7 days by isothermal conduction calorimetry. 1.2 This test method also outputs data on rate of heat of hydration versus time that is useful for other analytical purposes, as covered in Practice C1679. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM C1702-23 is classified under the following ICS (International Classification for Standards) categories: 17.200.10 - Heat. Calorimetry. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM C1702-23 has the following relationships with other standards: It is inter standard links to ASTM C186-17, ASTM C1679-14, ASTM C1679-13, ASTM E691-13, ASTM E691-11, ASTM C1679-09, ASTM E691-08, ASTM C1679-08, ASTM C1679-07, ASTM E691-05, ASTM E691-99. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM C1702-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: C1702 − 23
Standard Test Method for
Measurement of Heat of Hydration of Hydraulic
Cementitious Materials Using Isothermal Conduction
Calorimetry
This standard is issued under the fixed designation C1702; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 3. Terminology
3.1 Definitions of Terms Specific to This Standard:
1.1 This test method specifies the apparatus and procedure
for determining total heat of hydration of hydraulic cementi-
3.1.1 baseline, n—the time-series signal from the calorim-
tious materials at test ages up to 7 days by isothermal eter when measuring output from a sample of approximately
conduction calorimetry.
the same mass and thermal properties as a cement sample, but
which is not generating or consuming heat.
1.2 This test method also outputs data on rate of heat of
3.1.2 heat, n—the time integral of thermal power measured
hydration versus time that is useful for other analytical
purposes, as covered in Practice C1679. in joules (J).
3.1.3 isothermal conduction calorimeter, n—a calorimeter
1.3 The values stated in SI units are to be regarded as
that measures heat flow from a sample maintained at a constant
standard. No other units of measurement are included in this
temperature by intimate thermal contact with a constant
standard.
temperature heat sink.
1.4 This standard does not purport to address all of the
3.1.4 reference cell, n—a heat-flow measuring cell that is
safety concerns, if any, associated with its use. It is the
dedicated to measuring power from a sample that is generating
responsibility of the user of this standard to establish appro-
no heat.
priate safety, health, and environmental practices and deter-
mine the applicability of regulatory limitations prior to use. 3.1.4.1 Discussion—The purpose of the reference cell is to
1.5 This international standard was developed in accor- correct for baseline drift and other systematic errors that can
occur in heat-flow measuring equipment.
dance with internationally recognized principles on standard-
ization established in the Decision on Principles for the
3.1.5 sensitivity, n—the minimum change in thermal power
Development of International Standards, Guides and Recom-
reliably detectable by an isothermal calorimeter.
mendations issued by the World Trade Organization Technical
3.1.5.1 Discussion—For this application, sensitivity is taken
Barriers to Trade (TBT) Committee.
as ten times the random noise (standard deviation) in the
baseline signal.
2. Referenced Documents
3.1.6 thermal mass, n—the amount of thermal energy that
2.1 ASTM Standards:
can be stored by a material (J/K).
C1679 Practice for Measuring Hydration Kinetics of Hy-
3.1.6.1 Discussion—The thermal mass of a given material is
draulic Cementitious Mixtures Using Isothermal Calorim-
calculated by multiplying the mass by the specific heat capacity
etry
of the material. For the purpose of calculating the thermal mass
E691 Practice for Conducting an Interlaboratory Study to
used in this standard, the following specific heat capacities can
Determine the Precision of a Test Method
be used: The specific heat capacity of a typical unhydrated
portland cement and water is 0.75 and 4.18 J/(g·K), respec-
tively. Thus a mixture of A g of cement and B g of water has
a thermal mass of (0.75 × A + 4.18 × B) J/K. The specific heat
This test method is under the jurisdiction of ASTM Committee C01 on Cement
and is the direct responsibility of Subcommittee C01.26 on Heat of Hydration.
capacity of typical quartz and limestone is 0.75 and
Current edition approved Aug. 1, 2023. Published August 2023. Originally
0.84 J ⁄(g·K), respectively. The specific heat capacity of most
approved in 2009. Last previous edition approved in 2017 as C1702 – 17. DOI:
amorphous supplementary cementitious material, such as fly
10.1520/C1702-23.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or ash or slag, is approximately 0.8 J/(g·K).
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
3.1.7 thermal power, n—the heat production rate measured
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website. in joules per second (J/s).
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
C1702 − 23
3.1.7.1 Discussion—This is the property measured by the 6. Apparatus
calorimeter. The thermal power unit of measure is J/s, which is
6.1 Miscellaneous Equipment:
equivalent to the watt. The watt is also a common unit of
6.1.1 Balance—Accurate to 0.01 g.
measure used to represent thermal power.
6.1.2 Volumetric Dispenser—A device for measuring vol-
ume or mass of water, accurate to 0.1 mL. This could be a
4. Summary of Test Method
syringe, pipette, or weighing device.
4.1 Principle—An isothermal heat conduction calorimeter
6.1.3 Sample Holder—A device that holds the cement paste
consists of a constant-temperature heat sink to which two
and provides intimate contact with the calorimeter heat sensing
heat-flow sensors and sample holders are attached in a manner
device and prevents evaporation of mixing water. If using
resulting in good thermal conductivity. One heat-flow sensor
commercially manufactured equipment, consult the recom-
and sample holder contains the sample of interest. The other
mendations of the manufacturer in choosing sample holders.
heat-flow sensor is a reference cell containing a blank sample
6.1.4 Resistance Heater—An electrical device fabricated
that evolves no heat. The heat of hydration released by the
from material with similar heat capacity and shape as the test
reacting cementitious sample flows across the sensor and into
sample, but containing a resistor connected to a constant-
the heat sink. The output from the calorimeter is the difference
voltage power supply such that a stable output of 0.010 J ⁄s 6
in heat flow (thermal power) between the sample cell and the
0.0002 J/s can be generated (see Note 1).
reference cell. The heat-flow sensor actually senses a small
NOTE 1—A simple procedure for fabricating heaters and blanks having
temperature gradient that develops across the device, however
the same approximate shape and heat capacity as a sample is to make
the heat is removed from the hydrating sample fast enough
specimen similar to one used in a determination out of plaster of Paris
that, for practical purposes, the sample remains at a constant embedded with a small resistor. Plaster of Paris has only a transient heat
of hydration and is not aggressive to electronic components. A resistance
temperature (isothermal).
of 100 Ω to 300 Ω is a convenient value when using voltages of 0.1 V to
4.2 The output from the heat-flow sensor is an electrical
10 V to drive heat production.
voltage signal that is proportional to the thermal power from
6.1.5 Reference Specimen—A sample fabricated from an
the sample. This output must be calibrated to a known thermal
inert material with similar heat capacity and shape as the test
power. In this method this is accomplished by measurements
sample. This is used in the reference cell.
on a heat source that emits a constant and known thermal
6.1.6 Multimeter—An instrument for measuring DC voltage
power. The integral of the thermal power over the time of the
and resistance values for the resistance heater described in
test is the heat of hydration. Alternatively, a cementitious
6.1.4 to an accuracy of 1 %. This instrument is only required if
material with a known heat of hydration can be used for
the calorimeter does not contain built-in calibration capability.
calibration as described in Appendix X2.
6.1.7 Power Supply—A constant voltage DC power supply
4.3 Two methods are described. In Method A the sample and
with a power output range sufficient to simulate the maximum
water are both temperature equilibrated and mixed inside the
output of a hydrating cement sample (see Note 2). This
calorimeter. This method is the most direct way to determine
equipment is only required if an instrument does not contain
heat of hydration. In Method B the sample is mixed in the built-in calibration capability.
sample vial outside of the calorimeter using temperature
NOTE 2—A power output of at least 0.33 J/s is needed for most
equilibrated materials then put into the calorimeter. This
applications.
method offers certain practicality, but depending on the mate-
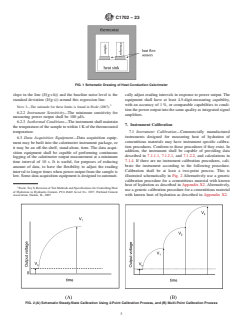
6.2 Calorimeter—The schematic design of a calorimeter is
rials being analyzed and procedures used for mixing and
given in Fig. 1. It shall consist of a sample holder for the test
handling, this method may suffer from small errors due to
and reference specimens, each thermally connected to heat-
periods of hydration being missed or spurious heat being
flow sensors, which are thermally connected to a constant-
introduced or taken away from the calorimeter during setup or
temperature heat sink. The actual design of an individual
combinations thereof.
instrument, whether commercial or homemade, may vary, but
it should follow the criteria given below. Any other suitable
5. Significance and Use
arrangement that satisfies 6.2.1 – 6.2.3 is acceptable.
5.1 This method is suitable for determining the total heat of
6.2.1 Instrument Stability—The baseline shall exhibit a low
hydration of hydraulic cement at constant temperature at ages
random noise level and be stable against drift. This property
up to 7 days to confirm specification compliance.
shall be verified on a new instrument and whenever there are
5.2 This method compliments Practice C1679 by providing questions about performance. The rate of change of the
details of calorimeter equipment, calibration, and operation. baseline measured during a time period of 3 days shall be
Practice C1679 emphasizes interpretation significant events in ≤20 μJ ⁄s per gram sample per hour of the test and a baseline
cement hydration by analysis of time dependent patterns of random noise level of ≤10 μJ/s per gram sample (see Note 3).
heat flow, but does not provide the level of detail necessary to In practice the baseline is measured for 3 days and a straight
give precision test results at specific test ages required for line is fitted to the power (J/(g·s)) versus time (h) data using a
specification compliance. linear regression procedure. The long term drift is then the
C1702 − 23
FIG. 1 Schematic Drawing of Heat Conduction Calorimeter
slope in the line (J/(g·s·h)) and the baseline noise level is the cally adjust reading intervals in response to power output. The
standard deviation (J/(g·s)) around this regression line. equipment shall have at least 4.5-digit-measuring capability,
with an accuracy of 1 %, or comparable capabilities to condi-
NOTE 3—The rationale for these limits is found in Poole (2007).
tion the power output into the same quality as integrated signal
6.2.2 Instrument Sensitivity—The minimum sensitivity for
amplifiers.
measuring power output shall be 100 μJ/s.
6.2.3 Isothermal Conditions—The instrument shall maintain
7. Instrument Calibration
the temperature of the sample to within 1 K of the thermostated
temperature. 7.1 Instrument Calibration—Commercially manufactured
instruments designed for measuring heat of hydration of
6.3 Data Acquisition Equipment—Data acquisition equip-
cementitious materials may have instrument specific calibra-
ment may be built into the calorimeter instrument package, or
tion procedures. Conform to these procedures if they exist. In
it may be an off-the-shelf, stand-alone, item. The data acqui-
addition, the instrument shall be capable of providing data
sition equipment shall be capable of performing continuous
described in 7.1.1.1, 7.1.2.1, and 7.1.2.2, and calculations in
logging of the calorimeter output measurement at a minimum
7.1.4. If there are no instrument calibration procedures, cali-
time interval of 10 s. It is useful, for purposes of reducing
brate the instrument according to the following procedure.
amount of data, to have the flexibility to adjust the reading
Calibration shall be at least a two-point process. This is
interval to longer times when power output from the sample is
illustrated schematically in Fig. 2 Alternatively use a generic
low. Some data acquisition equipment is designed to automati-
calibration procedure for a cementitious material with known
heat of hydration as described in Appendix X2. Alternatively,
Poole, Toy S, Revision of Test Methods and Specifications for Controlling Heat
use a generic calibration procedure for a cementitious material
of Hydration in Hydraulic-Cement, PCA R&D Serial No. 3007, Portland Cement
with known heat of hydration as described in Appendix X2.
Association, Skokie, IL, 2007
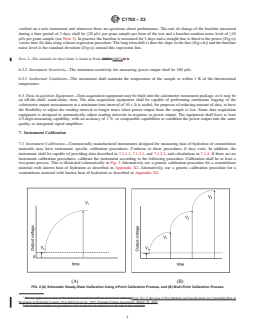
FIG. 2 (A) Schematic Steady-State Calibration Using 2-Point Calibration Process, and (B) Multi-Point Calibration Process
C1702 − 23
7.1.1 Mount the resistance heater and the blank specimen in tion to the next, then calibrations intervals must be reduced
their respective measuring cells and start data collection. This until this stability limit is reached.
step measures the baseline calorimeter output (in units of V or
mV) when no heat is being generated. 8. Procedure
7.1.1.1 Measure this baseline when it reaches a constant
8.1 The thermal mass of the inert reference specimen should
value (drift ≤ 20 μJ/s per gram sample per hour).
always be similar to the thermal mass of the target cement
7.1.1.2 Record this output as V for P = 0 (see Note 4).
0 0
paste. Verify that the calorimeter equipment temperature is
within 0.2 °C of target temperature with the proper mass of
NOTE 4—V may not be zero voltage, but may be a positive or negative
inert material charged in the reference cells no later than one
number. The practice of using a test cell and a reference cell usually results
in the V being a relatively small number but, depending on the variability day before performing a test. Determine that the calorimeter is
in properties of some hardware, it may not be zero.
at temperature equilibrium by verifying that the baseline is
stable over a period of 30 min or longer. The temperature of the
7.1.2 Power in the heater circuit is related to voltage and
heat sink during the test shall be 23.0 °C 6 1.0 °C, unless a
resistance by the following equation:
different temperature is required by the analysis.
P 5 I R (1)
NOTE 7—The time required to reach thermal equilibrium depends on
where:
the instrument. Generally, it is recommended to set the temperature
control unit of the calorimeter at target temperature at least 18 h before
P = power, J/s,
testing.
I = applied current, amperes, and
R = resistance, ohms. 8.1.1 Baseline Verification Test—This test is recommended
prior to testing and required whenever there is a change in the
Apply sufficient voltage to the heater circuit to generate a
operating temperature of the calorimeter or in ambient operat-
heat output of approximately 0.1 J/s, measured to an accuracy
ing conditions. For each active calorimeter cell, prepare a
of 5 %.
sample of water without any cement and without any mixing,
7.1.2.1 Allow the output to stabilize signal at a drift of
but with the same thermal mass as the inert reference speci-
≤0.1 % over 60 min or ≤0.05 % over 30 min.
men. Alternatively, use another inert material with equal
7.1.2.2 Record this output as V for a power P (see Note 5).
1 1
thermal mass as the inert reference specimen. Seal each vial
This is the minimum requirement for a calibration sequence. At
with a vapor-tight lid (see Note 8). For each active calorimeter
the users discretion any number of voltage levels may be used
cell, load the sample container with water or other inert
to characterize the operating range of the calorimeter.
material of equal thermal mass into the calorimeter and start
logging. Log the signal for a minimum of 24 h. Calculate the
NOTE 5—The early C A reaction of a typical portland cement evolves
a maximum power of about 0.02 J/(g·s). The alite phase typically evolves heat as a function of time per gram cement normally used in the
heat at a maximum power of about 0.002 J/(g·s) during the first 24 h of
calculation section, although no cement is used for this
hydration. A 5 g sample then generates power peaks in the range of
baseline verification test. A re-calibration is required if the
0.10 J ⁄s in the first few minutes after adding water, and in the range of
absolute value of the calculated heat per hour obtained 6 h from
0.010 J ⁄s in the first 24 h.
start of logging to the end of the test is higher than 0.10 J/(gh),
7.1.3 Calibration Coeffıcients—Calculate calibration coeffi-
where the mass (g) refers to the mass of cement intended to be
cients by fitting the power versus voltage output data to a to a
used.
mathematical relationship using standard curve fitting tech-
NOTE 8—The effectiveness of this sealing in preventing any evapora-
niques. Power (P), in units of J/s (or watts), is the dependent
tion (and its accompanying evaporative cooling) is variable depending on
variable (y) in the calibration equation, and output voltage (V),
the materials and techniques employed. Determining the mass of the
in units of mV, is the independent variable (x). This equation is
sealed vial to the nearest 0.001 g for a small (up to 10.000 g) sample or
0.01 g for a larger sample at the beginning and end of the test is a
then used to translate mV output to power units meaningful for
convenient method to assess the adequacy of the sealing operation for a
calculating heat flow (see Note 6).
sample with hydrating cementitious material. As a rule of thumb, for a w/c
= 0.5 cement paste, 0.3 % loss of water due to evaporation over 7 days,
NOTE 6—A linear calibration equation is found to be suitable in many
may, if not corrected for, result in a heat loss of approximately 3.7 J/g
instruments over the operating range necessary to analyze portland
cement. If the measured mass loss is assumed to be due to water
cements, as in the following equation: P = A + BV. In this case, the fitted
evaporation, it can be converted to an equivalent heat release (loss) using
coefficients A (y-axis intercept) and B (slope) are in units of J/s and
the known heat of vaporization of water of 43.99 kJ/mol or 2440 J/g at
J/(mV·s), respectively.
25 °C. A convenient method to approximate and compensate for the heat
7.1.4 In a multi-channel instrument containing several
loss due to evaporation during calibration is to measure the voltage signal
and mass loss with water in the sample vials as part of the baseline
calorimeters, all channels shall be calibrated individually.
calibration.
However, it is possible to calibrate all calorimeters simultane-
NOTE 9—The results from the baseline verification test can be used to
ously using multiple resistance heaters and having the same
recalculate the baseline value P in 7.1.
current passing through the heaters in all calorimeter cells.
NOTE 10—When performing the baseline verification test, use the same
thermal mass of water as in target cement paste.
7.1.5 Calibration shall be executed at regular intervals to
NOTE 11—Representative values of specific heat capacity for selected
determine the calibration coefficient. The length of the time
materials tested by this method are listed in Appendix X4.
intervals between calibrations is dependent on the instrument
NOTE 12—Calculation of thermal mass. The heat capacity of a typical
and the personnel, and must be determined empirically. If the
portland cement and water is 0.75 and 4.18 J/g/K, respectively. If, for
calibration coefficient differs more than 2 % from one calibra- example, a cement paste is prepared using 3.00 g cement and 1.5 g water,
-----------------
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: C1702 − 17 C1702 − 23
Standard Test Method for
Measurement of Heat of Hydration of Hydraulic
Cementitious Materials Using Isothermal Conduction
Calorimetry
This standard is issued under the fixed designation C1702; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*Scope
1.1 This test method specifies the apparatus and procedure for determining total heat of hydration of hydraulic cementitious
materials at test ages up to 7 days by isothermal conduction calorimetry.
1.2 This test method also outputs data on rate of heat of hydration versus time that is useful for other analytical purposes, as
covered in Practice C1679.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
C186 Test Method for Heat of Hydration of Hydraulic Cement (Withdrawn 2019)
C1679 Practice for Measuring Hydration Kinetics of Hydraulic Cementitious Mixtures Using Isothermal Calorimetry
E691 Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
3. Terminology
3.1 Definitions of Terms Specific to This Standard:
3.1.1 baseline, n—the time-series signal from the calorimeter when measuring output from a sample of approximately the same
mass and thermal properties as a cement sample, but which is not generating or consuming heat.
3.1.2 heat, n—the time integral of thermal power measured in joules (J).
This test method is under the jurisdiction of ASTM Committee C01 on Cement and is the direct responsibility of Subcommittee C01.26 on Heat of Hydration.
Current edition approved Feb. 1, 2017Aug. 1, 2023. Published February 2017August 2023. Originally approved in 2009. Last previous edition approved in 20152017 as
C1702 – 15b.C1702 – 17. DOI: 10.1520/C1702-17.10.1520/C1702-23.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
C1702 − 23
3.1.3 isothermal conduction calorimeter, n—a calorimeter that measures heat flow from a sample maintained at a constant
temperature by intimate thermal contact with a constant temperature heat sink.
3.1.4 reference cell, n—a heat-flow measuring cell that is dedicated to measuring power from a sample that is generating no heat.
3.1.4.1 Discussion—
The purpose of the reference cell is to correct for baseline drift and other systematic errors that can occur in heat-flow measuring
equipment.
3.1.5 sensitivity, n—the minimum change in thermal power reliably detectable by an isothermal calorimeter.
3.1.5.1 Discussion—
For this application, sensitivity is taken as ten times the random noise (standard deviation) in the baseline signal.
3.1.6 thermal mass, n—the amount of thermal energy that can be stored by a material (J/K).
3.1.6.1 Discussion—
The thermal mass of a given material is calculated by multiplying the mass by the specific heat capacity of the material. For the
purpose of calculating the thermal mass used in this standard, the following specific heat capacities can be used: The specific heat
capacity of a typical unhydrated portland cement and water is 0.75 and 4.18 J/(g·K), respectively. Thus a mixture of A g of cement
and B g of water has a thermal mass of (0.75 × A + 4.18 × B) J/K. The specific heat capacity of typical quartz and limestone is
0.75 and 0.84 J ⁄(g·K), respectively. The specific heat capacity of most amorphous supplementary cementitious material, such as
fly ash or slag, is approximately 0.8 J/(g·K).
3.1.7 thermal power, n—the heat production rate measured in joules per second (J/s).
3.1.7.1 Discussion—
This is the property measured by the calorimeter. The thermal power unit of measure is J/s, which is equivalent to the watt. The
watt is also a common unit of measure used to represent thermal power.
4. Summary of Test Method
4.1 Principle—An isothermal heat conduction calorimeter consists of a constant-temperature heat sink to which two heat-flow
sensors and sample holders are attached in a manner resulting in good thermal conductivity. One heat-flow sensor and sample
holder contains the sample of interest. The other heat-flow sensor is a reference cell containing a blank sample that evolves no heat.
The heat of hydration released by the reacting cementitious sample flows across the sensor and into the heat sink. The output from
the calorimeter is the difference in heat flow (thermal power) between the sample cell and the reference cell. The heat-flow sensor
actually senses a small temperature gradient that develops across the device, however the heat is removed from the hydrating
sample fast enough that, for practical purposes, the sample remains at a constant temperature (isothermal).
4.2 The output from the heat-flow sensor is an electrical voltage signal that is proportional to the thermal power from the sample.
This output must be calibrated to a known thermal power. In this method this is accomplished by measurements on a heat source
that emits a constant and known thermal power. The integral of the thermal power over the time of the test is the heat of hydration.
Alternatively, a cementitious material with a known heat of hydration can be used for calibration as described in Appendix X2.
4.3 Two methods are described. In Method A the sample and water are both temperature equilibrated and mixed inside the
calorimeter. This method is the most direct way to determine heat of hydration. In Method B the sample is mixed in the sample
vial outside of the calorimeter using temperature equilibrated materials then put into the calorimeter. This method offers certain
practicality, but depending on the materials being analyzed and procedures used for mixing and handling, this method may suffer
from small errors due to periods of hydration being missed or spurious heat being introduced or taken away from the calorimeter
during setup or combinations thereof.
5. Significance and Use
5.1 This method is suitable for determining the total heat of hydration of hydraulic cement at constant temperature at ages up to
7 days to confirm specification compliance. It gives test results equivalent to Test Method C186 up to 7 days of age (1).
5.2 This method compliments Practice C1679 by providing details of calorimeter equipment, calibration, and operation. Practice
C1702 − 23
C1679 emphasizes interpretation significant events in cement hydration by analysis of time dependent patterns of heat flow, but
does not provide the level of detail necessary to give precision test results at specific test ages required for specification compliance.
6. Apparatus
6.1 Miscellaneous Equipment:
6.1.1 Balance—Accurate to 0.01 g.
6.1.2 Volumetric Dispenser—A device for measuring volume or mass of water, accurate to 0.1 mL. This could be a syringe, pipette,
or weighing device.
6.1.3 Sample Holder—A device that holds the cement paste and provides intimate contact with the calorimeter heat sensing device
and prevents evaporation of mixing water. If using commercially manufactured equipment, consult the recommendations of the
manufacturer in choosing sample holders.
6.1.4 Resistance Heater—An electrical device fabricated from material with similar heat capacity and shape as the test sample,
but containing a resistor connected to a constant-voltage power supply such that a stable output of 0.0100.010 J ⁄s 6 0.0002 J/s
can be generated (see Note 1).
NOTE 1—A simple procedure for fabricating heaters and blanks having the same approximate shape and heat capacity as a sample is to make specimen
similar to one used in a determination out of plaster of Paris embedded with a small resistor. Plaster of Paris has only a transient heat of hydration and
is not aggressive to electronic components. A resistance of 100100 Ω to 300 Ω is a convenient value when using voltages of 0.10.1 V to 10 V to drive
heat production.
6.1.5 Reference Specimen—A sample fabricated from an inert material with similar heat capacity and shape as the test sample. This
is used in the reference cell.
6.1.6 Multimeter—An instrument for measuring DC voltage and resistance values for the resistance heater described in 6.1.4 to
an accuracy of 1 %. This instrument is only required if the calorimeter does not contain built-in calibration capability.
6.1.7 Power Supply—A constant voltage DC power supply with a power output range sufficient to simulate the maximum output
of a hydrating cement sample (see Note 2). This equipment is only required if an instrument does not contain built-in calibration
capability.
NOTE 2—A power output of at least 0.33 J/s is needed for most applications.
6.2 Calorimeter—The schematic design of a calorimeter is given in Fig. 1. It shall consist of a sample holder for the test and
reference specimens, each thermally connected to heat-flow sensors, which are thermally connected to a constant-temperature heat
sink. The actual design of an individual instrument, whether commercial or homemade, may vary, but it should follow the criteria
given below. Any other suitable arrangement that satisfies 6.2.1 – 6.2.3 is acceptable.
6.2.1 Instrument Stability—The baseline shall exhibit a low random noise level and be stable against drift. This property shall be
FIG. 1 Schematic Drawing of Heat Conduction Calorimeter
C1702 − 23
verified on a new instrument and whenever there are questions about performance. The rate of change of the baseline measured
during a time period of 3 days shall be ≤20 μJ ⁄s per gram sample per hour of the test and a baseline random noise level of ≤10
μJ/s per gram sample (see Note 3). In practice the baseline is measured for 3 days and a straight line is fitted to the power (J/(g·s))
versus time (h) data using a linear regression procedure. The long term drift is then the slope in the line (J/(g·s·h)) and the baseline
noise level is the standard deviation (J/(g·s)) around this regression line.
NOTE 3—The rationale for these limits is found in Poole (2007) (2007).(1 ).
6.2.2 Instrument Sensitivity—The minimum sensitivity for measuring power output shall be 100 μJ/s.
6.2.3 Isothermal Conditions—The instrument shall maintain the temperature of the sample to within 1 K of the thermostated
temperature.
6.3 Data Acquisition Equipment—Data acquisition equipment may be built into the calorimeter instrument package, or it may be
an off-the-shelf, stand-alone, item. The data acquisition equipment shall be capable of performing continuous logging of the
calorimeter output measurement at a minimum time interval of 10 s. It is useful, for purposes of reducing amount of data, to have
the flexibility to adjust the reading interval to longer times when power output from the sample is low. Some data acquisition
equipment is designed to automatically adjust reading intervals in response to power output. The equipment shall have at least
4.5-digit-measuring capability, with an accuracy of 1 %, or comparable capabilities to condition the power output into the same
quality as integrated signal amplifiers.
7. Instrument Calibration
7.1 Instrument Calibration—Commercially manufactured instruments designed for measuring heat of hydration of cementitious
materials may have instrument specific calibration procedures. Conform to these procedures if they exist. In addition, the
instrument shall be capable of providing data described in 7.1.1.1, 7.1.2.1, and 7.1.2.2, and calculations in 7.1.4. If there are no
instrument calibration procedures, calibrate the instrument according to the following procedure. Calibration shall be at least a
two-point process. This is illustrated schematically in Fig. 2 Alternatively use a generic calibration procedure for a cementitious
material with known heat of hydration as described in Appendix X2. Alternatively, use a generic calibration procedure for a
cementitious material with known heat of hydration as described in Appendix X2.
FIG. 2 (A) Schematic Steady-State Calibration Using 2-Point Calibration Process, and (B) Multi-Point Calibration Process
The last approved version of this historical standard is referenced on www.astm.org.Poole, Toy S, Revision of Test Methods and Specifications for Controlling Heat of
Hydration in Hydraulic-Cement, PCA R&D Serial No. 3007, Portland Cement Association, Skokie, IL, 2007
The boldface numbers in parentheses refer to the list of references at the end of this standard.
C1702 − 23
7.1.1 Mount the resistance heater and the blank specimen in their respective measuring cells and start data collection. This step
measures the baseline calorimeter output (in units of V or mV) when no heat is being generated.
7.1.1.1 Measure this baseline when it reaches a constant value (drift ≤ 20 μJ/s per gram sample per hour).
7.1.1.2 Record this output as V for P = 0 (see Note 4).
0 0
NOTE 4—V may not be zero voltage, but may be a positive or negative number. The practice of using a test cell and a reference cell usually results in
the V being a relatively small number but, depending on the variability in properties of some hardware, it may not be zero.
7.1.2 Power in the heater circuit is related to voltage and resistance by the following equation:
P 5 I R (1)
where:
P = power, J/s,
I = applied current, amperes, and
R = resistance, ohms.
Apply sufficient voltage to the heater circuit to generate a heat output of approximately 0.1 J/s, measured to an accuracy of 5 %.
7.1.2.1 Allow the output to stabilize signal at a drift of ≤0.1 % over 60 min or ≤0.05 % over 30 min.
7.1.2.2 Record this output as V for a power P (see Note 5). This is the minimum requirement for a calibration sequence. At the
1 1
users discretion any number of voltage levels may be used to characterize the operating range of the calorimeter.
NOTE 5—The early C A reaction of a typical portland cement evolves a maximum power of about 0.02 J/(g·s). The alite phase typically evolves heat at
a maximum power of about 0.002 J/(g·s) during the first 24 h of hydration. A 5-g5 g sample then generates power peaks in the range of 0.10 J ⁄s in the
first few minutes after adding water, and in the range of 0.010 J ⁄s in the first 24 h.
7.1.3 Calibration Coeffıcients—Calculate calibration coefficients by fitting the power versus voltage output data to a to a
mathematical relationship using standard curve fitting techniques. Power (P), in units of J/s (or watts), is the dependent variable
(y) in the calibration equation, and output voltage (V), in units of mV, is the independent variable (x). This equation is then used
to translate mV output to power units meaningful for calculating heat flow (see Note 6).
NOTE 6—A linear calibration equation is found to be suitable in many instruments over the operating range necessary to analyze portland cements, as
in the following equation: P = A + BV. In this case, the fitted coefficients A (y-axis intercept) and B (slope) are in units of J/s and J/(mV·s), respectively.
7.1.4 In a multi-channel instrument containing several calorimeters, all channels shall be calibrated individually. However, it is
possible to calibrate all calorimeters simultaneously using multiple resistance heaters and having the same current passing through
the heaters in all calorimeter cells.
7.1.5 Calibration shall be executed at regular intervals to determine the calibration coefficient. The length of the time intervals
between calibrations is dependent on the instrument and the personnel, and must be determined empirically. If the calibration
coefficient differs more than 2 % from one calibration to the next, then calibrations intervals must be reduced until this stability
limit is reached.
8. Procedure
8.1 The thermal mass of the inert reference specimen should always be similar to the thermal mass of the target cement paste.
Verify that the calorimeter equipment temperature is within 0.2°C0.2 °C of target temperature with the proper mass of inert
material charged in the reference cells no later than one day before performing a test. Determine that the calorimeter is at
temperature equilibrium by verifying that the baseline is stable over a period of 30 min or longer. The temperature of the heat sink
during the test shall be 23.0 6 1.0°C,23.0 °C 6 1.0 °C, unless a different temperature is required by the analysis.
NOTE 7—The time required to reach thermal equilibrium depends on the instrument. Generally, it is recommended to set the temperature control unit of
the calorimeter at target temperature at least 18 h before testing.
C1702 − 23
8.1.1 Baseline Verification Test—This test is recommended prior to testing and required whenever there is a change in the
operating temperature of the calorimeter or in ambient operating conditions. For each active calorimeter cell, prepare a sample of
water without any cement and without any mixing, but with the same thermal mass as the inert reference specimen. Alternatively,
use another inert material with equal thermal mass as the inert reference specimen. Seal each vial with a vapor-tight lid (see Note
8). For each active calorimeter cell, load the sample container with water or other inert material of equal thermal mass into the
calorimeter and start logging. Log the signal for a minimum of 24 h. Calculate the heat as a function of time per gram cement
normally used in the calculation section, although no cement is used for this baseline verification test. A re-calibration is required
if the absolute value of the calculated heat per hour obtained 6 h from start of logging to the end of the test is higher than 0.10
J/(gh), where the mass (g) refers to the mass of cement intended to be used.
NOTE 8—The effectiveness of this sealing in preventing any evaporation (and its accompanying evaporative cooling) is variable depending on the
materials and techniques employed. Determining the mass of the sealed vial to the nearest 0.001 g for a small (up to 10.000 g) sample or 0.01 g for a
larger sample at the beginning and end of the test is a convenient method to assess the adequacy of the sealing operation for a sample with hydrating
cementitious material. As a rule of thumb, for a w/c = 0.5 cement paste, 0.3 % loss of water due to evaporation over 7 days, may, if not corrected for,
result in a heat loss of approximately 3.7 J/g cement. If the measured mass loss is assumed to be due to water evaporation, it can be converted to an
equivalent heat release (loss) using the known heat of vaporization of water of 43.99 kJ/mol or 2440 J/g at 25°C.25 °C. A convenient method to
approximate and compensate for the heat loss due to evaporation during calibration is to measure the voltage signal and mass loss with water in the sample
vials as part of the baseline calibration.
NOTE 9—The results from the baseline verification test can be used to recalculate the baseline value P in 7.1.
NOTE 10—When performing the baseline verification test, use the same thermal mass of water as in target cement paste.
NOTE 11—Representative values of specific heat capacity for selected materials tested by this method are listed in Appendix X4.
NOTE 12—Calculation of thermal mass. The heat capacity of a typical portland cement and water is 0.75 and 4.18 J/g/K, respectively. If, for example,
a cement paste is prepared using 3.00 g cement and 1.5 g water, the resulting cement paste has an approximate thermal mass of (3.00 × 0.75 + 1.5 × 4.18)
= 8.52 J/K, which is also the target thermal mass of the inert reference specimen. If using water for the baseline verification test, the corresponding mass
of water used is (8.52 ⁄ 4.18) = 2.04 g. After completion of the baseline verification test, a fraction of this water (1.5 g in this example) can be used for
the heat of hydration tests in the procedure section.
8.2 Method A—This method is used when an instrument is configured so that cementitious materials and water can be temperature
equilibrated and mixed while in place in the calorimeter cell.
8.2.1 Weigh at least 3 g of cementitious material (see Note 13), the mass recorded to the nearest 0.01 g, and plac
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...