ASTM E1786-17a
(Test Method)Standard Test Method for Determination of Low Levels of Water in Liquid Chlorine by On-Line Infrared Spectrophotometry
Standard Test Method for Determination of Low Levels of Water in Liquid Chlorine by On-Line Infrared Spectrophotometry
SIGNIFICANCE AND USE
4.1 Trace amounts of water may be detrimental to the use of chlorine in some applications. The amount of water in the chlorine must be known to prevent problems during its use.
SCOPE
1.1 This test method is designed for the on-line determination of the content of water in liquid chlorine in the concentration range of 0.5 to 15 mg/kg (ppm).
1.2 The values stated in SI units are to be regarded as standard. The values given in parentheses are for information only.
1.3 Review the current Safety Data Sheets (SDS) for detailed information concerning toxicity, first aid procedures, and safety precautions.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. Specific hazards statements are given in Section 7 and Note 3.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Jun-2017
- Technical Committee
- D16 - Aromatic, Industrial, Specialty and Related Chemicals
- Drafting Committee
- D16.13 - Chlorine
Relations
- Effective Date
- 01-Jul-2017
- Effective Date
- 01-Mar-2017
- Effective Date
- 01-Nov-2016
- Effective Date
- 15-Dec-2008
- Effective Date
- 01-Jun-2007
- Effective Date
- 01-Mar-2006
- Effective Date
- 10-Jun-2002
- Effective Date
- 10-Feb-1999
- Effective Date
- 10-Feb-1999
Overview
ASTM E1786-17a is the internationally recognized standard test method developed by ASTM International for the determination of low levels of water in liquid chlorine. Using on-line infrared spectrophotometry (IR), this standard enables precise measurement of water content in liquid chlorine in the range of 0.5 to 15 mg/kg (ppm). Accurate analysis of trace water levels is critical, as even minimal amounts can negatively affect the use and quality of chlorine in various industrial and chemical applications.
This method is widely adopted for its ability to provide continuous, real-time monitoring, thereby supporting process control and compliance in chlorine production, transportation, and storage.
Key Topics
Measurement Range and Sensitivity
The standard method measures water in liquid chlorine at concentrations between 0.5 and 15 mg/kg, ensuring detection of even trace levels.On-Line Infrared Spectrophotometry
Employs a process IR spectrometer (typically an FTIR or dispersive instrument) and a specially designed IR cell, enabling on-line, automated measurement for continuous monitoring.Calibration and Quality Control
Calibration is achieved using prepared chlorine standards with known water concentrations. Rigorous quality control processes are integrated to ensure reliable results, referencing ASTM D6809 for QA/QC procedures if needed.Safety Considerations
Liquid chlorine is hazardous-procedures stress the importance of using SDS, proper protective equipment, and facility safety measures to prevent exposure and environmental release. Only qualified personnel should perform this analysis.Significance of Trace Water Determination
Water contamination in chlorine can cause corrosion, process inefficiencies, and product quality issues, making periodic and precise analysis essential for operational safety and product integrity.
Applications
Chlorine Production Facilities
On-line water content monitoring ensures product compliance and operational efficiency by mitigating the risks associated with water impurities.Chemical Manufacturing
In downstream processing where chlorine is used, knowledge of water content enables optimization of chemical reactions and product quality.Storage and Transport
Ensures conformity with safety regulations and prevents degradation of storage vessels, pipelines, and transport containers.Quality Assurance and Regulatory Compliance
Meeting industry and governmental standards for moisture content in chlorinated products, supporting regulatory reporting and audit requirements.Research and Development
Provides reliable analytical data for process improvement, safety evaluations, and innovation in chlorine handling and application.
Related Standards
- ASTM D1193 - Specification for Reagent Water, essential for preparing samples and standards.
- ASTM D6809 - Guide for Quality Control and Quality Assurance Procedures for Aromatic Hydrocarbons and Related Materials.
- ASTM E806 - Test Method for Carbon Tetrachloride and Chloroform in Liquid Chlorine by Direct Injection (Gas Chromatographic Procedure).
- 49 CFR 173 - U.S. Department of Transportation regulations on charging, requirements, and transport of compressed gases including chlorine.
- Chlorine Institute Pamphlet No. 1 – Chlorine Basics - Provides foundational information for chlorine handling and safety procedures.
Keywords: ASTM E1786, water determination in chlorine, on-line infrared spectrophotometry, FTIR, liquid chlorine analysis, chlorine quality assurance, trace water measurement, industrial chemical standards.
By following ASTM E1786-17a, organizations achieve reliable, consistent detection of water in liquid chlorine, supporting operational safety, product quality, and regulatory compliance in the chemical industry.
Buy Documents
ASTM E1786-17a - Standard Test Method for Determination of Low Levels of Water in Liquid Chlorine by On-Line Infrared Spectrophotometry
REDLINE ASTM E1786-17a - Standard Test Method for Determination of Low Levels of Water in Liquid Chlorine by On-Line Infrared Spectrophotometry
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM E1786-17a is a standard published by ASTM International. Its full title is "Standard Test Method for Determination of Low Levels of Water in Liquid Chlorine by On-Line Infrared Spectrophotometry". This standard covers: SIGNIFICANCE AND USE 4.1 Trace amounts of water may be detrimental to the use of chlorine in some applications. The amount of water in the chlorine must be known to prevent problems during its use. SCOPE 1.1 This test method is designed for the on-line determination of the content of water in liquid chlorine in the concentration range of 0.5 to 15 mg/kg (ppm). 1.2 The values stated in SI units are to be regarded as standard. The values given in parentheses are for information only. 1.3 Review the current Safety Data Sheets (SDS) for detailed information concerning toxicity, first aid procedures, and safety precautions. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. Specific hazards statements are given in Section 7 and Note 3. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 Trace amounts of water may be detrimental to the use of chlorine in some applications. The amount of water in the chlorine must be known to prevent problems during its use. SCOPE 1.1 This test method is designed for the on-line determination of the content of water in liquid chlorine in the concentration range of 0.5 to 15 mg/kg (ppm). 1.2 The values stated in SI units are to be regarded as standard. The values given in parentheses are for information only. 1.3 Review the current Safety Data Sheets (SDS) for detailed information concerning toxicity, first aid procedures, and safety precautions. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. Specific hazards statements are given in Section 7 and Note 3. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E1786-17a is classified under the following ICS (International Classification for Standards) categories: 71.040.50 - Physicochemical methods of analysis; 71.100.20 - Gases for industrial application. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E1786-17a has the following relationships with other standards: It is inter standard links to ASTM E1786-17, ASTM E806-17, ASTM D6809-02(2016), ASTM E806-08, ASTM D6809-02(2007), ASTM D1193-06, ASTM D6809-02, ASTM D1193-99, ASTM D1193-99e1. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E1786-17a is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E1786 − 17a
Standard Test Method for
Determination of Low Levels of Water in Liquid Chlorine by
On-Line Infrared Spectrophotometry
This standard is issued under the fixed designation E1786; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* 2.2 Federal Standards:
49 CFR 173Code of Federal RegulationsTitle 49Transpor-
1.1 This test method is designed for the on-line determina-
tation:Shippers’GeneralRequirementsforShipmentsand
tion of the content of water in liquid chlorine in the concen-
Packaging, including the following sections:
tration range of 0.5 to 15 mg/kg (ppm).
173.304Charging of Cylinders with Liquefied Compressed
1.2 The values stated in SI units are to be regarded as
Gas
standard. The values given in parentheses are for information
173.314Requirements for Compressed Gases in Tank Cars
only.
173.315Compressed Gases in Cargo Tanks and Portable
1.3 Review the current Safety Data Sheets (SDS) for de- Tank Containers
2.3 Other Document:
tailedinformationconcerningtoxicity,firstaidprocedures,and
safety precautions. Chlorine Institute Pamphlet No. 1— Chlorine Basics
1.4 This standard does not purport to address all of the
3. Summary of Test Method
safety concerns, if any, associated with its use. It is the
3.1 Liquid chlorine continuously flows through a special
responsibility of the user of this standard to establish appro-
infrared cell where it is maintained as a liquid under its own
priate safety, health, and environmental practices and deter-
pressure. A process infrared spectrometer scans from 400 to
mine the applicability of regulatory limitations prior to use.
4400 wavenumbers of the infrared transmission spectrum of
Specific hazards statements are given in Section 7 and Note 3.
liquid chlorine. This spectrum then is ratioed to one obtained
1.5 This international standard was developed in accor-
from the nitrogen-filled infrared cell previously. The ratioed
dance with internationally recognized principles on standard-
spectrumisconvertedtoabsorbance,andthenetabsorbanceof
ization established in the Decision on Principles for the
water band at 1596 wavenumbers, relative to a reference at
Development of International Standards, Guides and Recom-
1663 wavenumbers, is determined.
mendations issued by the World Trade Organization Technical
3.2 The amount of water corresponding to this net absor-
Barriers to Trade (TBT) Committee.
banceisdeterminedfromacalibrationcurvepreparedfromthe
2. Referenced Documents
infrared absorbencies of standards which contain concentra-
tions of water in liquid chlorine. These standards are prepared
2.1 ASTM Standards:
from manual samples of liquid chlorine in tantalum cylinders.
D1193Specification for Reagent Water
Sample from each cylinder is introduced into a calibration
D6809Guide for Quality Control and Quality Assurance
infraredcellandmaintainedasaliquidunderitsownpressure.
Procedures for Aromatic Hydrocarbons and Related Ma-
terials
4. Significance and Use
E806TestMethodforCarbonTetrachlorideandChloroform
4.1 Traceamountsofwatermaybedetrimentaltotheuseof
in Liquid Chlorine by Direct Injection (Gas Chromato-
chlorine in some applications. The amount of water in the
graphic Procedure)
chlorine must be known to prevent problems during its use.
5. Apparatus
This test method is under the jurisdiction of ASTM Committee D16 on
Aromatic, Industrial, Specialty and Related Chemicals and is the direct responsi-
5.1 Process Infrared Spectrometer, capable of measure-
bility of Subcommittee D16.13 on Chlorine.
ments in the 1600 wavenumber region. An FTIR with four
CurrenteditionapprovedJuly1,2017.PublishedJuly2017.Originallyapproved
in 1996. Last previous edition approved in 2017 as E1786–17. DOI: 10.1520/
E1786-17a. Code of Federal Regulations,AvailablefromU.S.GovernmentPrintingOffice,
For referenced ASTM standards, visit the ASTM website, www.astm.org, or Superintendent of Documents, 732 N. Capitol St., NW, Washington, DC 20401-
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM 0001, http://www.access.gpo.gov.
Standards volume information, refer to the standard’s Document Summary page on Available from The Chlorine Institute, Inc., 1300 Wilson Blvd., Suite 525,
the ASTM website. Arlington, VA 22209, https://www.chlorineinstitute.org.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E1786 − 17a
wavenumber resolution is the instrument of choice, but disper-
sive instruments also may be used to achieve similar results.
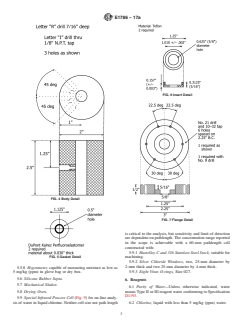
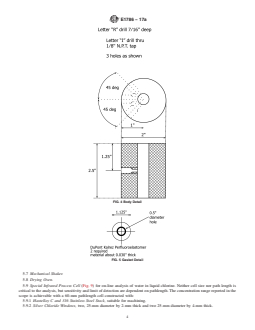
5.2 Special Infrared Calibration Cell (Fig. 1), as used for
calibration. Neither cell size nor pathlength are critical to the
analysis,butsensitivityandlimitofdetectionaredependenton
pathlength.TheconcentrationrangereportedintheSection1is
achievable with a 60-mm pathlength cell constructed with the
following: Figs. 2-7
5.2.1 Hastelloy C and 316 Stainless Steel Stock,suitablefor
machining,
5.2.2 Silver Chloride Windows, 0.5 cm×2.5 cm, and
5.2.3 Perfluoroelastomer Sheet,0.762mm(0.030in.)thick-
ness.
5.3 Ball Valves,Monel6.35mm( ⁄4-in.)valvewithpipeand
6.35 mm ( ⁄4-in.) tube ends.
5.4 Needle Valves, Nickel or Monel 6.35 mm ( ⁄4-in.) valve
with pipe and 6.35 mm ( ⁄4-in.) tube ends.
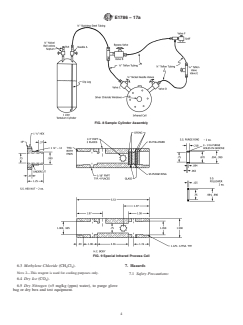
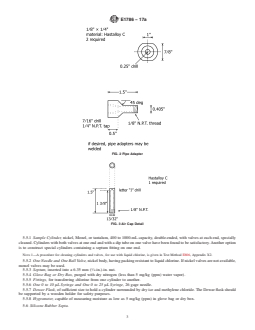
5.5 Sample Cylinder Assembly (Fig. 8), consisting of:
5.5.1 Sample Cylinder, nickel, Monel, or tantalum, 400 to
1000-mL capacity, double-ended, with valves at each end,
specially cleaned. Cylinders with both valves at one end and
withadiptubeononevalvehavebeenfoundtobesatisfactory.
Another option is to construct special cylinders containing a
septum fitting on one end.
NOTE 1—A procedure for cleaning cylinders and valves, for use with
FIG. 2 Pipe Adapter
FIG. 3 Air Cap Detail
liquid chlorine, is given in Test Method E806, Appendix X2.
5.5.2 One Needle and One Ball Valve, nickel body, having
packing resistant to liquid chlorine. If nickel valves are not
available, monel valves may be used.
5.5.3 Septum, inserted into a 6.35 mm ( ⁄4-in.)-in. nut.
5.5.4 Glove Bag or Dry Box, purged with dry nitrogen (less
than 5 mg/kg (ppm) water vapor).
5.5.5 Fittings, for transferring chlorine from one cylinder to
another.
5.5.6 One0to10 µL Syringe and One 0 to 25 µL Syringe,
26 gage needle.
5.5.7 Dewar Flask, of sufficient size to hold a cylinder
surrounded by dry ice and methylene chloride. The Dewar
flask should be supported by a wooden holder for safety
FIG. 1 Infrared Cell (Drawing Not to Scale) purposes.
E1786 − 17a
FIG. 6 Insert Detail
FIG. 4 Body Detail
FIG. 7 Flange Detail
is critical to the analysis, but sensitivity and limit of detection
are dependent on pathlength.The concentration range reported
in the scope is achievable with a 60-mm pathlength cell
constructed with:
5.9.1 Hastelloy C and 316 Stainless Steel Stock,suitablefor
FIG. 5 Gasket Detail machining.
5.9.2 Silver Chloride Windows, two, 25-mm diameter by
2-mm thick and two 25-mm diameter by 4-mm thick.
5.5.8 Hygrometer, capable of measuring moisture as low as
5.9.3 Eight Viton O-rings, Size 027.
5 mg/kg (ppm) in glove bag or dry box.
5.6 Silicone Rubber Septa.
6. Reagents
5.7 Mechanical Shaker.
6.1 Purity of Water—Unless otherwise indicated, water
5.8 Drying Oven. meansType II or III reagent water conforming to Specification
D1193.
5.9 Special Infrared Process Cell (Fig. 9) for on-line analy-
sis of water in liquid chlorine. Neither cell size nor path length 6.2 Chlorine, liquid with less than 5 mg/kg (ppm) water.
E1786 − 17a
FIG. 8 Sample Cylinder Assembly
FIG. 9 Special Infrared Process Cell
6.3 Methylene Chloride (CH Cl ). 7. Hazards
2 2
NOTE 2—This reagent is used for cooling purposes only.
7.1 Safety Precautions:
6.4 Dry Ice (CO ).
6.5 Dry Nitrogen (<5 mg/kg (ppm) water), to purge glove
bag or dry box and test equipment.
E1786 − 17a
7.1.1 Chlorine is a corrosive and toxic material. Use a cylinders are cooled with plant air or N having <5 mg/kg
well-ventilated fume hood to house all test equipment, except (ppm)moisture.Thevalvesareremoved,placedinadesiccator
the infrared spectrophotometer, when this material is analyzed with a suitable drying agent, and cooled to room temperature.
in the laboratory.
7.1.2 Personswhoarethoroughlyfamiliarwiththehandling 9. Preparation of Standards for Calibration
of chlorine should perform this analysis. An experienced
9.1 Obtainfourclean,evacuatedsamplecylinders.Useonly
person should not work alone. The analyst must be provided
cylinders that have been properly pressure-tested. Equip three
with adequate eye protection (chemical goggles are recom-
with a ball valve and a needle valve, label these Cylinders No.
mended) and an approved chlorine respirator. Splashes of
2 through No. 4, and record weight to the nearest gram. Equip
liquid chlorine destroy clothing, and if such clothing is next to
thefourthcylinderwithtwoneedlevalves,weightothenearest
the skin, will produce irritation and burns.
gram,record,andlabelNo.1.Onceitcontainschlorine,itwill
7.1.3 When sampling and working with chlorine out of
be used to purge the infrared cell before standards are loaded.
doors, warn people downwind from such operations of the
Usecapsonallvalves.Ifdiptubesareused,attachthediptube
possible release of chlorine.
to the needle valve. Check the hygrometer to make sure the
7.1.4 Dispose of excess chlorine in an environmentally safe
atmosphere in the glove bag or dry box contains <5 mg/kg
and acceptable manner. If chlorine cannot be disposed of in a
(ppm) moisture before performing any transfers of chlorine.
chlorine consuming process, provide a chlorine absorption
9.2 Load the four cylinders with liquid chlorine. The liquid
system. When the analysis and sampling regimen requires an
chlorine should contain less than 5 mg/kg (ppm) water. All
initialpurgingofchlorinefromacontainer,thepurgedchlorine
cylinders must contain at least 75% by volume of liquid
should be similarly handled.Avoid purging to the atmosphere.
chlorine (less than 25% vapor space).All cylinders should be
7.1.5 In the event chlorine is inhaled, use first aid immedi-
weighed to the nearest gram and recorded to determine the
ately.
weight of chlorine in each cylinder (Note 3). Place the filled
cylinders into the glove bag or dry box inside the hood. Place
8. Sampling for Calibration Standards
all fittings, tools and equipment, including the purged infrared
8.1 Carefully choose sampling points. Ensure that the
cell, in the glove bag or dry box. Wait until the hygrometer in
samplepointisassociatedwithflowingchlorineandisnotnear
the glove bag or dry box is reading <5 mg/kg (ppm) moisture
a “dead leg” where the concentrations of impurities in the
before proceeding.
chlorine will never change because the chlorine never moves.
9.3 Remove the cap onValveAof Cylinder No. 1 and blow
If sampling through secondary piping, purge that piping well
with N to remove any trapped moisture. Do this each time a
with nitrogen or dry air before being blocked in. Otherwise,
connection is made. Then connect the cylinder and the special
temperature variations can result in water vapor condensing
infrared cell to the filling apparatus (Fig. 8) in such a way that
inside the piping to contaminate the chlorine sample when it is
the liquid chlorine will flow into the cell and the valves on all
grabbed.
partsoftheapparatusareclosedpriortofillingthecell.Forthe
8.1.1 Finally, perform sampling at a sample point represen-
following operations, refer to 7.4 on venting chlorine.
tative of the chlorine needing to be analyzed; that is, sample
9.4 Open Valve F and then Valve A. Flush the filling
pure chlorine after all purification steps, drying steps, and so
apparatus by partially opening Valve B for a few s and then
forth, to ensure that the analytical results are meaningful.
closing it. Leave Valve A open.
8.1.1.1 Sampling from tank cars, barges, storage tanks, and
9.4.1 Open Valves E and C. Flush the cell by partially
large cylinders presents unique problems. Each facility,
opening Valve D and observing flow, then close Valve C and
however, must be capable of delivering a liquid sample (not
then Valve D.
gas). See Chlorine Institute Pamphlet No. 1. (See 49 CFR 173,
including Parts 173.304, 173.314, and 173.315.)
9.5 FilltheinfraredcellbyopeningValveCandobservethe
filling of the cell windows. Close Valve C and empty the cell
8.2 Collect samples from these facilities in the sample
by opening Valve D. Close Valve D.
cylinder assembly listed in 6.5. Follow proper and safe
sampling techniques. The cylinder must contain at least 75% 9.6 Fill and empty the cell once more, as described above.
by volume of liquid chlorine (less than 25% vapor space).
9.6.1 Close Valve A and open Valve B, to purge the filling
apparatus, then close.
NOTE 3—Do not allow the cylinder to become liquid-full. Tare the
cylinder, fill with water, and reweigh to determine the weight of water in
9.7 Remove the cylinder. Connect Cylinder No. 2 to the
thecylinder.Multiplythisweightby1.56(theassumedspecificgravityof
fillingapparatusafterblowingValveAwithN .Makesurethat
liquid chlorine) and by 0.75 to obtain the weight of chlorine to fill the
thecylinderisconnectedtotheapparatusinsuchawaythatthe
cylinder75%full.Forexample,thecylinderholds1000gofwater(1000
liquid chlorine will flow into the cell and that the valves on all
mL,assumingaspecificgravityof1.0).Thefilledcylinderwillhold1560
g of chlorine, and at 75% of capacity, it will contain 1170 g of chlorine. parts of the apparatus are closed prior to attempting to fill the
cell. For the following operations, refer to 7.1.4 on venting
8.3 Thoroughlydrythesamplecylindersbyplacingthemin
chlorine. Repeat 9.4 – 9.6.
an oven at 105°C for at least6hor preferably overnight. The
unheated dip tubes are placed in a desiccator. Valves also are 9.8 Fill the cell by opening Valve C. Close Valve C and
placed in the oven but not as a part of the cylinder (packings invertthecelltoensurecompletefillingofthecellwindowand
are resistant to this temperature). Af
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E1786 − 17 E1786 − 17a
Standard Test Method for
Determination of Low Levels of Water in Liquid Chlorine by
On-Line Infrared Spectrophotometry
This standard is issued under the fixed designation E1786; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This test method is designed for the on-line determination of the content of water in liquid chlorine in the concentration
range of 0.5 to 15 mg/kg (ppm).
1.2 The values stated in SI units are to be regarded as standard. The values given in parentheses are for information only.
1.3 Review the current Safety Data Sheets (SDS) for detailed information concerning toxicity, first aid procedures, and safety
precautions.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory
limitations prior to use. Specific hazards statements are given in Section 7 and Note 3.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D1193 Specification for Reagent Water
D6809 Guide for Quality Control and Quality Assurance Procedures for Aromatic Hydrocarbons and Related Materials
E806 Test Method for Carbon Tetrachloride and Chloroform in Liquid Chlorine by Direct Injection (Gas Chromatographic
Procedure)
2.2 Federal Standards:
49 CFR 173 Code of Federal Regulations Title 49 Transportation: Shippers’ General Requirements for Shipments and
Packaging, including the following sections:
173.304 Charging of Cylinders with Liquefied Compressed Gas
173.314 Requirements for Compressed Gases in Tank Cars
173.315 Compressed Gases in Cargo Tanks and Portable Tank Containers
2.3 Other Document:
Chlorine Institute Pamphlet No. 1 — Chlorine Basics
3. Summary of Test Method
3.1 Liquid chlorine continuously flows through a special infrared cell where it is maintained as a liquid under its own pressure.
A process infrared spectrometer scans from 400 to 4400 wavenumbers of the infrared transmission spectrum of liquid chlorine.
This test method is under the jurisdiction of ASTM Committee D16 on Aromatic Hydrocarbons Aromatic, Industrial, Specialty and Related Chemicals and is the direct
responsibility of Subcommittee D16.16 on Industrial and Specialty Product Standards.
Current edition approved March 1, 2017July 1, 2017. Published March 2017July 2017. Originally approved in 1996. Last previous edition approved in 20082017 as
E1786 – 08.E1786 – 17. DOI: 10.1520/E1786-17.10.1520/E1786-17a.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Code of Federal Regulations, Available from U.S. Government Printing Office, Superintendent of Documents, 732 N. Capitol St., NW, Washington, DC 20401-0001,
http://www.access.gpo.gov.
Available from The Chlorine Institute, Inc., 1300 Wilson Blvd., Suite 525, Arlington, VA 22209, https://www.chlorineinstitute.org.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E1786 − 17a
This spectrum then is ratioed to one obtained from the nitrogen-filled infrared cell previously. The ratioed spectrum is converted
to absorbance, and the net absorbance of water band at 1596 wavenumbers, relative to a reference at 1663 wavenumbers, is
determined.
3.2 The amount of water corresponding to this net absorbance is determined from a calibration curve prepared from the infrared
absorbencies of standards which contain concentrations of water in liquid chlorine. These standards are prepared from manual
samples of liquid chlorine in tantalum cylinders. Sample from each cylinder is introduced into a calibration infrared cell and
maintained as a liquid under its own pressure.
4. Significance and Use
4.1 Trace amounts of water may be detrimental to the use of chlorine in some applications. The amount of water in the chlorine
must be known to prevent problems during its use.
5. Apparatus
5.1 Process Infrared Spectrometer, capable of measurements in the 1600 wavenumber region. An FTIR with four wavenumber
resolution is the instrument of choice, but dispersive instruments also may be used to achieve similar results.
5.2 Special Infrared Calibration Cell (Fig. 1), as used for calibration. Neither cell size nor pathlength are critical to the analysis,
but sensitivity and limit of detection are dependent on pathlength. The concentration range reported in the Section 1 is achievable
with a 60-mm pathlength cell constructed with the following: Figs. 2-7
5.2.1 Hastelloy C and 316 Stainless Steel Stock, suitable for machining,
5.2.2 Silver Chloride Windows, 0.5 cm × 2.5 cm, and
5.2.3 Perfluoroelastomer Sheet, 0.762 mm (0.030 in.) thickness.
1 1
5.3 Ball Valves, Monel 6.35 mm ( ⁄4-in.) valve with pipe and 6.35 mm ( ⁄4-in.) tube ends.
1 1
5.4 Needle Valves, Nickel or Monel 6.35 mm ( ⁄4-in.) valve with pipe and 6.35 mm ( ⁄4-in.) tube ends.
5.5 Sample Cylinder Assembly (Fig. 8), consisting of:
FIG. 1 Infrared Cell (Drawing Not to Scale)
E1786 − 17a
FIG. 2 Pipe Adapter
FIG. 3 Air Cap Detail
5.5.1 Sample Cylinder, nickel, Monel, or tantalum, 400 to 1000-mL capacity, double-ended, with valves at each end, specially
cleaned. Cylinders with both valves at one end and with a dip tube on one valve have been found to be satisfactory. Another option
is to construct special cylinders containing a septum fitting on one end.
NOTE 1—A procedure for cleaning cylinders and valves, for use with liquid chlorine, is given in Test Method E806, Appendix X2.
5.5.2 One Needle and One Ball Valve, nickel body, having packing resistant to liquid chlorine. If nickel valves are not available,
monel valves may be used.
5.5.3 Septum, inserted into a 6.35 mm ( ⁄4-in.)-in. nut.
5.5.4 Glove Bag or Dry Box, purged with dry nitrogen (less than 5 mg/kg (ppm) water vapor).
5.5.5 Fittings, for transferring chlorine from one cylinder to another.
5.5.6 One 0 to 10 μL Syringe and One 0 to 25 μL Syringe, 26 gage needle.
5.5.7 Dewar Flask, of sufficient size to hold a cylinder surrounded by dry ice and methylene chloride. The Dewar flask should
be supported by a wooden holder for safety purposes.
5.5.8 Hygrometer, capable of measuring moisture as low as 5 mg/kg (ppm) in glove bag or dry box.
5.6 Silicone Rubber Septa.
E1786 − 17a
FIG. 4 Body Detail
FIG. 5 Gasket Detail
5.7 Mechanical Shaker.
5.8 Drying Oven.
5.9 Special Infrared Process Cell (Fig. 9) for on-line analysis of water in liquid chlorine. Neither cell size nor path length is
critical to the analysis, but sensitivity and limit of detection are dependent on pathlength. The concentration range reported in the
scope is achievable with a 60-mm pathlength cell constructed with:
5.9.1 Hastelloy C and 316 Stainless Steel Stock, suitable for machining.
5.9.2 Silver Chloride Windows, two, 25-mm diameter by 2-mm thick and two 25-mm diameter by 4-mm thick.
E1786 − 17a
FIG. 6 Insert Detail
FIG. 7 Flange Detail
5.9.3 Eight Viton O-rings, Size 027.
6. Reagents
6.1 Purity of Water—Unless otherwise indicated, water means Type II or III reagent water conforming to Specification D1193.
6.2 Chlorine, liquid with less than 5 mg/kg (ppm) water.
6.3 Methylene Chloride (CH Cl ).
2 2
NOTE 2—This reagent is used for cooling purposes only.
6.4 Dry Ice (CO ).
6.5 Dry Nitrogen (<5 mg/kg (ppm) water), to purge glove bag or dry box and test equipment.
7. Hazards
7.1 Safety Precautions:
7.1.1 Chlorine is a corrosive and toxic material. Use a well-ventilated fume hood to house all test equipment, except the infrared
spectrophotometer, when this material is analyzed in the laboratory.
E1786 − 17a
FIG. 8 Sample Cylinder Assembly
FIG. 9 Special Infrared Process Cell
7.1.2 Persons who are thoroughly familiar with the handling of chlorine should perform this analysis. An experienced person
should not work alone. The analyst must be provided with adequate eye protection (chemical goggles are recommended) and an
approved chlorine respirator. Splashes of liquid chlorine destroy clothing, and if such clothing is next to the skin, will produce
irritation and burns.
7.1.3 When sampling and working with chlorine out of doors, warn people downwind from such operations of the possible
release of chlorine.
7.1.4 Dispose of excess chlorine in an environmentally safe and acceptable manner. If chlorine cannot be disposed of in a
chlorine consuming process, provide a chlorine absorption system. When the analysis and sampling regimen requires an initial
purging of chlorine from a container, the purged chlorine should be similarly handled. Avoid purging to the atmosphere.
E1786 − 17a
7.1.5 In the event chlorine is inhaled, use first aid immediately.
8. Sampling for Calibration Standards
8.1 Carefully choose sampling points. Ensure that the sample point is associated with flowing chlorine and is not near a “dead
leg” where the concentrations of impurities in the chlorine will never change because the chlorine never moves. If sampling
through secondary piping, purge that piping well with nitrogen or dry air before being blocked in. Otherwise, temperature
variations can result in water vapor condensing inside the piping to contaminate the chlorine sample when it is grabbed.
8.1.1 Finally, perform sampling at a sample point representative of the chlorine needing to be analyzed; that is, sample pure
chlorine after all purification steps, drying steps, and so forth, to ensure that the analytical results are meaningful.
8.1.1.1 Sampling from tank cars, barges, storage tanks, and large cylinders presents unique problems. Each facility, however,
must be capable of delivering a liquid sample (not gas). See Chlorine Institute Pamphlet No. 1. (See 49 CFR 173, including Parts
173.304, 173.314, and 173.315.)
8.2 Collect samples from these facilities in the sample cylinder assembly listed in 6.5. Follow proper and safe sampling
techniques. The cylinder must contain at least 75 % by volume of liquid chlorine (less than 25 % vapor space).
NOTE 3—Do not allow the cylinder to become liquid-full. Tare the cylinder, fill with water, and reweigh to determine the weight of water in the cylinder.
Multiply this weight by 1.56 (the assumed specific gravity of liquid chlorine) and by 0.75 to obtain the weight of chlorine to fill the cylinder 75 % full.
For example, the cylinder holds 1000 g of water (1000 mL, assuming a specific gravity of 1.0). The filled cylinder will hold 1560 g of chlorine, and at
75 % of capacity, it will contain 1170 g of chlorine.
8.3 Thoroughly dry the sample cylinders by placing them in an oven at 105°C for at least 6 h or preferably overnight. The
unheated dip tubes are placed in a desiccator. Valves also are placed in the oven but not as a part of the cylinder (packings are
resistant to this temperature). After this treatment, the cylinders are cooled with plant air or N having <5 mg/kg (ppm) moisture.
The valves are removed, placed in a desiccator with a suitable drying agent, and cooled to room temperature.
9. Preparation of Standards for Calibration
9.1 Obtain four clean, evacuated sample cylinders. Use only cylinders that have been properly pressure-tested. Equip three with
a ball valve and a needle valve, label these Cylinders No. 2 through No. 4, and record weight to the nearest gram. Equip the fourth
cylinder with two needle valves, weigh to the nearest gram, record, and label No. 1. Once it contains chlorine, it will be used to
purge the infrared cell before standards are loaded. Use caps on all valves. If dip tubes are used, attach the dip tube to the needle
valve. Check the hygrometer to make sure the atmosphere in the glove bag or dry box contains <5 mg/kg (ppm) moisture before
performing any transfers of chlorine.
9.2 Load the four cylinders with liquid chlorine. The liquid chlorine should contain less than 5 mg/kg (ppm) water. All cylinders
must contain at least 75 % by volume of liquid chlorine (less than 25 % vapor space). All cylinders should be weighed to the
nearest gram and recorded to determine the weight of chlorine in each cylinder (Note 3). Place the filled cylinders into the glove
bag or dry box inside the hood. Place all fittings, tools and equipment, including the purged infrared cell, in the glove bag or dry
box. Wait until the hygrometer in the glove bag or dry box is reading <5 mg/kg (ppm) moisture before proceeding.
9.3 Remove the cap on Valve A of Cylinder No. 1 and blow with N to remove any trapped moisture. Do this each time a
connection is made. Then connect the cylinder and the special infrared cell to the filling apparatus (Fig. 8) in such a way that the
liquid chlorine will flow into the cell and the valves on all parts of the apparatus are closed prior to filling the cell. For the following
operations, refer to 7.4 on venting chlorine.
9.4 Open Valve F and then Valve A. Flush the filling apparatus by partially opening Valve B for a few s and then closing it.
Leave Valve A open.
9.4.1 Open Valves E and C. Flush the cell by partially opening Valve D and observing flow, then close Valve C and then Valve
D.
9.5 Fill the infrared cell by opening Valve C and observe the filling of the cell windows. Close Valve C and empty the cell by
opening Valve D. Close Valve D.
9.6 Fill and empty the cell once more, as described above.
9.6.1 Close Valve A and open Valve B, to purge the filling apparatus, then close.
9.7 Remove the cylinder. Connect Cylinder No. 2 to the filling apparatus after blowing Valve A with N . Make sure that the
cylinder is connected to the apparatus in such a way that the liquid chlorine will flow into the cell and that the valves on all parts
of the apparatus are closed prior to attempti
...




Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...