ASTM F2347-15
(Guide)Standard Guide for Characterization and Testing of Hyaluronan as Starting Materials Intended for Use in Biomedical and Tissue Engineered Medical Product Applications
Standard Guide for Characterization and Testing of Hyaluronan as Starting Materials Intended for Use in Biomedical and Tissue Engineered Medical Product Applications
SIGNIFICANCE AND USE
4.1 This guide contains a listing of those characterization parameters that are directly related to the functionality of hyaluronan. This guide can be used as an aid in the selection and characterization of the appropriate hyaluronan for a particular application. This guide is intended to give guidance in the methods and types of testing necessary to properly characterize, assess, and ensure consistency in the performance of a particular hyaluronan. It may have use in the regulation of these devices by appropriate authorities.
4.2 The hyaluronan covered by this guide may be gelled, cross-linked, extruded, or otherwise formulated into biomedical devices for use in tissue engineered medical products or drug delivery devices for implantation as determined to be appropriate, based on supporting biocompatibility and physical test data. Recommendations in this guide should not be interpreted as a guarantee of clinical success in any tissue engineered medical product or drug delivery application.
4.3 To ensure that the material supplied satisfies requirements for use in TEMPs, several general areas of characterization should be considered. These are: identity of hyaluronan, physical and chemical characterization and testing, impurities profile, and performance-related tests.
SCOPE
1.1 This guide covers the evaluation of hyaluronan suitable for use in biomedical or pharmaceutical applications, or both, including, but not limited to, Tissue Engineered Medical Products (TEMPs).
1.2 This guide addresses key parameters relevant to the characterization and purity of hyaluronan.
1.3 As with any material, some characteristics of hyaluronan may be altered by processing techniques, such as cross-linking and sterilization, required for the production of a specific formulation or device. Therefore, properties of fabricated forms of this polymer should be evaluated using test methods that are appropriate to ensure safety and efficacy and are not addressed in this guide.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory requirements prior to use.
General Information
- Status
- Published
- Publication Date
- 30-Nov-2015
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.42 - Biomaterials and Biomolecules for TEMPs
Relations
- Effective Date
- 01-Dec-2015
- Effective Date
- 15-Mar-2020
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Oct-2018
- Effective Date
- 01-Feb-2018
- Effective Date
- 01-Mar-2017
- Effective Date
- 01-Dec-2016
- Effective Date
- 01-Apr-2016
- Refers
ASTM F895-11(2016) - Standard Test Method for Agar Diffusion Cell Culture Screening for Cytotoxicity - Effective Date
- 01-Apr-2016
- Refers
ASTM E2975-15 - Standard Test Method for Calibration of Concentric Cylinder Rotational Viscometers - Effective Date
- 01-Sep-2015
- Refers
ASTM E2975-14 - Standard Test Method for Calibration of Concentric Cylinder Rotational Viscometers - Effective Date
- 01-Aug-2014
- Effective Date
- 01-Mar-2014
- Effective Date
- 01-Dec-2013
- Effective Date
- 01-Oct-2013
- Refers
ASTM E1953-07(2013) - Standard Practice for Description of Thermal Analysis and Rheology Apparatus - Effective Date
- 01-Mar-2013
Overview
ASTM F2347-15: Standard Guide for Characterization and Testing of Hyaluronan as Starting Materials Intended for Use in Biomedical and Tissue Engineered Medical Product Applications is a comprehensive guide developed by ASTM International. This standard provides key recommendations for the selection, evaluation, and testing of hyaluronan (hyaluronic acid, hyaluronate, and their salt forms) that are intended for biomedical, pharmaceutical, and tissue-engineered medical product (TEMP) applications.
Hyaluronan is a naturally occurring biopolymer widely used due to its biocompatibility, viscoelastic properties, and ability to form hydrogels. Its applications span viscosupplements, tissue engineering scaffolds, ocular devices, dermal fillers, and drug delivery systems. ASTM F2347-15 is crucial for ensuring that hyaluronan used in medical devices and related applications meets quality and regulatory requirements.
Key Topics
ASTM F2347-15 addresses several key issues related to the characterization and testing of hyaluronan:
- Material Identity: Methods for confirming the identity of hyaluronan, considering both its physical and chemical structure.
- Physicochemical Characterization: Evaluation of molecular weight, intrinsic viscosity, polydispersity, and rheological properties.
- Impurity Profile: Assessment of potential contaminants such as endotoxins, nucleic acids, proteins, sulfated glycosaminoglycans, heavy metals, and microbiological agents.
- Performance-Related Testing: Tests that relate to the biocompatibility and functionality of hyaluronan in the intended biomedical application.
- Product Source and Processing: Importance of documenting the origin (animal-derived vs. non-animal fermentation sources) and processing techniques, as these can impact performance and regulatory acceptance.
- Stability and Sterility: Considerations for stability studies and guidance on sterilization, including effects on molecular structure and functional properties.
- Regulatory Guidance: Emphasis on complying with current safety, toxicology, and biocompatibility guidelines (e.g., FDA, ISO 10993).
Applications
Implementing ASTM F2347-15 delivers practical value across various biomedical and pharmaceutical applications, including:
- Tissue Engineering: Selection and quality control of hyaluronan for use in scaffolds, cell encapsulation, and regenerative medicine.
- Implantable Medical Devices: Ensuring consistency, purity, and biocompatibility of hyaluronan in viscosupplements (e.g., osteoarthritis treatment), ocular implants, and dermal fillers.
- Drug Delivery: Supporting the development of hyaluronan-based drug carriers or hydrogels for controlled release formulations.
- Regulatory Submissions: Providing a framework for product dossiers and master files to facilitate regulatory approval processes.
- Quality Assurance: Enabling manufacturers and researchers to standardize testing, ensuring batch-to-batch consistency and reproducibility.
- Material Sourcing Decisions: Assisting in the comparison of hyaluronan from different origins (animal vs. microbial fermentation) to meet ethical, safety, and regulatory needs.
Related Standards
ASTM F2347-15 references and aligns with several important standards and guidance documents, including:
- USP and EP Monographs: Methods for sterility, endotoxins, microbial limits, and heavy metals.
- ASTM Standards: D2196 (rheological properties), E2975 (viscometer calibration), F748 (biological testing selection), F749 (material extracts evaluation), F756 (hemolytic properties), and others related to biocompatibility and cytotoxicity.
- ISO 10993 Series: Guidelines for biological evaluation and biocompatibility of medical devices.
- ICH Guidelines: Q1A (stability testing) and S2B (genotoxicity testing).
- AAMI Documents: Sterilization of medical devices.
- FDA Regulations: Particularly pertinent in device and drug master file submissions.
Following ASTM F2347-15 helps ensure that hyaluronan-based biomedical products meet international quality, safety, and regulatory standards, supporting innovation and patient safety in healthcare technologies.
Buy Documents
ASTM F2347-15 - Standard Guide for Characterization and Testing of Hyaluronan as Starting Materials Intended for Use in Biomedical and Tissue Engineered Medical Product Applications
REDLINE ASTM F2347-15 - Standard Guide for Characterization and Testing of Hyaluronan as Starting Materials Intended for Use in Biomedical and Tissue Engineered Medical Product Applications
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F2347-15 is a guide published by ASTM International. Its full title is "Standard Guide for Characterization and Testing of Hyaluronan as Starting Materials Intended for Use in Biomedical and Tissue Engineered Medical Product Applications". This standard covers: SIGNIFICANCE AND USE 4.1 This guide contains a listing of those characterization parameters that are directly related to the functionality of hyaluronan. This guide can be used as an aid in the selection and characterization of the appropriate hyaluronan for a particular application. This guide is intended to give guidance in the methods and types of testing necessary to properly characterize, assess, and ensure consistency in the performance of a particular hyaluronan. It may have use in the regulation of these devices by appropriate authorities. 4.2 The hyaluronan covered by this guide may be gelled, cross-linked, extruded, or otherwise formulated into biomedical devices for use in tissue engineered medical products or drug delivery devices for implantation as determined to be appropriate, based on supporting biocompatibility and physical test data. Recommendations in this guide should not be interpreted as a guarantee of clinical success in any tissue engineered medical product or drug delivery application. 4.3 To ensure that the material supplied satisfies requirements for use in TEMPs, several general areas of characterization should be considered. These are: identity of hyaluronan, physical and chemical characterization and testing, impurities profile, and performance-related tests. SCOPE 1.1 This guide covers the evaluation of hyaluronan suitable for use in biomedical or pharmaceutical applications, or both, including, but not limited to, Tissue Engineered Medical Products (TEMPs). 1.2 This guide addresses key parameters relevant to the characterization and purity of hyaluronan. 1.3 As with any material, some characteristics of hyaluronan may be altered by processing techniques, such as cross-linking and sterilization, required for the production of a specific formulation or device. Therefore, properties of fabricated forms of this polymer should be evaluated using test methods that are appropriate to ensure safety and efficacy and are not addressed in this guide. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory requirements prior to use.
SIGNIFICANCE AND USE 4.1 This guide contains a listing of those characterization parameters that are directly related to the functionality of hyaluronan. This guide can be used as an aid in the selection and characterization of the appropriate hyaluronan for a particular application. This guide is intended to give guidance in the methods and types of testing necessary to properly characterize, assess, and ensure consistency in the performance of a particular hyaluronan. It may have use in the regulation of these devices by appropriate authorities. 4.2 The hyaluronan covered by this guide may be gelled, cross-linked, extruded, or otherwise formulated into biomedical devices for use in tissue engineered medical products or drug delivery devices for implantation as determined to be appropriate, based on supporting biocompatibility and physical test data. Recommendations in this guide should not be interpreted as a guarantee of clinical success in any tissue engineered medical product or drug delivery application. 4.3 To ensure that the material supplied satisfies requirements for use in TEMPs, several general areas of characterization should be considered. These are: identity of hyaluronan, physical and chemical characterization and testing, impurities profile, and performance-related tests. SCOPE 1.1 This guide covers the evaluation of hyaluronan suitable for use in biomedical or pharmaceutical applications, or both, including, but not limited to, Tissue Engineered Medical Products (TEMPs). 1.2 This guide addresses key parameters relevant to the characterization and purity of hyaluronan. 1.3 As with any material, some characteristics of hyaluronan may be altered by processing techniques, such as cross-linking and sterilization, required for the production of a specific formulation or device. Therefore, properties of fabricated forms of this polymer should be evaluated using test methods that are appropriate to ensure safety and efficacy and are not addressed in this guide. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory requirements prior to use.
ASTM F2347-15 is classified under the following ICS (International Classification for Standards) categories: 11.120.99 - Other standards related to pharmaceutics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F2347-15 has the following relationships with other standards: It is inter standard links to ASTM F2347-11, ASTM E1953-20, ASTM F749-20, ASTM F1903-18, ASTM F1439-03(2018), ASTM F756-17, ASTM E2975-16, ASTM F748-16, ASTM F895-11(2016), ASTM E2975-15, ASTM E2975-14, ASTM E1953-14, ASTM F756-13, ASTM F1439-03(2013), ASTM E1953-07(2013). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F2347-15 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F2347 − 15
Standard Guide for
Characterization and Testing of Hyaluronan as Starting
Materials Intended for Use in Biomedical and Tissue
Engineered Medical Product Applications
This standard is issued under the fixed designation F2347; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
INTRODUCTION
Hyaluronan, which in this guide will encompass hyaluronic acid, hyaluronate, and its salt forms, is
the simplest of the glycosaminoglycans. Hyaluronan is soluble in water and forms highly viscous
solutions.Hyaluronanisfoundinubiquitouslyinthebodyaspartoftheextracellularmatrixoftissues,
with high concentrations in the synovial fluid, vitreous humor, and skin, as well as in cartilage.
Hyaluronan has found uses in a variety of products ranging from viscosupplements (treatment of
osteoarthritis), adhesion prevention (prevention of post-surgical adhesions), viscoelastics (ocular
protection), and dermal implants (lip augmentation and wrinkle removal). New applications, such as
scaffolds for tissue engineering, are emerging. The aim of this guide is to identify key parameters
relevant to the characterization of hyaluronan for the development of new commercial applications of
hyaluronan for the biomedical and pharmaceutical industries.
1. Scope* priate safety and health practices and determine the applica-
bility of regulatory requirements prior to use.
1.1 This guide covers the evaluation of hyaluronan suitable
for use in biomedical or pharmaceutical applications, or both,
2. Referenced Documents
including, but not limited to, Tissue Engineered Medical
2.1 ASTM Standards:
Products (TEMPs).
E1953 Practice for Description of Thermal Analysis and
1.2 This guide addresses key parameters relevant to the
Rheology Apparatus
characterization and purity of hyaluronan.
E2975 Test Method for Calibration of Concentric Cylinder
Rotational Viscometers
1.3 As with any material, some characteristics of hyaluro-
nan may be altered by processing techniques, such as cross- F619 Practice for Extraction of Medical Plastics
F748 PracticeforSelectingGenericBiologicalTestMethods
linking and sterilization, required for the production of a
specific formulation or device. Therefore, properties of fabri- for Materials and Devices
F749 Practice for Evaluating Material Extracts by Intracuta-
cated forms of this polymer should be evaluated using test
methods that are appropriate to ensure safety and efficacy and neous Injection in the Rabbit
F756 Practice for Assessment of Hemolytic Properties of
are not addressed in this guide.
Materials
1.4 The values stated in SI units are to be regarded as
F763 Practice for Short-Term Screening of Implant Materi-
standard. No other units of measurement are included in this
als
standard.
F813 Practice for Direct Contact Cell Culture Evaluation of
1.5 This standard does not purport to address all of the
Materials for Medical Devices
safety concerns, if any, associated with its use. It is the
F895 TestMethodforAgarDiffusionCellCultureScreening
responsibility of the user of this standard to establish appro-
for Cytotoxicity
F981 Practice for Assessment of Compatibility of Biomate-
rials for Surgical Implants with Respect to Effect of
This guide is under the jurisdiction of ASTM Committee F04 on Medical and
Surgical Materials and Devicesand is the direct responsibility of Subcommittee
F04.42 on Biomaterials and Biomolecules for TEMPs. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved Dec. 1, 2015. Published February 2016. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 2003. Last previous edition approved in 2011 as F2347 – 11. DOI: Standards volume information, refer to the standard’s Document Summary page on
10.1520/F2347-15. the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2347 − 15
Materials on Muscle and Bone ICH Q1A Harmonized Tripartite Guidance for Stability
F1439 Guide for Performance of Lifetime Bioassay for the TestingofNewDrugSubstancesandProducts(September
Tumorigenic Potential of Implant Materials 2001, Revision 1)
F1903 Practice for Testing For Biological Responses to FDA Guideline on Validation of the Limulus Amebocyte
Particles In Vitro Test as an End-Product Endotoxin Test for Human and
F1904 Practice for Testing the Biological Responses to Animal Parenteral Drugs, Biological Products and Health-
Particles in vivo care Products, DHHS, December 1987
2.2 USP Documents: FDA Interim Guidance for Human and Veterinary Drug
USP <61> Microbial Limit Tests Products and Biologicals, Kinetic LAL Techniques,
USP <71> Sterility Tests DHHS, July 15, 1991
USP <85> Bacterial Endotoxins Tests AAMI TIR No. 7: 1999 Chemical Sterilants and High Level
USP <231> Heavy Metals Disinfectants: A Guide to Selection and Use
USP <731> Loss on Drying AAMI ST67/CDV–2: 1999 Sterilization of Medical
USP <1211> Sterilization and Sterility Assurance of Com- Devices—Requirements for Products Labeled “Sterile”
pendial Articles 21 CFR 312 FDATitle 21, Food and Drugs, Investigational
4 9
2.3 EP Documents: New Drug Applications
EP Monograph 1472 Sodium Hyaluronate
3. Terminology
EP 2.6.1 Sterility
3.1 Definitions:
2.4 Other Referenced Documents:
3.1.1 decomposition, n—structural changes of hyaluronan
ISO 10993 Biological Evaluation of Medical Devices
duetoexposuretoenvironmental,chemical,orthermalfactors.
ISO 10993-1 Biological Evaluation of Medical Devices—
Decomposition may occur at temperatures as low as 121°C
Part 1: Evaluation and Testing
during autoclaving. Decomposition can result in deleterious
ISO 10993-7 Biological Evaluation of Medical Devices—
changes to the hyaluronan.
Part 7: Ethylene Oxide Sterilization Residuals
ISO 10993-9 Biological Evaluation of Medical Devices—
3.1.2 degradation, n—change in the chemical structure,
Part 9: Framework for Identification and Quantification of
physical properties or appearance of a material. Degradation of
Potential Degradation Products
polysaccharides occurs via cleavage of the glycosidic bonds,
ISO 10993-17 Biological Evaluation of Medical Devices—
usually by acid catalyzed hydrolysis. Degradation can also
Part 17: Establishment ofAllowable Limits for Leachable
occur thermally and by alkali. It is important to note that
Substances
degradation is not synonymous with decomposition. Degrada-
ISO 14160: 1998 Sterilization of Single-Use Medical De-
tion is often used as a synonym for depolymerization when
vices Incorporating Materials of Animal Origin—
referring to polymers. Degradation (depolymerization) of hy-
Validation and Routine Control of Sterilization by Liquid
aluronan may also occur enzymatically by the action of
Chemical Sterilants
hyaluronidases.
ISO 11737-1: 1995 Sterilization of Medical Devices—
3.1.3 depolymerization, n—reduction in length of a polymer
Microbiological Methods—Part 1: Estimation of Popula-
chain to form shorter polymeric units. Depolymerization may
tion of Microorganisms on Products
reduce the polymer chain to smaller molecular weight
ISO 11737-2: 1998 Sterilization of Medical Devices—
polymers, oligomeric, or monomeric units, or combination
Microbiological Methods—Part 2: Tests of Sterility Per-
thereof. In hyaluronan, acid hydrolysis of the glycosidic bonds
formed in the Validation of a Sterilization Process
is the primary mechanism.
ISO 13408-1: 1998 Aseptic Processing of Health Care
5 3.1.4 endotoxin,n—pyrogenichighmolarmasslipopolysac-
Products—Part 1: General Requirements
charide (LPS) complex associated with the cell wall of
ISO EN 12442-1 Animal Tissues and Their Derivative
gram-negative bacteria.
Utilized in the Manufacture of Medical Devices—Part 1:
5 3.1.4.1 Discussion—Though endotoxins are pyrogens, not
Analysis and Management of Risk
all pyrogens are endotoxins. Endotoxins are specifically de-
ISO EN 12442-3 Animal Tissues and Their Derivative
tected through a Limulus Amebocyte Lysate (LAL) test.
Utilized in the Manufacture of Medical Devices—Part 3:
3.1.5 hyaluronan, n—a polysaccharide with a disaccharide
Validation of the Elimination and/or inactivation of Virus
repeating unit composed of D-glucuronic acid and N-acetyl-
and Transmissible Agents
D-glucosamine in β-(1→3) linkage. Each disaccharide unit is
ICH S2B A Standard Battery for Genotoxicity Testing of
attached to the next byβ-(1→4) bonds. Hyaluronan is a linear
Pharmaceuticals (July 1997)
polymer.Othercommonnamesarehyaluronicacidandsodium
hyaluronate.
Available from U.S. Pharmacopeia (USP), 12601 Twinbrook Pkwy., Rockville,
MD 20852.
Available from European Directorate for the Quality of Medicines (EDQM), Available from U.S. Food and Drug Administration, 5600 Fishers Lane,
Council of Europe, BP 907, 67029 Strasbourg, France. Rockville, MD 20857-0001.
Available fromAmerican National Standards Institute (ANSI), 25 W. 43rd St., Available from Association for the Advancement of Medical Instrumentation,
4th Floor, New York, NY 10036. 1110 North Glebe Rd., Suite 220, Arlington, VA 22201-4795.
6 9
Available from International Conference on Harmonization (ICH) Secretariat, Available from Standardization Documents Order Desk, DODSSP, Bldg. 4,
c/o IFPMA, 30 rue de St-Jean, P.O. Box 758, 1211 Geneva 13, Switzerland. Section D, 700 Robbins Ave., Philadelphia, PA 19111-5098
F2347 − 15
3.1.6 hydrocolloid, n—a water-soluble polymer of colloidal 5.1.2 Fourier Transform Infrared Spectroscopy (FT-IR)—
nature when hydrated. Almost all organic chemical compounds absorb infrared radia-
tion at frequencies characteristic for the functional groups in
3.1.7 molecular mass average (molecular weight average),
the compound. A FT-IR spectrum will show absorption bands
n—thegivenmolecularweight(Mw)ofhyaluronanwillalways
relating to bond stretching and bending and can therefore serve
represent an average of all of the molecules in the population.
as a unique fingerprint of a specific compound. Direct FT-IR
The most common ways to express the Mw are as the number
analysis of hyaluronan powder is perhaps the easiest technique
¯ ¯
average (M ) and the weight average (M ).The two averages
n w
to perform. One method utilizes a horizontal attenuated total
are defined by the following equations:
reflectance (HATR) accessory with a zinc-selenium (ZnSe)
N M w M N M
(i i i (i i i (i i i
crystal (or equivalent) having a sample trough and a pressure
H H
M 5 and M 5 5
n w
N w N M plate. Record background and sample spectra between 4000
(i i (i i (i i i
-1
and 600 cm at an appropriate resolution. Label the peaks.
where:
-1
Typical frequencies (cm ) for hyaluronan (sodium salt) are
N = number of molecules having a specific molecular
i
3275-3390(b),1615(s),1405(m),1377(m),1150,1077,1045
weight M, and
i (s), 946 (m), 893 (w). The peak designators are: sh: sharp; s:
w = weightofmoleculeshavingaspecificmolecularweight
i
strong; m: medium; w: weak; b: broad.Atypical FT-IR HATR
M.
i
spectrum is shown in Fig. 1. A reference spectrum can be
¯ ¯
InapolydispersemolecularpopulationtherelationM >M obtained from the European Pharmacopoeia.
w n
¯ ¯
is always valid. The coefficient M /M is referred to as the
w n
5.2 Physical and Chemical Characterization of Hyaluro-
polydispersity index, and will typically be in the range 1.2 to
nan:
3.0 for commercial hyaluronan.
5.2.1 The composition and sequential structure of hyaluro-
3.1.8 non-animal derived, n—a term describing the absence
nan can be determined by the following method: High-
1 13
of any animal-derived tissue, proteins, or products in the
resolution H- and C-nuclear magnetic resonance spectros-
manufacturing process.
copy (NMR). Hyaluronan should be dissolved in D O. If the
resulting solution is viscous, viscosity may be reduced by
3.1.9 pyrogen, n—any substance that produces fever when
chemical or enzymatic depolymerization. A typical H-NMR
administered parenterally.
spectrum of hyaluronan is shown below. Hyaluronan is char-
acterized by calculating parameters such as glucuronic acid:
4. Significance and Use
N-acetylglucosamine ratio. Some literature references to the
4.1 This guide contains a listing of those characterization
determination of composition and structure of hyaluronan are
parameters that are directly related to the functionality of
given in the References section (1-4).
hyaluronan. This guide can be used as an aid in the selection
5.2.2 Molecularmass(molecularweight)ofhyaluronanwill
and characterization of the appropriate hyaluronan for a
define certain performance characteristics such as viscosity or
particular application. This guide is intended to give guidance
gel strength, or both.As such and depending on the sensitivity
in the methods and types of testing necessary to properly
of a particular end use to these variations, determination of
characterize,assess,andensureconsistencyintheperformance
molecular mass directly or indirectly may be necessary. Com-
of a particular hyaluronan. It may have use in the regulation of
mercial hyaluronan is polydisperse with respect to molecular
these devices by appropriate authorities.
weight (M ). M may be expressed as the number average
w w
4.2 The hyaluronan covered by this guide may be gelled,
(M ) or the weight average (M ). Molecular weights may be
N W
cross-linked, extruded, or otherwise formulated into biomedi-
determined by methods such as, but not limited to the follow-
cal devices for use in tissue engineered medical products or
ing:
drug delivery devices for implantation as determined to be
5.2.2.1 Molecular Weight Determination Based on Intrinsic
appropriate, based on supporting biocompatibility and physical
Viscosity—The intrinsic viscosity describes a polymer’s ability
test data. Recommendations in this guide should not be
to form viscous solutions in water and is directly proportional
interpreted as a guarantee of clinical success in any tissue
to the average molecular weight of the polymer. The intrinsic
engineered medical product or drug delivery application.
viscosity is a characteristic of the polymer under specified
solvent and temperature conditions; it is independent of con-
4.3 To ensure that the material supplied satisfies require-
centration. The intrinsic viscosity (η) is directly related to the
ments for use in TEMPs, several general areas of characteriza-
molecular weight of a polymer through the Mark-Houwink-
tion should be considered. These are: identity of hyaluronan,
a
Sakurada (MHS) equation: [η]= KM . For hyaluronan, K is
physical and chemical characterization and testing, impurities
0.00057 and the exponent (a) is 0.75 at the following condi-
profile, and performance-related tests.
tions: 0.15 M NaCl in phosphate buffer, pH 7.5, 20°C (5).By
measuringtheintrinsicviscosity,theviscosityaveragemolecu-
5. Chemical and Physical Test Methods
lar weight can be determined if K and a are accurately known
5.1 Identity of Hyaluronan—The identity of hyaluronan can
be established by several methods including, but not limited to
EDQM, European Pharmacopoeia, Council of Europe, B.P. 907, F-67029
the following:
Strasbourg France; www.pheur.org
5.1.1 Sodium Hyaluronate Monograph EP Monograph
The boldface numbers in parentheses refer to the list of references at the end
1472. of this standard.
F2347 − 15
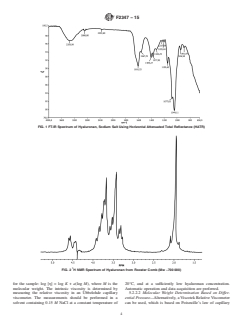
FIG. 1 FT-IR Spectrum of Hyaluronan, Sodium Salt Using Horizontal Attenuated Total Reflectance (HATR)
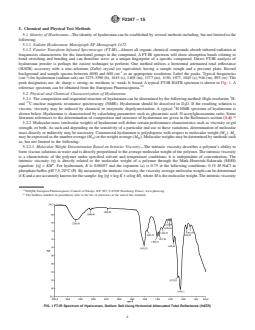
FIG. 2 H NMR Spectrum of Hyaluronan from Rooster Comb (Mw ~700 000)
for the sample: log [η] = log K + a(log M), where M is the 20°C, and at a sufficiently low hyaluronan concentration.
molecular weight. The intrinsic viscosity is determined by Automatic operation and data acquisition are preferred.
measuring the relative viscosity in an Ubbelohde capillary 5.2.2.2 Molecular Weight Determination Based on Differ-
viscometer. The measurements should be performed in a ential Pressure—Alternatively, a Viscotek Relative Viscometer
solvent containing 0.15 M NaCl at a constant temperature of can be used, which is based on Poiseuille’s law of capillary
F2347 − 15
flow: the pressure drop of a fluid flowing through a capillary is issue. In such cases, calculation of the polydispersity will be
directly proportional to the viscosity. important.Typicallythisisbetween1.2and3.0forcommercial
hyaluronan.
∆P 5ηQR
5.2.3 Depending on the final use and the required perfor-
where:
mance control, other characterization assays can include, but
∆P = the pressure drop across the capillary measured by the
are not limited to the following:
differential pressure transducer (DPT),
5.2.3.1 Viscosity in Aqueous Solution—Viscosity is defined
η = the viscosity,
as a liquid’s resistance to flow. The molecular mass of
Q = the flow rate, and
hyaluronanwilldeterminetheextenttowhichitwillthickenan
R = the resistance of the capillary.
aqueous solution. Therefore, a simple viscosity test may yield
Two capillaries are connected in series with the sample
information on the relative differences in molecular mass
injection valve located between capillary one (1) and capillary
among hyaluronan samples. To allow comparison between
two (2). The sample is injected in capillary two (2) and the
laboratories, the viscometer used must be calibrated with
pressure change is detected by the DPT. The relative viscosity
traceable standards (see Test Method E2975). The viscosity
is determined by the ratio of the pressures divided by the
measured will depend on several parameters related to how the
instrument constant K.
testing is conducted. Both rotational viscometers (Practice
E1953) and “cone on plate” rheometers may be used. Impor-
η 5 P /P K
r 2 1
tant parameters to control include, but are not limited to:
The instrument constant K is the ratio of the resistances of
(1) Temperature—The temperature at which the measure-
capillary one (1) and two (2) at the base line where both
ment is performed is critical. An increase in temperature will,
capillaries contain pure solvent. Specific viscosity, inherent
in almost every case, result in a decrease in the viscosity.
viscosity, reduced viscosity, and intrinsic viscosity values can
Consistent and controlled temperature (that is, with a standard
be calculated from relative viscosity as follows:
temperature bath) is critical to achieving reproducible results.
η 5η 2 1 Typically, the temperature used to measure viscosity can be
sp r
20°C, 25°C, or 37°C, or combination thereof.
where:
(2) Hyaluronan Concentration—The moisture content of
η = specific viscosity.
sp
the hyaluronan must be known in order to prepare correct
concentrations of hyaluronan (see 5.2.3.2).
η 5η /C
red sp
(3) Ionic Strength—The viscosity of a hyaluronan solution
where:
is sensitive to the ionic environment in which the measurement
η = reduced viscosity and C is the concentration.
red
ismade.Themostimportantaspectistokeeptheionicstrength
consistent. Typically viscosity measurements should be made
η 5 lim~η /C! as C→0
int sp
in a standardized ionic environment of known ionic strength.
where:
(4) MolecularMass—Viscositymeasurementsaresensitive
η = the intrinsic viscosity.
int to the molecular mass of hyaluronan. The following is one
suggestion concerning the measurement of hyaluronan
From the intrinsic viscosity values, molecular weight can be
viscosity,butanyappropriatemethodwouldapply.Tomeasure
calculated using the Mark-Houwink-Sakurada equation.
the apparent viscosity of hyaluronan, prepare a solution in
5.2.2.3 Molecular Weight and Polydispersity Determination
deionized water with a concentration (w/w, corrected for dry
by Size Exclusion Chromatography with Multiple Angle Laser
matter content) appropriate for the end use. The viscosity is
Light Scattering Detection (SEC-MALLS)—The method of
measured using a rotational viscometer (see Practice E1953)at
choice is to use refractive index coupled to multiple angle laser
20 6 0.2°C (or other controlled temperature) using the
light scattering detection (MALLS). For separation of the
appropriate spindle, spindle rotation speed and a temperature-
hyaluronan into different molecular weight fractions, a hydro-
controlled water bath.
philic column with the appropriate pore size is required. Such
(5) Shear Rate—Hyaluronan is sensitive to shear and the
columns include, but are not limited to those mentioned in the
viscosity may vary as a function of the shear rate.
techniques below and in Refs (6, 7). The precision of these
5.2.3.2 Dry Matter Content—Hyaluronan from various sup-
techniques must be determined as results can vary by 5 to
pliers may contain different moisture contents. The dry matter
20 %. Typical methods using these techniques include, but are
content determination is based upon the removal of water and
not limited to:
other volatile substances (such as alcohol) from the sample.
(1) Using 0.2 M NaCl as the mobile phase with separation
Normally with hyaluronan, gravimetric techniques are used.
using TSK 3000 and TSK 6000 columns.
They are adapted directly from USP <731> and utilize a
(2) Using 150 mM NaCl, 50 mM phosphate buffer as the
calibrated drying oven at 105°C, or EP2.2.32 by drying at 100
mobile phase with separation using a Biogel column.
to 110°C over diphosphorus pentoxide for 6 h.
5.2.2.4 Polydispersity—Depending on the end use and the
sensitivity of the application to the molecular mass, the
NOTE 1—Dried hyaluronan can reabsor
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F2347 − 11 F2347 − 15
Standard Guide for
Characterization and Testing of Hyaluronan as Starting
Materials Intended for Use in Biomedical and Tissue
Engineered Medical Product Applications
This standard is issued under the fixed designation F2347; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
INTRODUCTION
Hyaluronan, which in this guide will encompass hyaluronic acid, hyaluronate, and its salt forms, is
the simplest of the glycosaminoglycans. Hyaluronan is soluble in water and forms highly viscous
solutions. Hyaluronan is found in ubiquitously in the body as part of the extracellular matrix of tissues,
with high concentrations in the synovial fluid, vitreous humor, and skin, as well as in cartilage.
Hyaluronan has found uses in a variety of products ranging from viscosupplements (treatment of
osteoarthritis), adhesion prevention (prevention of post-surgical adhesions), viscoelastics (ocular
protection), and dermal implants (lip augmentation and wrinkle removal). New applications, such as
scaffolds for tissue engineering, are emerging. The aim of this guide is to identify key parameters
relevant to the characterization of hyaluronan for the development of new commercial applications of
hyaluronan for the biomedical and pharmaceutical industries.
1. Scope Scope*
1.1 This guide covers the evaluation of hyaluronan suitable for use in biomedical or pharmaceutical applications, or both,
including, but not limited to, Tissue Engineered Medical Products (TEMPs).
1.2 This guide addresses key parameters relevant to the characterization and purity of hyaluronan.
1.3 As with any material, some characteristics of hyaluronan may be altered by processing techniques, such as cross-linking and
sterilization, required for the production of a specific formulation or device. Therefore, properties of fabricated forms of this
polymer should be evaluated using test methods that are appropriate to ensure safety and efficacy and are not addressed in this
guide.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory
requirements prior to use.
2. Referenced Documents
2.1 ASTM Standards:
D2196E1953 Test Methods for Rheological Properties of Non-Newtonian Materials by Rotational ViscometerPractice for
Description of Thermal Analysis and Rheology Apparatus
E2975 Test Method for Calibration of Concentric Cylinder Rotational Viscometers
F619 Practice for Extraction of Medical Plastics
F748 Practice for Selecting Generic Biological Test Methods for Materials and Devices
F749 Practice for Evaluating Material Extracts by Intracutaneous Injection in the Rabbit
This guide is under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devicesand is the direct responsibility of Subcommittee F04.42
on Biomaterials and Biomolecules for TEMPs.
Current edition approved March 1, 2011Dec. 1, 2015. Published March 2011February 2016. Originally approved in 2003. Last previous edition approved in 20032011
as F2347 – 03.F2347 – 11. DOI: 10.1520/F2347-11.10.1520/F2347-15.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2347 − 15
F756 Practice for Assessment of Hemolytic Properties of Materials
F763 Practice for Short-Term Screening of Implant Materials
F813 Practice for Direct Contact Cell Culture Evaluation of Materials for Medical Devices
F895 Test Method for Agar Diffusion Cell Culture Screening for Cytotoxicity
F981 Practice for Assessment of Compatibility of Biomaterials for Surgical Implants with Respect to Effect of Materials on
Muscle and Bone
F1251 Terminology Relating to Polymeric Biomaterials in Medical and Surgical Devices (Withdrawn 2012)
F1439 Guide for Performance of Lifetime Bioassay for the Tumorigenic Potential of Implant Materials
F1903 Practice for Testing For Biological Responses to Particles In Vitro
F1904 Practice for Testing the Biological Responses to Particles in vivo
F1905 Practice For Selecting Tests for Determining the Propensity of Materials to Cause Immunotoxicity (Withdrawn 2011)
F1906 Practice for Evaluation of Immune Responses In Biocompatibility Testing Using ELISA Tests, Lymphocyte Proliferation,
and Cell Migration (Withdrawn 2011)
2.2 USP Documents:
USP <61> Microbial Limit Tests
USP <71> Sterility Tests
USP <85> Bacterial Endotoxins Tests
USP <231> Heavy Metals
USP <731> Loss on Drying
USP <1211> Sterilization and Sterility Assurance of Compendial Articles
2.3 EP Documents:
EP Monograph 1472 Sodium Hyaluronate
EP 2.6.1 Sterility
2.4 Other Referenced Documents:
ISO 10993 Biological Evaluation of Medical Devices
ISO 10993-1 Biological Evaluation of Medical Devices—Part 1: Evaluation and Testing
ISO 10993-7 Biological Evaluation of Medical Devices—Part 7: Ethylene Oxide Sterilization Residuals
ISO 10993-9 Biological Evaluation of Medical Devices—Part 9: Framework for Identification and Quantification of Potential
Degradation Products
ISO 10993-17 Biological Evaluation of Medical Devices—Part 17: Establishment of Allowable Limits for Leachable Substances
ISO 14160: 1998 Sterilization of Single-Use Medical Devices Incorporating Materials of Animal Origin—Validation and
Routine Control of Sterilization by Liquid Chemical Sterilants
ISO 11737-1: 1995 Sterilization of Medical Devices—Microbiological Methods—Part 1: Estimation of Population of
Microorganisms on Products
ISO 11737-2: 1998 Sterilization of Medical Devices—Microbiological Methods—Part 2: Tests of Sterility Performed in the
Validation of a Sterilization Process
ISO 13408-1: 1998 Aseptic Processing of Health Care Products—Part 1: General Requirements
ISO EN 12442-1 Animal Tissues and Their Derivative Utilized in the Manufacture of Medical Devices—Part 1: Analysis and
Management of Risk
ISO EN 12442-3 Animal Tissues and Their Derivative Utilized in the Manufacture of Medical Devices—Part 3: Validation of
the Elimination and/or inactivation of Virus and Transmissible Agents
ICH S2B A Standard Battery for Genotoxicity Testing of Pharmaceuticals (July 1997)
ICH Q1A Harmonized Tripartite Guidance for Stability Testing of New Drug Substances and Products (September 2001,
Revision 1)
FDA Guideline on Validation of the Limulus Amebocyte Test as an End-Product Endotoxin Test for Human and Animal
Parenteral Drugs, Biological Products and Healthcare Products, DHHS, December 1987
FDA Interim Guidance for Human and Veterinary Drug Products and Biologicals, Kinetic LAL Techniques, DHHS, July 15,
AAMI TIR No. 7: 1999 Chemical Sterilants and High Level Disinfectants: A Guide to Selection and Use
AAMI ST67/CDV–2: 1999 Sterilization of Medical Devices—Requirements for Products Labeled “Sterile”
21 CFR 312 FDA Title 21, Food and Drugs, Investigational New Drug Applications
Available from U.S. Pharmacopeia (USP), 12601 Twinbrook Pkwy., Rockville, MD 20852.
Available from European Directorate for the Quality of Medicines (EDQM), Council of Europe, BP 907, 67029 Strasbourg, France.
Available from American National Standards Institute (ANSI), 25 W. 43rd St., 4th Floor, New York, NY 10036.
Available from International Conference on Harmonization (ICH) Secretariat, c/o IFPMA, 30 rue de St-Jean, P.O. Box 758, 1211 Geneva 13, Switzerland.
Available from U.S. Food and Drug Administration, 5600 Fishers Lane, Rockville, MD 20857-0001.
Available from Association for the Advancement of Medical Instrumentation, 1110 North Glebe Rd., Suite 220, Arlington, VA 22201-4795.
Available from Standardization Documents Order Desk, DODSSP, Bldg. 4, Section D, 700 Robbins Ave., Philadelphia, PA 19111-5098
F2347 − 15
3. Terminology
3.1 Definitions:
3.1.1 decomposition, n—structural changes of hyaluronan due to exposure to environmental, chemical, or thermal factors.
Decomposition may occur at temperatures as low as 121°C during autoclaving. Decomposition can result in deleterious changes
to the hyaluronan.
3.1.2 degradation, n—change in the chemical structure, physical properties or appearance of a material. Degradation of
polysaccharides occurs via cleavage of the glycosidic bonds, usually by acid catalyzed hydrolysis. Degradation can also occur
thermally and by alkali. It is important to note that degradation is not synonymous with decomposition. Degradation is often used
as a synonym for depolymerization when referring to polymers. Degradation (depolymerization) of hyaluronan may also occur
enzymatically by the action of hyaluronidases.
3.1.3 depolymerization, n—reduction in length of a polymer chain to form shorter polymeric units. Depolymerization may
reduce the polymer chain to smaller molecular weight polymers, oligomeric, or monomeric units, or combination thereof. In
hyaluronan, acid hydrolysis of the glycosidic bonds is the primary mechanism.
3.1.4 endotoxin, n—pyrogenic high molar mass lipopolysaccharide (LPS) complex associated with the cell wall of
gram-negative bacteria.
3.1.4.1 Discussion—
Though endotoxins are pyrogens, not all pyrogens are endotoxins. Endotoxins are specifically detected through a Limulus
Amebocyte Lysate (LAL) test.
3.1.5 hyaluronan, n—a polysaccharide with a disaccharide repeating unit composed of D-glucuronic acid and N-acetyl-D-
glucosamine in β-(1→3) linkage. Each disaccharide unit is attached to the next by β-(1→4) bonds. Hyaluronan is a linear polymer.
Other common names are hyaluronic acid and sodium hyaluronate.
3.1.6 hydrocolloid, n—a water-soluble polymer of colloidal nature when hydrated.
3.1.7 molecular mass average (molecular weight average), n—the given molecular weight (Mw) of hyaluronan will always
represent an average of all of the molecules in the population. The most common ways to express the Mw are as the number
average (M¯ ) and the weight average (M¯ ). The two averages are defined by the following equations:
n w
N M w M N M
(i i i (i i i (i i i
H H
M 5 and M 5 5
n w
N w N M
i i i i
(i (i (i
where:
N = number of molecules having a specific molecular weight M , and
i i
w = weight of molecules having a specific molecular weight M .
i i
In a polydisperse molecular population the relation M¯ > M¯ is always valid. The coefficient M¯ / M¯ is referred to as the
w n w n
polydispersity index, and will typically be in the range 1.2 to 3.0 for commercial hyaluronan.
3.1.8 non-animal derived, n—a term describing the absence of any animal-derived tissue, proteins, or products in the
manufacturing process.
3.1.9 pyrogen, n—any substance that produces fever when administered parenterally.
4. Significance and Use
4.1 This guide contains a listing of those characterization parameters that are directly related to the functionality of hyaluronan.
This guide can be used as an aid in the selection and characterization of the appropriate hyaluronan for a particular application.
This guide is intended to give guidance in the methods and types of testing necessary to properly characterize, assess, and ensure
consistency in the performance of a particular hyaluronan. It may have use in the regulation of these devices by appropriate
authorities.
4.2 The hyaluronan covered by this guide may be gelled, cross-linked, extruded, or otherwise formulated into biomedical
devices for use in tissue engineered medical products or drug delivery devices for implantation as determined to be appropriate,
based on supporting biocompatibility and physical test data. Recommendations in this guide should not be interpreted as a
guarantee of clinical success in any tissue engineered medical product or drug delivery application.
4.3 To ensure that the material supplied satisfies requirements for use in TEMPs, several general areas of characterization should
be considered. These are: identity of hyaluronan, physical and chemical characterization and testing, impurities profile, and
performance-related tests.
F2347 − 15
5. Chemical and Physical Test Methods
5.1 Identity of Hyaluronan—The identity of hyaluronan can be established by several methods including, but not limited to the
following:
5.1.1 Sodium Hyaluronate Monograph EP Monograph 1472.
5.1.2 Fourier Transform Infrared Spectroscopy (FT-IR)—Almost all organic chemical compounds absorb infrared radiation at
frequencies characteristic for the functional groups in the compound. A FT-IR spectrum will show absorption bands relating to
bond stretching and bending and can therefore serve as a unique fingerprint of a specific compound. Direct FT-IR analysis of
hyaluronan powder is perhaps the easiest technique to perform. One method utilizes a horizontal attenuated total reflectance
(HATR) accessory with a zinc-selenium (ZnSe) crystal (or equivalent) having a sample trough and a pressure plate. Record
-1
background and sample spectra between 4000 and 600 cm at an appropriate resolution. Label the peaks. Typical frequencies
-1
(cm ) for hyaluronan (sodium salt) are 3275-3390 (b), 1615 (s), 1405 (m), 1377 (m), 1150, 1077, 1045 (s), 946 (m), 893 (w). The
peak designators are: sh: sharp; s: strong; m: medium; w: weak; b: broad. A typical FT-IR HATR spectrum is shown in Fig. 1. A
reference spectrum can be obtained from the European Pharmacopoeia.
5.2 Physical and Chemical Characterization of Hyaluronan:
5.2.1 The composition and sequential structure of hyaluronan can be determined by the following method: High-resolution H-
and C-nuclear magnetic resonance spectroscopy (NMR). Hyaluronan should be dissolved in D O. If the resulting solution is
viscous, viscosity may be reduced by chemical or enzymatic depolymerization. A typical H-NMR spectrum of hyaluronan is
shown below. Hyaluronan is characterized by calculating parameters such as glucuronic acid: N-acetylglucosamine ratio. Some
literature references to the determination of composition and structure of hyaluronan are given in the References section (1-4).
5.2.2 Molecular mass (molecular weight) of hyaluronan will define certain performance characteristics such as viscosity or gel
strength, or both. As such and depending on the sensitivity of a particular end use to these variations, determination of molecular
mass directly or indirectly may be necessary. Commercial hyaluronan is polydisperse with respect to molecular weight (M ). M
w w
may be expressed as the number average (M ) or the weight average (M ). Molecular weights may be determined by methods such
N W
as, but not limited to the following:
5.2.2.1 Molecular Weight Determination Based on Intrinsic Viscosity—The intrinsic viscosity describes a polymer’s ability to
form viscous solutions in water and is directly proportional to the average molecular weight of the polymer. The intrinsic viscosity
is a characteristic of the polymer under specified solvent and temperature conditions; it is independent of concentration. The
intrinsic viscosity (η) is directly related to the molecular weight of a polymer through the Mark-Houwink-Sakurada (MHS)
a
equation: [η] = KM . For hyaluronan, K is 0.00057 and the exponent (a) is 0.75 at the following conditions: 0.15 M NaCl in
phosphate buffer, pH 7.5, 20°C (5). By measuring the intrinsic viscosity, the viscosity average molecular weight can be determined
if K and a are accurately known for the sample: log [η] = log K + a(log M), where M is the molecular weight. The intrinsic viscosity
EDQM, European Pharmacopoeia, Council of Europe, B.P. 907, F-67029 Strasbourg France; www.pheur.org
The boldface numbers in parentheses refer to the list of references at the end of this standard.
FIG. 1 FT-IR Spectrum of Hyaluronan, Sodium Salt Using Horizontal Attenuated Total Reflectance (HATR)
F2347 − 15
FIG. 2 H NMR Spectrum of Hyaluronan from Rooster Comb (Mw ~700 000)
is determined by measuring the relative viscosity in an Ubbelohde capillary viscometer. The measurements should be performed
in a solvent containing 0.15 M NaCl at a constant temperature of 20°C, and at a sufficiently low hyaluronan concentration.
Automatic operation and data acquisition are preferred.
5.2.2.2 Molecular Weight Determination Based on Differential Pressure—Alternatively, a Viscotek Relative Viscometer can be
used, which is based on Poiseuille’s law of capillary flow: the pressure drop of a fluid flowing through a capillary is directly
proportional to the viscosity.
ΔP 5 ηQR
where:
ΔP = the pressure drop across the capillary measured by the differential pressure transducer (DPT),
η = the viscosity,
Q = the flow rate, and
R = the resistance of the capillary.
Two capillaries are connected in series with the sample injection valve located between capillary one (1) and capillary two (2).
The sample is injected in capillary two (2) and the pressure change is detected by the DPT. The relative viscosity is determined
by the ratio of the pressures divided by the instrument constant K.
η 5 P /P K
r 2 1
The instrument constant K is the ratio of the resistances of capillary one (1) and two (2) at the base line where both capillaries
contain pure solvent. Specific viscosity, inherent viscosity, reduced viscosity, and intrinsic viscosity values can be calculated from
relative viscosity as follows:
η 5 η 2 1
sp r
where:
η = specific viscosity.
sp
η 5 η /C
red sp
F2347 − 15
where:
η = reduced viscosity and C is the concentration.
red
η 5 lim η /C as C→0
~ !
int sp
where:
η = the intrinsic viscosity.
int
From the intrinsic viscosity values, molecular weight can be calculated using the Mark-Houwink-Sakurada equation.
5.2.2.3 Molecular Weight and Polydispersity Determination by Size Exclusion Chromatography with Multiple Angle Laser Light
Scattering Detection (SEC-MALLS)—The method of choice is to use refractive index coupled to multiple angle laser light
scattering detection (MALLS). For separation of the hyaluronan into different molecular weight fractions, a hydrophilic column
with the appropriate pore size is required. Such columns include, but are not limited to those mentioned in the techniques below
and in Refs (6, 7). The precision of these techniques must be determined as results can vary by 5 to 20 %. Typical methods using
these techniques include, but are not limited to:
(1) Using 0.2 M NaCl as the mobile phase with separation using TSK 3000 and TSK 6000 columns.
(2) Using 150 mM NaCl, 50 mM phosphate buffer as the mobile phase with separation using a Biogel column.
5.2.2.4 Polydispersity—Depending on the end use and the sensitivity of the application to the molecular mass, the presence of
a wide range of hyaluronan fractions may be an issue. In such cases, calculation of the polydispersity will be important. Typically
this is between 1.2 and 3.0 for commercial hyaluronan.
5.2.3 Depending on the final use and the required performance control, other characterization assays can include, but are not
limited to the following:
5.2.3.1 Viscosity in Aqueous Solution—Viscosity is defined as a liquid’s resistance to flow. The molecular mass of hyaluronan
will determine the extent to which it will thicken an aqueous solution. Therefore, a simple viscosity test may yield information on
the relative differences in molecular mass among hyaluronan samples. To allow comparison between laboratories, the viscometer
used must be calibrated with traceable standards (see Test MethodsMethod D2196E2975). The viscosity measured will depend on
several parameters related to how the testing is conducted. Both rotational viscometers (Practice E1953) and “cone on plate”
rheometers may be used. Important parameters to control include, but are not limited to:
(1) Temperature—The temperature at which the measurement is performed is critical. An increase in temperature will, in
almost every case, result in a decrease in the viscosity. Consistent and controlled temperature (that is, with a standard temperature
bath) is critical to achieving reproducible results. Typically, the temperature used to measure viscosity can be 20°C, 25°C, or 37°C,
or combination thereof.
(2) Hyaluronan Concentration—The moisture content of the hyaluronan must be known in order to prepare correct
concentrations of hyaluronan (see 5.2.3.2).
(3) Ionic Strength—The viscosity of a hyaluronan solution is sensitive to the ionic environment in which the measurement is
made. The most important aspect is to keep the ionic strength consistent. Typically viscosity measurements should be made in a
standardized ionic environment of known ionic strength.
(4) Molecular Mass—Viscosity measurements are sensitive to the molecular mass of hyaluronan. The following is one
suggestion concerning the measurement of hyaluronan viscosity, but any appropriate method would apply. To measure the apparent
viscosity of hyaluronan, prepare a solution in deionized water with a concentration (w/w, corrected for dry matter content)
appropriate for the end use. The viscosity is measured using a rotational viscometer (for example,(see Practice E1953Brookfield
type) ) at 20 6 0.2°C (or other controlled temperature) using the appropriate spindle, spindle rotation speed and a
temperature-controlled water bath.
(5) Shear Rate—Hyaluronan is sensitive to shear and the viscosity may vary as a function of the shear rate.
5.2.3.2 Dry Matter Content—Hyaluronan from various suppliers may contain different moisture contents. The dry ma
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...