ASTM D4891-13(2018)
(Test Method)Standard Test Method for Heating Value of Gases in Natural Gas and Flare Gases Range by Stoichiometric Combustion
Standard Test Method for Heating Value of Gases in Natural Gas and Flare Gases Range by Stoichiometric Combustion
SIGNIFICANCE AND USE
5.1 This test method provides an accurate and reliable procedure to measure the total heating value of a fuel gas, on a continuous basis, which is used for regulatory compliance, custody transfer, and process control.
5.2 Some instruments which conform to the requirements set forth in this test method can have response times on the order of 1 min or less and can be used for on-line measurement and control.
5.3 The method is sensitive to the presence of oxygen and nonparaffin fuels. For components not listed and composition ranges that fall outside those in Table 1 and Table 2, modifications in the method and changes to the calibration gas or gasses being used may be required to obtain correct results.
SCOPE
1.1 This test method covers the determination of the heating value of natural gases and similar gaseous mixtures within the range of composition shown in Table 1, and Table 2 that covers flare components but is not intended to limit the components to be measured in flare gases.
1.2 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.3 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Aug-2018
- Technical Committee
- D03 - Gaseous Fuels
Relations
- Effective Date
- 01-Apr-2017
- Effective Date
- 01-May-2013
- Effective Date
- 01-Nov-2011
- Effective Date
- 01-May-2010
- Effective Date
- 01-Oct-2008
- Effective Date
- 01-Nov-2005
- Effective Date
- 10-May-2003
- Effective Date
- 10-May-1999
- Effective Date
- 10-Nov-1998
Overview
ASTM D4891-13(2018) provides a standardized test method for determining the total heating value of natural gases and flare gases by stoichiometric combustion. Developed and maintained by ASTM International, this method ensures an accurate and reliable procedure for continuous measurement of fuel gas heating value, which is essential for regulatory compliance, custody transfer, and process control in the natural gas industry.
The method is specifically designed for a wide range of natural gas and flare gas components, providing guidance for various fuel compositions. By focusing on stoichiometric combustion, it delivers repeatable and reproducible results, supporting critical operations in the measurement, billing, and regulatory sectors of the energy industry.
Key Topics
- Stoichiometric Combustion: Measurement is based on achieving the exact air-to-fuel ratio needed for the complete combustion of all combustibles, converting them into water and carbon dioxide.
- Continuous and On-line Measurement: Instruments following this ASTM standard can deliver real-time data, typically with response times of a minute or less.
- Calibration and Standardization: Detailed procedures ensure the reliability of results through regular calibration using gases of known heating values. Best practices for accuracy and repeatability are outlined.
- Sensitivity and Applicability:
- The method is sensitive to oxygen content and the presence of nonparaffinic fuels.
- For gases outside the specified composition ranges, method adjustments and calibration changes may be needed to ensure the results are valid.
- Precision and Bias: Interlaboratory studies confirmed high precision and minimal bias, with repeatability intervals and reproducibility defined for user reference.
- Safety Considerations: Users are responsible for following appropriate safety, health, and environmental protocols.
Applications
The ASTM D4891 test method is essential in several areas within the energy and petrochemical sectors, including:
- Regulatory Compliance: Satisfies requirements for accurate fuel gas measurement for environmental and commercial regulation.
- Custody Transfer: Provides a basis for fair and accurate billing when natural gas or mixed gases change ownership.
- Process Control: Enables precise, real-time monitoring and adjustment of industrial combustion processes, optimizing fuel use and emissions.
- Environmental Monitoring: Facilitates the assessment of flare gases, supporting compliance with environmental standards.
- Gas Quality Assessment: Assists utilities and industries in maintaining consistent fuel gas quality for processes and customers.
Typical users include natural gas producers, refineries, pipeline operators, utility companies, process engineers, laboratory technicians, and regulatory bodies.
Related Standards
For comprehensive gas measurement and process control, ASTM D4891 should be considered alongside other standards:
- ASTM D1826: Test Method for Calorific (Heating) Value of Gases in Natural Gas Range by Continuous Recording Calorimeter.
- ASTM E691: Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method.
- EPA-600/2-85-106: Evaluation of the Efficiency of Industrial Flares: Flare Head Design and Gas Composition.
- Wobbe Index Methods: For gas interchangeability assessments.
Practical Value
Utilizing ASTM D4891 ensures consistent, traceable, and internationally recognized measurement of the heating value of gaseous fuels. This contributes to:
- Enhanced operational efficiency and process safety
- Accurate regulatory reporting and billing
- Reliable quality control across the natural gas and petrochemical industries
Keywords: ASTM D4891, heating value, fuel gas, natural gas, flare gas, stoichiometric combustion, energy measurement, regulatory compliance, custody transfer, process control, gas analysis, ASTM standards.
Buy Documents
ASTM D4891-13(2018) - Standard Test Method for Heating Value of Gases in Natural Gas and Flare Gases Range by Stoichiometric Combustion
Get Certified
Connect with accredited certification bodies for this standard

ABS Quality Evaluations Inc.
American Bureau of Shipping quality certification.

Element Materials Technology
Materials testing and product certification.

ABS Group Brazil
ABS Group certification services in Brazil.
Sponsored listings
Frequently Asked Questions
ASTM D4891-13(2018) is a standard published by ASTM International. Its full title is "Standard Test Method for Heating Value of Gases in Natural Gas and Flare Gases Range by Stoichiometric Combustion". This standard covers: SIGNIFICANCE AND USE 5.1 This test method provides an accurate and reliable procedure to measure the total heating value of a fuel gas, on a continuous basis, which is used for regulatory compliance, custody transfer, and process control. 5.2 Some instruments which conform to the requirements set forth in this test method can have response times on the order of 1 min or less and can be used for on-line measurement and control. 5.3 The method is sensitive to the presence of oxygen and nonparaffin fuels. For components not listed and composition ranges that fall outside those in Table 1 and Table 2, modifications in the method and changes to the calibration gas or gasses being used may be required to obtain correct results. SCOPE 1.1 This test method covers the determination of the heating value of natural gases and similar gaseous mixtures within the range of composition shown in Table 1, and Table 2 that covers flare components but is not intended to limit the components to be measured in flare gases. 1.2 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.3 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 This test method provides an accurate and reliable procedure to measure the total heating value of a fuel gas, on a continuous basis, which is used for regulatory compliance, custody transfer, and process control. 5.2 Some instruments which conform to the requirements set forth in this test method can have response times on the order of 1 min or less and can be used for on-line measurement and control. 5.3 The method is sensitive to the presence of oxygen and nonparaffin fuels. For components not listed and composition ranges that fall outside those in Table 1 and Table 2, modifications in the method and changes to the calibration gas or gasses being used may be required to obtain correct results. SCOPE 1.1 This test method covers the determination of the heating value of natural gases and similar gaseous mixtures within the range of composition shown in Table 1, and Table 2 that covers flare components but is not intended to limit the components to be measured in flare gases. 1.2 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.3 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D4891-13(2018) is classified under the following ICS (International Classification for Standards) categories: 75.060 - Natural gas; 75.160.30 - Gaseous fuels. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D4891-13(2018) has the following relationships with other standards: It is inter standard links to ASTM D1826-94(2017), ASTM E691-13, ASTM E691-11, ASTM D1826-94(2010), ASTM E691-08, ASTM E691-05, ASTM D1826-94(2003), ASTM E691-99, ASTM D1826-94(1998). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D4891-13(2018) is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D4891 − 13 (Reapproved 2018)
Standard Test Method for
Heating Value of Gases in Natural Gas and Flare Gases
Range by Stoichiometric Combustion
This standard is issued under the fixed designation D4891; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 3.2 Definitions of Terms Specific to This Standard:
3.2.1 combustion ratio, n—the ratio of combustion air to
1.1 Thistestmethodcoversthedeterminationoftheheating
gaseous fuel.
value of natural gases and similar gaseous mixtures within the
3.2.2 burned gas parameter, n—apropertyoftheburnedgas
rangeofcompositionshowninTable1,andTable2thatcovers
after combustion which is a function of the combustion ratio.
flarecomponentsbutisnotintendedtolimitthecomponentsto
be measured in flare gases.
3.2.3 criticalcombustionratio,n—foraspecificburnedgas
parameter, the combustion ratio at which a plot of burned gas
1.2 This standard does not purport to address all of the
parameter versus combustion ratio has either maximum value
safety concerns, if any, associated with its use. It is the
or maximum slope.
responsibility of the user of this standard to establish appro-
priate safety, health, and environmental practices and deter-
3.2.4 combustion air requirement index (CARI), n—is the
mine the applicability of regulatory limitations prior to use.
amount of air required for complete combustion of the gas
1.3 This international standard was developed in accor-
being measured and can be used to index against other
dance with internationally recognized principles on standard-
measured values such as the Wobbe Index or Heating Value.
ization established in the Decision on Principles for the
3.2.5 stoichiometricratio,n—thecombustionratiowhenthe
Development of International Standards, Guides and Recom-
quantity of combustion air is just sufficient to convert all of the
mendations issued by the World Trade Organization Technical
combustibles in the fuel to water and carbon dioxide.
Barriers to Trade (TBT) Committee.
4. Summary of Test Method
2. Referenced Documents
2 4.1 Air is mixed with the gaseous fuel to be tested. The
2.1 ASTM Standards:
mixture is burned and the air-fuel ratio is adjusted so that
D1826TestMethodforCalorific(Heating)ValueofGasesin
essentially a stoichiometric proportion of air is present. More
Natural Gas Range by Continuous Recording Calorimeter
exactly, the adjustment is made so that the air-fuel ratio is in a
E691Practice for Conducting an Interlaboratory Study to
constant proportion to the stoichiometric ratio that is a relative
Determine the Precision of a Test Method
3 measure of the heating value. To set this ratio, a characteristic
2.2 EPA Standard:
property of the burned gas is measured, such as temperature or
EPA-600/2-85-106EvaluationoftheEfficiencyofIndustrial
oxygen concentration.
Flares: Flare Head Design and Gas Composition
5. Significance and Use
3. Terminology
5.1 This test method provides an accurate and reliable
3.1 All of the terms defined in Test Method D1826 are
procedure to measure the total heating value of a fuel gas, on
included by reference.
a continuous basis, which is used for regulatory compliance,
custody transfer, and process control.
ThistestmethodisunderthejurisdictionofASTMCommitteeD03onGaseous
Fuels and is the direct responsibility of Subcommittee D03.03 on Determination of
5.2 Some instruments which conform to the requirements
Heating Value and Relative Density of Gaseous Fuels.
set forth in this test method can have response times on the
Current edition approved Sept. 1, 2018. Published September 2018. Originally
orderof1minorlessandcanbeusedforon-linemeasurement
approved in 1989. Last previous edition approved in 2013 as D4891–13. DOI:
10.1520/D4891-13R18.
and control.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
5.3 The method is sensitive to the presence of oxygen and
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Standards volume information, refer to the standard’s Document Summary page on
nonparaffin fuels. For components not listed and composition
the ASTM website.
ranges that fall outside those in Table 1 and Table 2, modifi-
Available from United States Environmental Protection Agency (EPA), Ariel
cations in the method and changes to the calibration gas or
Rios Bldg., 1200 Pennsylvania Ave., NW, Washington, DC 20004, http://
www.epa.gov. gasses being used may be required to obtain correct results.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D4891 − 13 (2018)
TABLE 1 Natural Gas Components and Range of Composition TABLE 2 Natural Gas Components and Range of Composition
A
Covered Covered
Compound CAS Number
Compound Concentration Range, mole, %
Volatile Analytes
Helium 0.01 to 5
Acetone 67-64-1
Nitrogen 0.01 to 20
Acetonitrile 75-05-8
Carbon dioxide 0.01 to 10
Acrolein 107-05-8
Methane 50 to 100
Acrylonitrile 107-13-1
Ethane 0.01 to 20
Benzene 71-43-2 2
Propane 0.01 to 20
1,3-Butadiene 106-99-0
n-butane 0.01 to 10
Carbon disulfide 75-15-0
isobutane 0.01 to 10
Chlorobenzene 108-90-7
n-pentane 0.01 to 2
Cumene 98-82-8
Isopentane 0.01 to 2
(isopropylbenzene)
Hexanes and heavier 0.01 to 2
1,2-Dibromoethane 106-93-4
Ethylbenzene 100-41-4 2,2,4
Hexane 110-54-3
Methanol 67-56-1
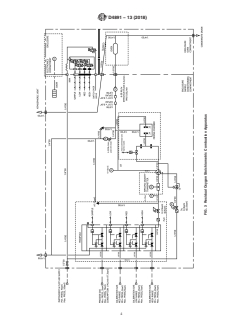
6. Apparatus
Methyl isobutyl ketone 108-10-1
Methyl t-butyl ether 1634-04-4
6.1 A suitable apparatus for carrying out the stoichiometric
Methylene chloride 75-09-2
combustion method will have at least the following four
Nitrobenzene 98-95-3
components: flow meter or regulator, or both; combustion Nitropropane 79-46-9
Pentane2 109-66-0
chamber; burned gas sensor; and electronics. The requirement
Styrene 100-42-5
for each of these components is discussed below. The detailed
Tetrachloroethene 127-18-4
design of each of these components can vary. Three different Toluene 108-88-3
Trichloroethene 79-01-6
apparatus are shown in Fig. 1, Fig. 2 and Fig. 3. In each figure
Trimethylpentane 2 540-84-1
the equivalent of the four necessary components are enclosed
Xylenes (mixed isomers) 1330-20-7
Trimethylpentane 2 540-84-1
in dashed lines.
Xylenes (mixed isomers) 1330-20-7
6.2 Overview—Airandfuelentertheapparatusandtheflow
Semi-volatile Analytes
of each is measured. Alternatively, only one gas flow need be
Acenaphthene 83-32-9
measured if the flow of the other is kept the same during
Acenaphthylene 208-96-8
measurement and calibration.This is illustrated in Fig. 2. Next
Aniline 62-53-3
Anthracene 120-12-7
there is a combustion chamber in which the air and fuel are
Benzidine1 92-87-5
mixedandburned.Thiscanbeassimpleasabunsenormeeker
Benz[a]anthracene 56-55-3
burner, but precautions should be taken that subsequent mea-
Benzo[b]fluoranthene 205-99-2
Benzo[k]fluoranthene 207-08-9
surements of burned gas characteristics are not influenced by
Benzo[g,h,i]perylene 191-24-2
ambient conditions. Finally, there is a sensor in the burned gas
Benzo[a]pyrene 50-32-8
which measures a property of this gas that is sensitive to the Benzo[e]pyrene2 192-97-2
Biphenyl2, 92-52-4
combustion ratio and has a unique feature at the stoichiometric
Cresol (mixed isomers) 1319-77-3
ratio. Two such properties are temperature and oxygen
Chrysene 218-01-9
concentrations, and either can be measured. Dibenz[a,h]anthracene 53-70-3
Dibenzofuran 132-64-9
6.3 Flow Meter or Regulator, or both—The flow measure-
Dibenzo(a,e)pyrene 192-65-4
3,3’- Dimethoxybenzidine 119-90-4
ment part of the apparatus should have an accuracy and
Dimethylaminobenzene 60-11-7
precision of the order of 0.1%. Likewise, if the flow is to be
7,12- 57-97-6
kept constant, the flow regulator should maintain this constant
Dimethylbenz(a)anthracene
3,3’- Dimethylbenzidine 119-93-7
valuewithin0.1%.Themeterorregulatorfornaturalgasmust
á,á- 122-09-8
maintain this precision and accuracy over the density and
Dimethylphenethylamine
viscosityrangesconsistentwiththecompositionrangeinTable
2,4-Dimethylphenol 105-67-9
Fluoranthene 206-44-0
1 or Table 2.
Fluorene 86-73-7
6.4 Combustion Chamber: Indeno(1,2,3-cd)pyrene 193-39-5
Isophorone 78-59-1
6.4.1 There are two different types of combustion chambers
3-Methylcholanthrene 56-49-5
thatmaybeused.Inthefirsttypetheairandfuelaremixedand
2-Methylnaphthalene 91-57-6
burned in a single burner. The apparatus shown in Fig. 1 has Naphthalene 91-20-3
Perylene2 198-55-0
this type of combustion chamber.
Phenanthrene 85-01-8
6.4.2 In the second type of combustion chamber, the air and
Phenol 108-95-2
fuel are each divided into two streams, and combustion takes 1,4-Phenylenediamine 106-50-3
Pyrene 129-00-0
place simultaneously in two burners. The division of air flow
o-Toluidine 95-53-4
must be such that the proportion of air going to each burner
Aldehydes
always remains the same. Likewise the division of fuel flow
Methanol 67-56-1
must always remain the same even through fuel composition
Formaldehyde 50-00-0
changes.Anotherrequirementisthattheflowdivisionsbesuch
Acetaldehyde 75-07-0
thatoneburnerhasamixturewithaslightlyhighercombustion
D4891 − 13 (2018)
TABLE 2 Continued
Compound CAS Number
Propanal 123-38-6
C1 to C5 Hydrocarbons
Description Compound CAS Number
C1 Alkanes Methane 74-82-8
C2 Alkanes Ethane 74-84-0
C3 Alkanes Propane 74-98-6
n-Butane 106-97-8
C4 Alkanes
Isobutane 75-28-5
n-Pentane 109-66-0
C5 Alkanes 2-Methylbutane 78-78-4
Cyclopentane 287-92-3
C2 Olefins Ethylene 74-85-1
C2 Alkanes Acetylene 74-86-2
C3 Olefins Propylene 115-07-1
C4 Olefins 1-Butene 106-98-9
2-Butene 107-01-7
Isobutene 115-11-7
C5 Olefins 1-Pentene 109-67-1
FIG. 2 Stoichiometric Combustion Apparatus
Cis-2-pentene 627-20-3
Trans-2-pentene 646-04-8
2-Methyl-1-butene 563-46-2
ratiothantheother.TheapparatusshowninFig.2hasthistype
3-Methyl-1-butene 563-45-1
of combustion chamber.
2-Methyl-2-butene 513-35-9
Cylcopentene 142-29-0
6.4.3 A third type utilizes a combustion oven operating in
C3 Alkadienes Propadiene 463-49-0
excess of 800°C (1472°F) to assure the combustion of gases
C4 Alkadienes 1,2-Butadiene 590-19-2
1,3-Butadiene 106-99-0 withinthenaturalorflaregascompositionsbeingcombustedas
C5 Alkadienes 1,2-Pentadiene 591-95-7
shown in Fig. 3.
1-cis-3-Pentadiene 1574-41-0
1-trans-3- Pentadiene 2004-70-8 6.5 Burned Gas Sensor:
1,4-Pentadiene 591-93-5
6.5.1 The burned or combusted gas sensor must measure a
2,3-Pentadiene 591-96-8
characteristic of the burned gas which is a function of the
3-Methyl-1,2- butadiene 598-25-4
2-Methyl-1,3- butadiene 78-79-5
combustion ratio and for which there is a critical combustion
Cyclopentadiene 542-92-7
ratio related to the stoichiometric ratio.Acombustion chamber
Heating Value Range
of the first type (Fig. 1) would have one sensor in the bu
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...