ASTM D6888-16(2023)
(Test Method)Standard Test Method for Available Cyanides with Ligand Displacement and Flow Injection Analysis (FIA) Utilizing Gas Diffusion Separation and Amperometric Detection
Standard Test Method for Available Cyanides with Ligand Displacement and Flow Injection Analysis (FIA) Utilizing Gas Diffusion Separation and Amperometric Detection
SIGNIFICANCE AND USE
5.1 Cyanide and hydrogen cyanide are highly toxic. Regulations have been established to require the monitoring of cyanide in industrial and domestic wastes and surface waters.3
5.2 This test method is applicable for natural water, saline waters, metallurgical process solutions, and wastewater effluent.
5.3 The method may be used for process control in wastewater treatment facilities.
SCOPE
1.1 This test method is used to determine the concentration of available inorganic cyanide in an aqueous wastewater or effluent. The method detects the cyanides that are free (HCN and CN-) and metal-cyanide complexes that are easily dissociated into free cyanide ions. The method does not detect the less toxic strong metal-cyanide complexes, cyanides that are not “amenable to chlorination.”
1.2 Total cyanide can be determined for samples that have been distilled as described in Test Methods D2036, Test Method A, Total Cyanides after Distillation. The cyanide complexes are dissociated and absorbed into the sodium hydroxide capture solution, which can be analyzed with this test method; therefore, ligand exchange reagents from 8.12 and 8.13 would not be required when determining total cyanide after distillation.
1.3 This procedure is applicable over a range of approximately 2 μg/L to 400 μg/L (parts per billion) available cyanides. Higher concentrations can be analyzed by dilution or lower injection volume.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific hazard statements are given in 8.6 and Section 9.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 14-Nov-2023
- Technical Committee
- D19 - Water
- Drafting Committee
- D19.06 - Methods for Analysis for Organic Substances in Water
Relations
- Effective Date
- 15-Nov-2023
- Effective Date
- 15-Nov-2023
- Effective Date
- 01-May-2020
- Effective Date
- 01-May-2020
- Effective Date
- 01-Apr-2016
- Effective Date
- 15-Dec-2015
- Effective Date
- 15-Nov-2023
- Effective Date
- 15-Nov-2023
- Effective Date
- 15-Nov-2023
- Effective Date
- 15-Nov-2023
- Effective Date
- 15-Nov-2023
- Effective Date
- 15-Nov-2023
- Effective Date
- 15-Nov-2023
- Effective Date
- 15-Nov-2023
- Effective Date
- 15-Nov-2023
Overview
ASTM D6888-16(2023) is the internationally recognized standard test method for determining the concentration of available inorganic cyanide in aqueous samples, including wastewater, effluent, natural waters, and process solutions. Using ligand displacement and flow injection analysis (FIA) with gas diffusion separation and amperometric detection, this method provides accurate monitoring of cyanide species that are relevant for environmental regulation and industrial process control. The test specifically detects free cyanide (hydrogen cyanide and cyanide ion) and weakly bound metal-cyanide complexes, ensuring compliance with safety and environmental guidelines.
Key Topics
Scope and Applicability
- Designed for analysis of available cyanide in natural water, saline water, metallurgical process solutions, and wastewater effluent.
- Intended for process control in wastewater treatment facilities.
- Applicable for concentrations ranging from approximately 2 µg/L to 400 µg/L; higher concentrations can be measured by dilution.
Test Method Fundamentals
- Employs ligand displacement to release cyanides bound to nickel or mercury.
- Utilizes flow injection analysis, which introduces the sample into an acidic stream, releasing hydrogen cyanide gas.
- Gas diffusion allows HCN to pass through a hydrophobic membrane into an alkaline acceptor stream.
- Captured cyanide is measured amperometrically using a silver electrode, generating a response proportional to cyanide concentration.
Cyanide Species Detected
- Measures available cyanides: free cyanides and weak acid dissociable or weakly bound complex cyanides.
- Does not detect less toxic, strong metal-cyanide complexes or cyanides not amenable to chlorination.
Calibration and Precision
- Recommends rigorous calibration and verification daily, with strict quality assurance protocols.
- Provides guidance on sample treatment, calibration curve fitting, and interference mitigation to ensure data accuracy.
Environmental and Safety Considerations
- Addresses hazards associated with cyanide handling and requires adherence to established safety, health, and waste disposal practices.
- Developed according to WTO Technical Barriers to Trade (TBT) principles, supporting international regulatory acceptance.
Applications
ASTM D6888-16(2023) offers practical value across multiple fields, supporting:
Environmental Monitoring
- Assists in compliance with water quality regulations by detecting toxic cyanide species in industrial wastewater, effluents, and surface waters.
- Enables monitoring programs for domestic waste streams and protects aquatic environments from hazardous cyanide contamination.
Industrial Process Control
- Essential for metallurgical operations managing cyanide-bearing process solutions.
- Facilitates optimization and regulatory compliance in wastewater treatment processes in mining, chemical, and manufacturing industries.
Public Health and Safety
- Provides data for risk assessment and management by reliably quantifying available cyanide.
- Used to meet regulatory reporting requirements and to demonstrate adherence to environmental health standards.
Related Standards
Leveraging ASTM D6888-16(2023) often involves referencing additional standards for comprehensive water analysis. Notable related standards include:
- ASTM D2036 – Test Methods for Cyanides in Water (for total cyanide determination after distillation)
- ASTM D1129 – Terminology Relating to Water
- ASTM D1193 – Specification for Reagent Water
- ASTM D3856 – Guide for Management Systems in Laboratories Engaged in Analysis of Water
- ASTM D5847 – Practice for Writing Quality Control Specifications for Standard Test Methods for Water Analysis
- ASTM D7365 – Practice for Sampling, Preservation, and Mitigating Interferences in Water Samples for Analysis of Cyanide
Together, these standards establish a robust framework for reliable water analysis, environmental monitoring, and regulatory compliance concerning cyanide-containing waters.
Keywords: ASTM D6888, cyanide determination, available cyanide, flow injection analysis, gas diffusion, amperometric detection, wastewater monitoring, cyanide in water, environmental standards, water quality compliance
Buy Documents
ASTM D6888-16(2023) - Standard Test Method for Available Cyanides with Ligand Displacement and Flow Injection Analysis (FIA) Utilizing Gas Diffusion Separation and Amperometric Detection
Get Certified
Connect with accredited certification bodies for this standard

BSMI (Bureau of Standards, Metrology and Inspection)
Taiwan's standards and inspection authority.
Sponsored listings
Frequently Asked Questions
ASTM D6888-16(2023) is a standard published by ASTM International. Its full title is "Standard Test Method for Available Cyanides with Ligand Displacement and Flow Injection Analysis (FIA) Utilizing Gas Diffusion Separation and Amperometric Detection". This standard covers: SIGNIFICANCE AND USE 5.1 Cyanide and hydrogen cyanide are highly toxic. Regulations have been established to require the monitoring of cyanide in industrial and domestic wastes and surface waters.3 5.2 This test method is applicable for natural water, saline waters, metallurgical process solutions, and wastewater effluent. 5.3 The method may be used for process control in wastewater treatment facilities. SCOPE 1.1 This test method is used to determine the concentration of available inorganic cyanide in an aqueous wastewater or effluent. The method detects the cyanides that are free (HCN and CN-) and metal-cyanide complexes that are easily dissociated into free cyanide ions. The method does not detect the less toxic strong metal-cyanide complexes, cyanides that are not “amenable to chlorination.” 1.2 Total cyanide can be determined for samples that have been distilled as described in Test Methods D2036, Test Method A, Total Cyanides after Distillation. The cyanide complexes are dissociated and absorbed into the sodium hydroxide capture solution, which can be analyzed with this test method; therefore, ligand exchange reagents from 8.12 and 8.13 would not be required when determining total cyanide after distillation. 1.3 This procedure is applicable over a range of approximately 2 μg/L to 400 μg/L (parts per billion) available cyanides. Higher concentrations can be analyzed by dilution or lower injection volume. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific hazard statements are given in 8.6 and Section 9. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 Cyanide and hydrogen cyanide are highly toxic. Regulations have been established to require the monitoring of cyanide in industrial and domestic wastes and surface waters.3 5.2 This test method is applicable for natural water, saline waters, metallurgical process solutions, and wastewater effluent. 5.3 The method may be used for process control in wastewater treatment facilities. SCOPE 1.1 This test method is used to determine the concentration of available inorganic cyanide in an aqueous wastewater or effluent. The method detects the cyanides that are free (HCN and CN-) and metal-cyanide complexes that are easily dissociated into free cyanide ions. The method does not detect the less toxic strong metal-cyanide complexes, cyanides that are not “amenable to chlorination.” 1.2 Total cyanide can be determined for samples that have been distilled as described in Test Methods D2036, Test Method A, Total Cyanides after Distillation. The cyanide complexes are dissociated and absorbed into the sodium hydroxide capture solution, which can be analyzed with this test method; therefore, ligand exchange reagents from 8.12 and 8.13 would not be required when determining total cyanide after distillation. 1.3 This procedure is applicable over a range of approximately 2 μg/L to 400 μg/L (parts per billion) available cyanides. Higher concentrations can be analyzed by dilution or lower injection volume. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific hazard statements are given in 8.6 and Section 9. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D6888-16(2023) is classified under the following ICS (International Classification for Standards) categories: 17.120.10 - Flow in closed conduits. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D6888-16(2023) has the following relationships with other standards: It is inter standard links to ASTM D6888-16, ASTM D6696-16(2023), ASTM D1129-13(2020)e1, ASTM D1129-13(2020)e2, ASTM D6696-16, ASTM D3856-11(2015), ASTM D2036-09(2022), ASTM D7728-18, ASTM E800-20, ASTM D7572-15(2022), ASTM D7365-09a(2022), ASTM E1600-23, ASTM D7237-18, ASTM D7284-20, ASTM D8273-20. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D6888-16(2023) is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D6888 − 16 (Reapproved 2023)
Standard Test Method for
Available Cyanides with Ligand Displacement and Flow
Injection Analysis (FIA) Utilizing Gas Diffusion Separation
and Amperometric Detection
This standard is issued under the fixed designation D6888; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope mendations issued by the World Trade Organization Technical
Barriers to Trade (TBT) Committee.
1.1 This test method is used to determine the concentration
of available inorganic cyanide in an aqueous wastewater or
2. Referenced Documents
effluent. The method detects the cyanides that are free (HCN
-
2.1 ASTM Standards:
and CN ) and metal-cyanide complexes that are easily disso-
D1129 Terminology Relating to Water
ciated into free cyanide ions. The method does not detect the
D1193 Specification for Reagent Water
less toxic strong metal-cyanide complexes, cyanides that are
D2036 Test Methods for Cyanides in Water
not “amenable to chlorination.”
D3856 Guide for Management Systems in Laboratories
1.2 Total cyanide can be determined for samples that have
Engaged in Analysis of Water
been distilled as described in Test Methods D2036, Test
D5847 Practice for Writing Quality Control Specifications
Method A, Total Cyanides after Distillation. The cyanide
for Standard Test Methods for Water Analysis
complexes are dissociated and absorbed into the sodium
D6696 Guide for Understanding Cyanide Species
hydroxide capture solution, which can be analyzed with this
D7365 Practice for Sampling, Preservation and Mitigating
test method; therefore, ligand exchange reagents from 8.12 and
Interferences in Water Samples for Analysis of Cyanide
8.13 would not be required when determining total cyanide
after distillation.
3. Terminology
1.3 This procedure is applicable over a range of approxi-
3.1 Definitions:
mately 2 μg ⁄L to 400 μg ⁄L (parts per billion) available cya-
3.1.1 For definitions of terms used in this standard, refer to
nides. Higher concentrations can be analyzed by dilution or
Terminology D1129 and Guide D6696.
lower injection volume.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 available cyanides, n—Inorganic cyanides that are
1.4 The values stated in SI units are to be regarded as
-
free (HCN and CN ) and metal-cyanide complexes that are
standard. No other units of measurement are included in this
easily dissociated into free cyanide ions.
standard.
3.2.1.1 Discussion—Available cyanide does not include the
1.5 This standard does not purport to address all of the
less toxic strong metal-cyanide complexes, cyanides that are
safety concerns, if any, associated with its use. It is the
not “amenable to chlorination” and includes weak acid disso-
responsibility of the user of this standard to establish appro-
ciable or weak and dissociable (WAD) cyanides for use in the
priate safety, health, and environmental practices and deter-
implementation of International Cyanide Management Code.
mine the applicability of regulatory limitations prior to use.
Specific hazard statements are given in 8.6 and Section 9.
4. Summary of Test Method
1.6 This international standard was developed in accor-
4.1 Complex cyanides bound with nickel or mercury are
dance with internationally recognized principles on standard-
released by ligand displacement by the addition of a ligand
ization established in the Decision on Principles for the
displacement agent, when necessary.
Development of International Standards, Guides and Recom-
4.2 Other weak and dissociable cyanide species do not
require ligand displacement.
This test method is under the jurisdiction of ASTM Committee D19 on Water
and is the direct responsibility of Subcommittee D19.06 on Methods for Analysis for
Organic Substances in Water. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved Nov. 15, 2023. Published December 2023. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 2003. Last previous addition approved in 2016 as D6888 – 16. DOI: Standards volume information, refer to the standard’s Document Summary page on
10.1520/D6888-16R23. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D6888 − 16 (2023)
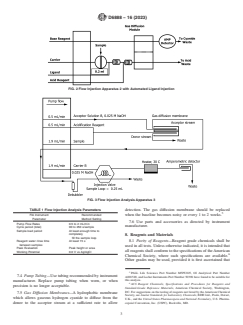
FIG. 1 Flow Injection Analysis Apparatus 1
4.3 The sample is introduced into a flow injection analysis 6.2 Sulfide above 50 mg/L will diffuse through the gas
(FIA) system where it is acidified to form hydrogen cyanide diffusion membrane and can be detected in the amperometric
(HCN). The hydrogen cyanide gas diffuses through a hydro- flowcell. Oxidized products of sulfide can also rapidly convert
- -
phobic gas diffusion membrane, from the acidic donor stream CN to SCN at a high pH. Refer to Practice D7365 for sulfide
into an alkaline acceptor stream. removal procedures.
4.4 The captured cyanide is sent to an amperometric flow- 6.3 Refer to Practice D7365 for further information on
cell detector with a silver-working electrode. In the presence of mitigating interferences in water samples for the analysis of
cyanide, silver in the working electrode is oxidized at the cyanide.
applied potential. The anodic current measured is proportional
7. Apparatus
to the concentration of cyanide in the standard or sample
injected.
7.1 The instrument should be equipped with a precise
sample introduction system, a gas diffusion manifold with
4.5 Calibrations and data are processed with the instru-
hydrophobic membrane, and an amperometric detection sys-
ment’s data acquisition software.
tem to include a silver working electrode, a Ag/AgCl reference
electrode, and a Pt or stainless steel counter electrode. Ex-
5. Significance and Use
amples of the apparatus schematics are shown in Figs. 1-3.
5.1 Cyanide and hydrogen cyanide are highly toxic. Regu-
Example instrument settings are shown in Table 1.
lations have been established to require the monitoring of
NOTE 1—The instrument settings in Table 1 are only examples. The
cyanide in industrial and domestic wastes and surface waters.
analyst may modify the settings as long as performance of the method has
not been degraded. Contact the instrument manufacturer for recommended
5.2 This test method is applicable for natural water, saline
instrument parameters.
waters, metallurgical process solutions, and wastewater efflu-
7.2 An autosampler is recommended but not required to
ent.
automate sample injections and increase throughput. Autosam-
5.3 The method may be used for process control in waste-
plers are usually available as an option from the instrument’s
water treatment facilities.
manufacturer.
7.3 Data Acquisition System—Use the computer hardware
6. Interferences
and software recommended by the instrument manufacturer to
6.1 High levels of carbonate can release CO into the
control the apparatus and to collect data from the detector.
acceptor stream and cause an interference with the amperomet-
ric detector that result in a slight masking effect (15 % negative
bias with 20 ppb cyanide in 1500 ppm carbonate). Refer to 11.2
OI Analytical CNSolution 3100, FS3100, or Flow Solution IV and Lachat
Instruments QuikChem Automated Ion Analyzer using Method 10-204-00-5-A have
for sample pretreatment.
been found to be suitable for this analysis. If you are aware of alternative suppliers,
please provide this information to ASTM International Headquarters. Your com-
ments will receive careful consideration at a meeting of the responsible technical
3 1
40 CFR Part 136. committee, which you may attend.
D6888 − 16 (2023)
FIG. 2 Flow Injection Apparatus 2 with Automated Ligand Injection
FIG. 3 Flow Injection Analysis Apparatus 3
TABLE 1 Flow Injection Analysis Parameters
detection. The gas diffusion membrane should be replaced
FIA Instrument Recommended when the baseline becomes noisy or every 1 to 2 weeks.
Parameter Method Setting
7.6 Use parts and accessories as directed by instrument
Pump Flow Rates 0.5 to 2 mL/min
manufacturer.
Cycle period (total) 90 to 250 s/sample
Sample load period At least enough time to
completely
8. Reagents and Materials
fill the sample loop
Reagent water rinse time At least 15 s 8.1 Purity of Reagents—Reagent grade chemicals shall be
between samples
used in all tests. Unless otherwise indicated, it is intended that
Peak Evaluation Peak height or area
all reagents shall conform to the specifications of the American
Working Potential 0.0 V vs Ag/AgCl
Chemical Society, where such specifications are available.
Other grades may be used, provided it is first ascertained that
PALL Life Sciences Part Number M5PU025, OI Analytical Part Number
7.4 Pump Tubing—Use tubing recommended by instrument
A001520, and Lachat Instruments Part Number 50398 have found to be suitable for
this analysis.
manufacturer. Replace pump tubing when worn, or when
ACS Reagent Chemicals, Specifications and Procedures for Reagents and
precision is no longer acceptable.
Standard-Grade Reference Materials, American Chemical Society, Washington,
DC. For suggestions on the testing of reagents not listed by the American Chemical
7.5 Gas Diffusion Membranes—A hydrophobic membrane
Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset,
which allows gaseous hydrogen cyanide to diffuse from the
U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharma-
donor to the acceptor stream at a sufficient rate to allow copeial Convention, Inc. (USPC), Rockville, MD.
D6888 − 16 (2023)
the reagent is of sufficiently high purity to permit its use 8.11 Carrier A—Use water as the carrier.
without lessening the accuracy of the determination.
8.12 Ligand Exchange Reagent 1 (TEP Solution)—Weigh
8.2 Purity of Water—Unless otherwise indicated, references
0.10 g tetraethylenepentamine (TEP) into a 100 mL volumetric
to water shall be understood to mean reagent water conforming flask. Dilute to volume with laboratory water. The solution
to Type II grade of Specification D1193.
should be stored at room temperature.
8.3 Sodium Hydroxide Solution (1.00 M)—Dissolve 40 g
8.13 Ligand Exchange Reagent 2 (Dithizone Solution)—
NaOH in laboratory water and dilute to 1 L.
Weigh 0.010 g of dithizone into a 100 mL volumetric flask
containing 1.0 mL of 1.00 M NaOH (see 8.3). Dilute to volume
8.4 Acceptor Solution A (0.10 M NaOH)—Dissolve 4.0 g
with laboratory water. Sonicate if necessary until all of the
NaOH in laboratory water and dilute to 1 L.
dithizone has dissolved. The solution should be stored at room
8.5 Acceptor Solution B, Carrier B (0.025 M NaOH)—
temperature.
Dissolve 1.0 g NaOH in laboratory water and dilute to 1 L.
NOTE 2—Commercially prepared or alternative ligand exchange re-
-
agents can be used if equivalent results can be demonstrated. Commercial
8.6 Stock Cyanide Solution (1000 μg/mL CN )—Dissolve
reagents should be used in accordance with manufacturer’s instructions.
2.51 g of KCN and 2.0 g of NaOH in 1 L of water. Standardize
with silver nitrate solution as described in Test Methods 8.14 Mixed Ligand Exchange Reagent, for automated ligand
addition as shown in Fig. 2—Transfer 0.125 mL of WAD
D2036, Section 16.2. Store the solution under refrigeration and
check concentration approximately every 6 months and correct Reagent A and 0.250 mL of WAD Reagent B8 into a 100 mL
volumetric flask containing 50 mL laboratory water. Dilute to
if necessary. (Warning—Because KCN is highly toxic, avoid
contact or inhalation.) volume with laboratory water and mix. The solution should be
stored at room temperature
8.7 Intermediate Cyanide Standards:
-
8.7.1 Intermediate Cyanide Standard 1 (100 μg/mL CN )— 8.15 Mercury (II) Cyanide Stock Solution—Weigh 0.4854 g
Pipette 10.0 mL of stock cyanide solution (see 8.6) into a Hg(CN) into a 100 mL volumetric flask. Place 1.0 mL of 1.00
100 mL volumetric flask containing 1 mL of 1.0 M NaOH (see M NaOH (see 8.3) in the flask and dilute to volume with
-
8.3). Dilute to volume with laboratory water. Store under laboratory water. Hg(CN) as CN = 1000 mg/L. The solution
refrigeration. The standard should be stable for 6 months. must be stored in an amber glass bottle under refrigeration. The
-
8.7.2 Intermediate Cyanide Standard 2 (10 μg/mL CN )— standard should be stable for 6 months.
Pipette 10.0 mL of Intermediate Cyanide Standard 1 (see 8.7.1)
8.16 Mercury (II) Cyanide Intermediate Solution—Pipet
into a 100 mL volumetric flask containing 1.0 mL of 1.00 M
10.0 mL of the mercury (II) cyanide stock solution (see 8.15)
NaOH (see 8.3). Dilute to volume with laboratory water. Store
into a 100 mL volumetric flask containing 1.0 mL of 1.00 M
under refrigeration. The standard should be stable for 6
NaOH (see 8.3). Dilute to volume with laboratory grade water.
months. -
Hg(CN) as CN = 100 mg/L. The solution must be stored in an
8.8 Working Cyanide Calibration Standards—Prepare fresh amber glass bottle under refrigeration. The standard should be
weekly as described in 8.8.1 and 8.8.2 ranging in concentration stable for 6 months.
-
from 2 μg ⁄L to 400 μg ⁄L CN .
8.17 Mercury (II) Cyanide Recovery Solution—Pipet 100 μL
8.8.1 Calibration Standards (20 μg ⁄L, 50 μg ⁄L, 100 μg ⁄L,
of mercury II cyanide intermediate solution (see 8.16) into a
-
200 μg ⁄L, and 400 μg/L CN )—Pipette 20 μL, 50 μL, 100 μL,
100 mL volumetric flask containing 1.0 mL of 1.00 M NaOH
200 μL, and 400 μL of Intermediate Cyanide Standard 1 (see
(see 8.3). Dilute to volume with laboratory water. Hg(CN) as
8.7.1) into separate 100 mL volumetric flasks containing
-
CN = 100 μg ⁄L. Prepare fresh weekly.
1.0 mL of 1.00 M NaOH (see 8.3). Dilute to volume with
8.18 Potassium Nickel Cyanide Stock Solution—Weigh
laboratory water.
-
0.2488 g of K Ni(CN) H O in a 100 mL volumetric flask.
8.8.2 Calibration Standards (2 μL and 10 μg/L CN )— 2 4 2
Place 1.0 mL of 1.00 M NaOH (see 8.3) in the flask and dilute
Pipette 20 μL and 100 μL of Intermediate Cyanide Standard 2
-
to volume with laboratory water. K Ni(CN) as CN =
(see 8.7.2) into separate 100 mL volumetric flasks containing 2 4
1000 mg ⁄L. The solution must be stored in an amber glass
1.0 mL of 1.00 M NaOH (see 8.3). Dilute to volume with
bottle under refrigeration. The standard should be stable for 6
laboratory water.
months.
8.9 Cyanide Electrode Stabilization Solution (2 mg/L as
-
8.19 Potassium Nickel Cyanide Intermediate Solution—
CN )—Pipette 200 μL of Stock Cyanide (see 8.6) into a 100 mL
Pipet 10.0 mL of the potassium nickel cyanide stock solution
volumetric flask containing 1.0 mL of 1.00 M NaOH (see 8.3).
(see 8.18) into a 100 mL volumetric flask containing 1.0 mL of
Dilute to volume with laboratory water. The solution should be
1.00 M NaOH (see 8.3). Dilute to volume with laboratory
stored under refrigeration.
-
grade water. K Ni(CN) as CN = 100 mg/L. The solution must
2 4
8.10 Acetate Buffer—Dissolve 410 g of sodium acetate
be stored in an amber glass bottle under refrigeration. The
trihydrate (NaC H O ·3H O) in 500 mL of laboratory water.
2 3 2 2
standard should be stable for 6 months.
Add glacial acetic acid (approximately 500 mL) to yield a pH
of 4.5.
OI Analytical WAD Reagents A and B, PN A001416 and A001417 have found
Commerical Solutions of Stock Cyanide may be substituted. to be suitable for this analysis.
D6888 − 16 (2023)
8.20 Potassium Nickel Cyani
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...