ASTM F2255-05(2015)
(Test Method)Standard Test Method for Strength Properties of Tissue Adhesives in Lap-Shear by Tension Loading
Standard Test Method for Strength Properties of Tissue Adhesives in Lap-Shear by Tension Loading
SIGNIFICANCE AND USE
4.1 Materials and devices that function at least in part by adhering to living tissues are finding increasing use in surgical procedures either as adjuncts to sutures and staples, or as frank replacements for those devices in a wide variety of medical procedures. While the nature and magnitude of the forces involved varies greatly with indication and with patient specific circumstances, all uses involve to some extent the ability of the material to resist imposed mechanical forces. Therefore, the mechanical properties of the materials, and in particular the adhesive properties, are important parameters in evaluating their fitness for use. In addition, the mechanical properties of a given adhesive composition can provide a useful means of determining product consistency for quality control, or as a means for determining the effects of various surface treatments on the substrate prior to use of the device.
4.2 The complexity and variety of individual applications for tissue adhesive devices, even within a single indicated use (surgical procedure) is such that the results of a single-lap-shear test are not suitable for determining allowable design stresses without thorough analysis and understanding of the application and adhesive behaviors.
4.3 This test method may be used for comparing adhesives or bonding processes for susceptibility to fatigue and environmental changes, but such comparisons must be made with great caution since different adhesives may respond differently to varying conditions.
SCOPE
1.1 This test method is intended to provide a means for comparison of the adhesive strengths of tissue adhesives intended for use as surgical adhesives or sealants, or both, on soft tissue. With the appropriate choice of substrate, it may also be used for purposes of quality control in the manufacture of tissue adhesive based medical devices.
1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
General Information
- Status
- Published
- Publication Date
- 30-Apr-2015
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.15 - Material Test Methods
Relations
- Effective Date
- 01-May-2015
- Effective Date
- 01-Jun-2014
- Effective Date
- 01-Jul-2012
- Effective Date
- 01-May-2012
- Effective Date
- 01-Dec-2011
- Effective Date
- 01-Jan-2011
- Effective Date
- 01-Jun-2010
- Effective Date
- 01-Nov-2009
- Effective Date
- 01-Apr-2009
- Effective Date
- 01-Dec-2008
- Effective Date
- 01-Oct-2008
- Effective Date
- 15-Aug-2008
- Effective Date
- 01-Mar-2008
- Effective Date
- 01-Jan-2007
- Effective Date
- 01-Dec-2006
Overview
ASTM F2255-05(2015) is the Standard Test Method for Strength Properties of Tissue Adhesives in Lap-Shear by Tension Loading, developed by ASTM International. This standardized test method is designed to evaluate and compare the adhesive strength of tissue adhesives - materials intended for use as surgical adhesives or sealants on soft tissue. Tissue adhesives are increasingly utilized in medical procedures, serving either as adjuncts to traditional wound closure devices like sutures and staples, or as complete replacements.
By providing a reliable and repeatable method to measure the lap-shear strength of tissue adhesives, this standard supports product development, quality control, and the assessment of performance characteristics necessary for regulatory and clinical considerations. The results of this method offer crucial information for manufacturers, clinicians, and regulatory bodies engaged in the evaluation and usage of tissue adhesive medical devices.
Key Topics
- Tissue Adhesive Strength Evaluation: The standard specifies a testing process to determine the shear strength when tissue adhesives are subjected to tension loading in a single-lap-shear configuration.
- Substrate Selection: Guidance is given for using appropriate substrates, including fresh or frozen porcine skin for comparative tests and model substrates for quality control purposes.
- Test Specimens: Criteria are detailed for preparing test specimens, ensuring proper alignment, size, and moisture levels to simulate clinical conditions.
- Testing Conditions: Recommendations include test temperature, specimen conditioning, and environmental controls to ensure consistent results.
- Measurement and Reporting: The standard requires calculation of maximum load, shear strength, and detailed reporting of test conditions, adhesive types, substrate characteristics, and failure modes.
- Quality Control: The method outlines procedures for batch consistency checks in manufacturing, enabling evaluation of lot-to-lot consistency and effects of surface treatments.
Applications
ASTM F2255 is widely applicable within the medical device and biomaterials sectors, particularly for:
- Surgical adhesives and sealants: Evaluating the mechanical strength of products used for wound closure, hemostasis, and prevention of fluid leakage.
- Product development: Enabling comparative analysis of new tissue adhesive formulations, ensuring they meet mechanical performance requirements.
- Quality assurance and manufacturing: Providing a standardized approach for ongoing quality control of tissue adhesive devices, verifying consistency, and detecting process variations.
- Research and regulatory assessments: Supporting laboratories and regulatory professionals in generating reliable data for scientific publication and regulatory submissions.
Note: The test method is particularly relevant for comparative analysis, batch testing, and screening the effects of formulation or substrate variations. However, results from this single-lap-shear test are not sufficient as an exclusive basis for clinical design stress determination, due to the variability of clinical applications.
Related Standards
- ASTM D907 - Terminology of Adhesives: Provides definitions for adhesive-related terminology referenced in this standard.
- ASTM E4 - Practices for Force Verification of Testing Machines: Ensures calibration and verification of testing apparatus.
- Standards for Tissue Banking (American Association of Tissue Banks): Offers guidelines for handling biological tissue used in testing.
- Other ASTM F04 Standards: Covering medical and surgical materials and related test methods.
Conclusion
ASTM F2255-05(2015) plays a critical role in promoting the consistent and rigorous assessment of tissue adhesive products, enhancing patient safety, product reliability, and the advancement of medical technologies. This standard is valuable for researchers, manufacturers, and quality assurance professionals seeking reliable, reproducible, and comparable data on the mechanical strength of tissue adhesives in healthcare applications.
Keywords: ASTM F2255, tissue adhesive, lap-shear, shear strength, surgical adhesives, quality control, medical device testing, biomaterials, adhesive testing standard.
Buy Documents
ASTM F2255-05(2015) - Standard Test Method for Strength Properties of Tissue Adhesives in Lap-Shear by Tension Loading
Get Certified
Connect with accredited certification bodies for this standard

Control Union Certifications
Global certification for agriculture and sustainability.

Smithers Quality Assessments
US management systems and product certification.

Bureau Veritas Bangladesh
Bureau Veritas certification services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM F2255-05(2015) is a standard published by ASTM International. Its full title is "Standard Test Method for Strength Properties of Tissue Adhesives in Lap-Shear by Tension Loading". This standard covers: SIGNIFICANCE AND USE 4.1 Materials and devices that function at least in part by adhering to living tissues are finding increasing use in surgical procedures either as adjuncts to sutures and staples, or as frank replacements for those devices in a wide variety of medical procedures. While the nature and magnitude of the forces involved varies greatly with indication and with patient specific circumstances, all uses involve to some extent the ability of the material to resist imposed mechanical forces. Therefore, the mechanical properties of the materials, and in particular the adhesive properties, are important parameters in evaluating their fitness for use. In addition, the mechanical properties of a given adhesive composition can provide a useful means of determining product consistency for quality control, or as a means for determining the effects of various surface treatments on the substrate prior to use of the device. 4.2 The complexity and variety of individual applications for tissue adhesive devices, even within a single indicated use (surgical procedure) is such that the results of a single-lap-shear test are not suitable for determining allowable design stresses without thorough analysis and understanding of the application and adhesive behaviors. 4.3 This test method may be used for comparing adhesives or bonding processes for susceptibility to fatigue and environmental changes, but such comparisons must be made with great caution since different adhesives may respond differently to varying conditions. SCOPE 1.1 This test method is intended to provide a means for comparison of the adhesive strengths of tissue adhesives intended for use as surgical adhesives or sealants, or both, on soft tissue. With the appropriate choice of substrate, it may also be used for purposes of quality control in the manufacture of tissue adhesive based medical devices. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
SIGNIFICANCE AND USE 4.1 Materials and devices that function at least in part by adhering to living tissues are finding increasing use in surgical procedures either as adjuncts to sutures and staples, or as frank replacements for those devices in a wide variety of medical procedures. While the nature and magnitude of the forces involved varies greatly with indication and with patient specific circumstances, all uses involve to some extent the ability of the material to resist imposed mechanical forces. Therefore, the mechanical properties of the materials, and in particular the adhesive properties, are important parameters in evaluating their fitness for use. In addition, the mechanical properties of a given adhesive composition can provide a useful means of determining product consistency for quality control, or as a means for determining the effects of various surface treatments on the substrate prior to use of the device. 4.2 The complexity and variety of individual applications for tissue adhesive devices, even within a single indicated use (surgical procedure) is such that the results of a single-lap-shear test are not suitable for determining allowable design stresses without thorough analysis and understanding of the application and adhesive behaviors. 4.3 This test method may be used for comparing adhesives or bonding processes for susceptibility to fatigue and environmental changes, but such comparisons must be made with great caution since different adhesives may respond differently to varying conditions. SCOPE 1.1 This test method is intended to provide a means for comparison of the adhesive strengths of tissue adhesives intended for use as surgical adhesives or sealants, or both, on soft tissue. With the appropriate choice of substrate, it may also be used for purposes of quality control in the manufacture of tissue adhesive based medical devices. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
ASTM F2255-05(2015) is classified under the following ICS (International Classification for Standards) categories: 59.080.01 - Textiles in general; 83.180 - Adhesives. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F2255-05(2015) has the following relationships with other standards: It is inter standard links to ASTM F2255-05(2010), ASTM E4-14, ASTM D907-12a, ASTM D907-12, ASTM D907-11a, ASTM D907-11, ASTM E4-10, ASTM E4-09a, ASTM E4-09, ASTM E4-08, ASTM D907-08b, ASTM D907-08a, ASTM D907-08, ASTM E4-07, ASTM D907-06. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F2255-05(2015) is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F2255 − 05 (Reapproved 2015)
Standard Test Method for
Strength Properties of Tissue Adhesives in Lap-Shear by
Tension Loading
This standard is issued under the fixed designation F2255; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope for use in closing wounds (surgical or traumatic) or for sealing
against leakage of body fluids.
1.1 This test method is intended to provide a means for
comparison of the adhesive strengths of tissue adhesives
3.2.2 tissue sealant—a surface coating with adequate adhe-
intended for use as surgical adhesives or sealants, or both, on
sive strength to prevent leakage of body fluids.
softtissue.Withtheappropriatechoiceofsubstrate,itmayalso
be used for purposes of quality control in the manufacture of
4. Significance and Use
tissue adhesive based medical devices.
4.1 Materials and devices that function at least in part by
1.2 The values stated in SI units are to be regarded as
adhering to living tissues are finding increasing use in surgical
standard. No other units of measurement are included in this
procedureseitherasadjunctstosuturesandstaples,orasfrank
standard.
replacements for those devices in a wide variety of medical
1.3 This standard does not purport to address all of the procedures. While the nature and magnitude of the forces
safety concerns, if any, associated with its use. It is the involvedvariesgreatlywithindicationandwithpatientspecific
responsibility of the user of this standard to establish appro- circumstances,allusesinvolvetosomeextenttheabilityofthe
priate safety and health practices and determine the applica- material to resist imposed mechanical forces. Therefore, the
bility of regulatory limitations prior to use. mechanical properties of the materials, and in particular the
adhesive properties, are important parameters in evaluating
2. Referenced Documents
their fitness for use. In addition, the mechanical properties of a
given adhesive composition can provide a useful means of
2.1 ASTM Standards:
determining product consistency for quality control, or as a
D907Terminology of Adhesives
meansfordeterminingtheeffectsofvarioussurfacetreatments
E4Practices for Force Verification of Testing Machines
on the substrate prior to use of the device.
2.2 American Association of Tissue Banks Standards:
Standards for Tissue Banking 4.2 The complexity and variety of individual applications
for tissue adhesive devices, even within a single indicated use
3. Terminology (surgical procedure) is such that the results of a single-lap-
shear test are not suitable for determining allowable design
3.1 Definitions—Many terms in this test method are defined
stresses without thorough analysis and understanding of the
in Terminology D907.
application and adhesive behaviors.
3.2 Definitions:
4.3 This test method may be used for comparing adhesives
3.2.1 tissue adhesive—for the purposes of this test method,
or bonding processes for susceptibility to fatigue and environ-
tissue adhesive is defined as a compound or system intended
mentalchanges,butsuchcomparisonsmustbemadewithgreat
caution since different adhesives may respond differently to
varying conditions.
ThistestmethodisunderthejurisdictionofASTMCommitteeF04onMedical
andSurgicalMaterialsandDevicesandisthedirectresponsibilityofSubcommittee
F04.15 on Material Test Methods.
5. Apparatus
CurrenteditionapprovedMay1,2015.PublishedJuly2015.Originallyapproved
in 2003. Last previous edition approved in 2010 as F2255–05 (2010). DOI:
5.1 Testing Machine, of the constant-rate-of-crosshead-
10.1520/F2255-05R15.
movement type and comprising essentially the following:
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM 5.1.1 Fixed Member, a fixed or essentially stationary mem-
Standards volume information, refer to the standard’s Document Summary page on
ber carrying one grip.
the ASTM website.
5.1.2 Movable Member, a movable member carrying a
Available from the American Association of Tissue Banks (AATB), 1350
Beverly Rd., Suite 220-A, McLean, VA 22101. second grip.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2255 − 05 (2015)
5.1.3 Grips,forholdingthetestspecimenbetweenthefixed AmericanAssociation of Tissue Banks. The specimens should
member and the movable member of the testing machine can be brought to the test temperature or other prescribed tempera-
be either the fixed or self-aligning type. ture (such as body temperature) prior to application of the
5.1.3.1 Fixed Grips are rigidly attached to the fixed and adhesive.
movable members of the testing machine. When this type of 6.2.2 Fixed tissue should not be used since it has been
gripisusedextremecareshouldbetakentoensurethatthetest demonstrated that fixatives cause large alterations in the
specimen is inserted and clamped so that the long axis of the mechanical properties of the tissue and it is probable that the
test specimen coincides with the direction of pull through the adhesive strength would be affected as well.
centerline of the grip assembly. 6.2.3 Ifthetargetorganisofasizeorgeometry,orboth,that
5.1.3.2 Self-aligning Grips are attached to the fixed and does not allow fabrication of test samples as shown in Fig. 1,
movablemembersofthetestingmachineinsuchamannerthat a tissue of similar origin but larger size should be used. For
they will move freely into alignment as soon as any load is example, if the intended indication is for anastomosis of small
applied so that the long axis of the test specimen will coincide blood vessels, a larger vessel should be substituted.
with the direction of the applied pull through the center line of 6.2.4 The thickness of the tissue sample should be mini-
the grip assembly. The specimens should be aligned as per- mized and should not exceed 5 mm.Thicker samples will lead
fectly as possible with the direction of pull so that no rotary to distortion of the substrate and mixed loading (shear and
motion that may induce slippage or damage to the sample will tension).Itisalsoimportantthatthethicknessbeasuniformas
occur in the grips; there is a limit to the amount of misalign- possible.
ment self-aligning grips will accommodate.
6.3 Substrates for Quality Control Testing:
5.1.4 Drive Mechanism,forimpartingtothemovablemem-
6.3.1 For testing that is undertaken as part of a quality
berauniform,controlledvelocitywithrespecttothestationary
control process in the manufacturing of a tissue adhesive
member, with this velocity to be regulated as specified in 9.3.
device, the use of freshly harvested tissue is highly inconve-
5.1.5 Load Indicator, a suitable load-indicating mechanism
nient and may also lead to unacceptable variation in the test
capable of showing the total tensile load carried by the test
results, especially if the failure occurs in the adherend (sub-
specimen when held by the grips. This mechanism shall be
strate failure). Since the purpose of quality control testing is to
essentially free of inertia lag at the specified rate of testing and
demonstrate consistency in the device, substitution of a model
shall indicate the load with an accuracy of 61% of the
substrate is preferred so long as it is demonstrated that the
indicated value, or better. The accuracy of the testing machine
adhesive does bond to the adherand. For devices that require
shall be verified in accordance with Practices E4.
contact with tissues to cure, Mediskin XenoGraft should be
5.2 Temperature-controlling Equipment, capable of main- used for quality control testing as well as comparative testing.
taining the test temperature to 62°C. If ambient laboratory
7. Test Specimen
conditionsareemployedthesamedegreeofcontrolisrequired.
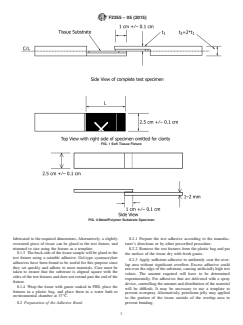
7.1 Specimens with Soft-tissue Substrates shall conform to
Awaterbathorenvironmentalchambercapableofmaintaining
the form shown in Fig. 1.The length of the tissue substrate (L)
37°C is required for testing on tissue substrates.
attached to each specimen holder should be at least 1.5 times
6. Test Substrate
the length of the overlap area in order to ensure that the failure
occurs at the overlap bond and doesn’t pull the tissue substrate
6.1 For comparative testing, either fresh or frozen split
off of the specimen holder. For very strong adhesives, L may
thickness porcine skin graft may be used.
need to be 2 to 3 times the overlap length. The tissue can be
6.1.1 Frozen split thickness porcine skin that has been
bonded to the specimen holder with any suitable adhesive.
aseptically prepared is available commercially and is preferred
Gel-type cyanoacrylate adhesives have been found to be
due to ease of use and the potential for more consistent
convenient for this purpose since they adhere well to moist
properties.Itshouldbethawedaccordingtothemanufacturer’s
tissues and cure quickly.
instructions prior to use. Unused graft may be kept at 2 to 8°C
for up to two weeks after thawing.
7.2 Specimens with Polymer or Metal Substrates shall
6.1.2 Iffreshskinischosen,itshouldbepreparedaccording
conform to the form and dimensions shown in Fig. 2.
to the method in Appendix X1.
7.3 Number of Test Specimens—Test at least 10 specimens
6.2 Application Specific Testing:
of each type. Discard results if failure occurs between the test
6.2.1 The strength of any adhesive is highly dependent on
fixture and the tissue sample and test additional samples to
the test substrate, or adherend. For a specific application, the
obtain a total of 10 valid tests. Tissue substrates tend to give
preferred substrate is freshly harvested tissue from the target
higher variances and may require more samples to attain a
organ of a domestic food animal. Tissue from bovine, porcine,
reasonable estimate of the mean strength.
orovineoriginispreferredduetowideavailabilityandthefact
8. Sample Preparation
that relatively large samples of tissue can be harvested from a
single source. Ideally, the tissue should be used within 24 h of 8.1 Tissue Preparation:
harvest,andshouldbekeptbetween5and10°Cpriortotesting 8.1.1 Tissue substrate materials should be kept moist at all
if it cannot be used immediately after harvesting. Storage and times with phosphate buffered saline (PBS).
handling of tissue samples should be carried out according to 8.1.2 The substrate will be cut to the dimensions shown in
the guidelines set forth in Standards forTissue Banking by the Fig. 1 using a template and a fresh scalpel blade or a cutter
F2255 − 05 (2015)
FIG. 1 Soft Tissue Fixture
FIG. 2 Met
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...