ASTM D8345-21
(Test Method)Standard Test Method for Determination of an Emission Parameter for Phthalate Esters and Other Non-Phthalate Plasticizers from Planar Polyvinyl Chloride Indoor Materials for Use in Mass Transfer Modeling Calculations
Standard Test Method for Determination of an Emission Parameter for Phthalate Esters and Other Non-Phthalate Plasticizers from Planar Polyvinyl Chloride Indoor Materials for Use in Mass Transfer Modeling Calculations
SIGNIFICANCE AND USE
5.1 The conventional approach for characterizing VOC emissions from building materials and products (for example, Test Methods D6007 and D8142; Practices D6177, D6330, D6670, D6803, D7143; Guide D5116; and ISO 16000-6 standards) results in modeling VOC emissions in the indoor environment using area-specific emission rates (µg h-1 m-2). These approaches work for most chemicals classified as VOCs, because most VOC emissions are controlled by the internal mass transfer processes (diffusion of the chemical through the material) and most VOCs sorb to minimal extent to chamber walls. Hence, chamber area-specific emission rates can be directly applied to models of indoor environments.
5.2 In contrast, chemicals classified as SVOCs will sorb strongly to chamber walls and are controlled by the external mass transfer process (migration through the air boundary layer on the material surface). When used for the equilibrium gas phase concentration of certain SVOCs above source materials, conventional chamber emission characterization approaches are typically time-consuming taking up to several months due to sorption of analytes to chamber walls. Due to SVOC’s external mass transfer limitation, the SVOC area-specific emission rate (µg h-1 m-2) measured in a test chamber can be different from that for the same material in a real indoor environment. To accurately model SVOC concentrations in indoor environments, a mass transfer approach to determine gas phase concentrations in equilibrium with the material phase is needed.
5.3 Modeling emissions in a real environment using a mass transfer framework requires knowledge of the convective mass transfer coefficient (hm), the initial SVOC concentration in the material (Co), the diffusion coefficient in the material (D), and the concentration in the air immediately above the material surface (y0). Typically, the convective mass transfer coefficient, hm, and diffusion coefficient, D, can be estimated. The initial concentration in the...
SCOPE
1.1 Planar polyvinyl chloride (vinyl) indoor materials can contain semi-volatile organic compounds (SVOCs), such as phthalate esters and other non-phthalate plasticizers, that can emit into indoor air. Phthalate esters and other non-phthalate plasticizers that have been measured using this standard are listed in Table 1 and are referred to as SVOCs in the remainder of this document.
1.2 The SVOCs listed in Table 1 are present in a wide range of products and not limited to planar polyvinyl chloride (vinyl) indoor materials. This standard discusses specific planar polyvinyl chloride materials due to method development and associated quality control data produced from testing these materials. The materials inclusion in this standard does not indicate the SVOC source strength of specific polyvinyl chloride planar materials relative to other products.
1.3 This method describes the design of a 1 L environmental chamber with minimal exposed chamber walls.
1.4 This method measures the steady-state gas phase concentration of SVOCs in the chamber. Samples of products are tested at specified conditions of temperature, airflow rate, and elapsed time in a specially designed chamber with dry air. Air samples are collected periodically using sorbent sampling tubes at the chamber exhausts at controlled flow rates, and then analyzed by thermal desorption-gas chromatography-mass spectrometry (TD-GC-MS).
1.5 This method determines the SVOC convective gas-phase mass transfer coefficient across the material surface, hm, from the known dimethyl phthalate mass transfer coefficient in the chamber (1).2
1.6 Using the steady-state gas phase concentration and mass transfer coefficient, the method estimates the gas-phase concentration of SVOC in equilibrium with the material phase (y0) at a specified temperature. The obtained y0 data can be used to predict emissions in real indoor environments. However, exposure modeling is beyond t...
General Information
- Status
- Published
- Publication Date
- 28-Feb-2021
- Technical Committee
- D22 - Air Quality
- Drafting Committee
- D22.05 - Indoor Air
Relations
- Effective Date
- 01-Sep-2023
- Effective Date
- 01-Sep-2020
- Effective Date
- 15-Mar-2020
- Effective Date
- 01-Mar-2020
- Effective Date
- 01-Nov-2019
- Effective Date
- 01-Aug-2019
- Effective Date
- 01-Sep-2018
- Effective Date
- 01-Nov-2017
- Effective Date
- 01-Oct-2017
- Effective Date
- 01-Oct-2017
- Effective Date
- 01-Mar-2017
- Effective Date
- 01-Oct-2016
- Effective Date
- 01-Nov-2015
- Effective Date
- 15-Oct-2015
- Effective Date
- 01-Jul-2015
Overview
ASTM D8345-21 is a standard test method developed by ASTM International for determining emission parameters of phthalate esters and other non-phthalate plasticizers-categorized as semi-volatile organic compounds (SVOCs)-from planar polyvinyl chloride (PVC) indoor materials. The data produced by this method support accurate mass transfer modeling calculations essential for predicting and managing SVOC concentrations and emissions in indoor environments. Unlike volatile organic compounds (VOCs), SVOC emissions are governed by external mass transfer and tend to sorb to chamber walls, requiring specialized test setups. This standard presents a robust approach to measure and model SVOC emissions, contributing to healthier indoor air quality and informed material choices.
Key Topics
- Purpose and Scope: Focuses on measuring gas-phase concentrations of SVOCs, including commonly used plasticizers in PVC materials such as phthalate esters, under controlled environmental conditions.
- Specialized Chamber Design: Uses a 1 L stainless steel environmental chamber with minimal exposed surfaces apart from the test material, facilitating reliable SVOC emission measurements.
- Sample Preparation and Measurement:
- Samples of planar PVC materials are exposed to controlled temperatures, airflow, and time.
- Air sampling is conducted using sorbent tubes, later analyzed by thermal desorption-gas chromatography-mass spectrometry (TD-GC-MS).
- Mass Transfer Parameters:
- Determines key parameters like the convective mass transfer coefficient (hm) and steady-state gas-phase concentration (y0), critical for mass transfer modeling.
- Rapid and Accurate Measurement: Reduces testing time compared to conventional chamber methods, enabling more efficient evaluation for real-world indoor applications.
Applications
- Indoor Air Quality Modeling: Provides foundational data to model SVOC emissions from building materials for use in exposure and risk assessments.
- Product Development and Quality Control: Assists manufacturers in evaluating the emission profiles of plasticizers from PVC products such as vinyl flooring, wall coverings, and mattress covers.
- Regulatory Compliance: Supports compliance with environmental and health regulations concerning indoor air pollutants, including phthalates and other SVOCs.
- Material Selection and Building Design: Offers architects, builders, and facility managers scientific data to inform material choices and design decisions for improved indoor environmental quality.
- Comparative Emission Performance: Though the method discusses PVC, principles and data may extend to a wider range of SVOC-containing indoor products.
Related Standards
- ASTM D6007: Determination of formaldehyde concentrations in air from wood products using small-scale chambers.
- ASTM D8142: Testing of chemical emissions from spray polyurethane foam insulation.
- ASTM D5116: Guide for small-scale environmental chamber determinations of organic emissions.
- ISO 16000-6: International standard for the determination of volatile organic compounds in indoor and chamber air.
- EPA Method 8270E: Analysis of semivolatile organic compounds by GC/MS.
- CPSC-CH-C1001-09.4: Procedure for determination of phthalates in consumer products.
Practical Value
Implementing ASTM D8345-21 enables stakeholders to:
- Obtain reliable, rapid measurements of SVOC emissions from PVC and similar materials, crucial for safe indoor environments.
- Generate accurate emission data for input in advanced indoor air models, supporting public health evaluations and product sustainability initiatives.
- Harmonize testing practices with international indoor air quality standards, paving the way for global acceptance of building products.
- Enhance transparency and comparability of material emissions data, contributing to better consumer confidence and regulatory adherence.
By adopting this standard, laboratories, manufacturers, regulators, and building professionals gain a scientifically sound tool for addressing contemporary indoor air quality concerns related to plasticizer emissions.
Buy Documents
ASTM D8345-21 - Standard Test Method for Determination of an Emission Parameter for Phthalate Esters and Other Non-Phthalate Plasticizers from Planar Polyvinyl Chloride Indoor Materials for Use in Mass Transfer Modeling Calculations
Get Certified
Connect with accredited certification bodies for this standard

NSF International
Global independent organization facilitating standards development and certification.

Smithers Quality Assessments
US management systems and product certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.
Sponsored listings
Frequently Asked Questions
ASTM D8345-21 is a standard published by ASTM International. Its full title is "Standard Test Method for Determination of an Emission Parameter for Phthalate Esters and Other Non-Phthalate Plasticizers from Planar Polyvinyl Chloride Indoor Materials for Use in Mass Transfer Modeling Calculations". This standard covers: SIGNIFICANCE AND USE 5.1 The conventional approach for characterizing VOC emissions from building materials and products (for example, Test Methods D6007 and D8142; Practices D6177, D6330, D6670, D6803, D7143; Guide D5116; and ISO 16000-6 standards) results in modeling VOC emissions in the indoor environment using area-specific emission rates (µg h-1 m-2). These approaches work for most chemicals classified as VOCs, because most VOC emissions are controlled by the internal mass transfer processes (diffusion of the chemical through the material) and most VOCs sorb to minimal extent to chamber walls. Hence, chamber area-specific emission rates can be directly applied to models of indoor environments. 5.2 In contrast, chemicals classified as SVOCs will sorb strongly to chamber walls and are controlled by the external mass transfer process (migration through the air boundary layer on the material surface). When used for the equilibrium gas phase concentration of certain SVOCs above source materials, conventional chamber emission characterization approaches are typically time-consuming taking up to several months due to sorption of analytes to chamber walls. Due to SVOC’s external mass transfer limitation, the SVOC area-specific emission rate (µg h-1 m-2) measured in a test chamber can be different from that for the same material in a real indoor environment. To accurately model SVOC concentrations in indoor environments, a mass transfer approach to determine gas phase concentrations in equilibrium with the material phase is needed. 5.3 Modeling emissions in a real environment using a mass transfer framework requires knowledge of the convective mass transfer coefficient (hm), the initial SVOC concentration in the material (Co), the diffusion coefficient in the material (D), and the concentration in the air immediately above the material surface (y0). Typically, the convective mass transfer coefficient, hm, and diffusion coefficient, D, can be estimated. The initial concentration in the... SCOPE 1.1 Planar polyvinyl chloride (vinyl) indoor materials can contain semi-volatile organic compounds (SVOCs), such as phthalate esters and other non-phthalate plasticizers, that can emit into indoor air. Phthalate esters and other non-phthalate plasticizers that have been measured using this standard are listed in Table 1 and are referred to as SVOCs in the remainder of this document. 1.2 The SVOCs listed in Table 1 are present in a wide range of products and not limited to planar polyvinyl chloride (vinyl) indoor materials. This standard discusses specific planar polyvinyl chloride materials due to method development and associated quality control data produced from testing these materials. The materials inclusion in this standard does not indicate the SVOC source strength of specific polyvinyl chloride planar materials relative to other products. 1.3 This method describes the design of a 1 L environmental chamber with minimal exposed chamber walls. 1.4 This method measures the steady-state gas phase concentration of SVOCs in the chamber. Samples of products are tested at specified conditions of temperature, airflow rate, and elapsed time in a specially designed chamber with dry air. Air samples are collected periodically using sorbent sampling tubes at the chamber exhausts at controlled flow rates, and then analyzed by thermal desorption-gas chromatography-mass spectrometry (TD-GC-MS). 1.5 This method determines the SVOC convective gas-phase mass transfer coefficient across the material surface, hm, from the known dimethyl phthalate mass transfer coefficient in the chamber (1).2 1.6 Using the steady-state gas phase concentration and mass transfer coefficient, the method estimates the gas-phase concentration of SVOC in equilibrium with the material phase (y0) at a specified temperature. The obtained y0 data can be used to predict emissions in real indoor environments. However, exposure modeling is beyond t...
SIGNIFICANCE AND USE 5.1 The conventional approach for characterizing VOC emissions from building materials and products (for example, Test Methods D6007 and D8142; Practices D6177, D6330, D6670, D6803, D7143; Guide D5116; and ISO 16000-6 standards) results in modeling VOC emissions in the indoor environment using area-specific emission rates (µg h-1 m-2). These approaches work for most chemicals classified as VOCs, because most VOC emissions are controlled by the internal mass transfer processes (diffusion of the chemical through the material) and most VOCs sorb to minimal extent to chamber walls. Hence, chamber area-specific emission rates can be directly applied to models of indoor environments. 5.2 In contrast, chemicals classified as SVOCs will sorb strongly to chamber walls and are controlled by the external mass transfer process (migration through the air boundary layer on the material surface). When used for the equilibrium gas phase concentration of certain SVOCs above source materials, conventional chamber emission characterization approaches are typically time-consuming taking up to several months due to sorption of analytes to chamber walls. Due to SVOC’s external mass transfer limitation, the SVOC area-specific emission rate (µg h-1 m-2) measured in a test chamber can be different from that for the same material in a real indoor environment. To accurately model SVOC concentrations in indoor environments, a mass transfer approach to determine gas phase concentrations in equilibrium with the material phase is needed. 5.3 Modeling emissions in a real environment using a mass transfer framework requires knowledge of the convective mass transfer coefficient (hm), the initial SVOC concentration in the material (Co), the diffusion coefficient in the material (D), and the concentration in the air immediately above the material surface (y0). Typically, the convective mass transfer coefficient, hm, and diffusion coefficient, D, can be estimated. The initial concentration in the... SCOPE 1.1 Planar polyvinyl chloride (vinyl) indoor materials can contain semi-volatile organic compounds (SVOCs), such as phthalate esters and other non-phthalate plasticizers, that can emit into indoor air. Phthalate esters and other non-phthalate plasticizers that have been measured using this standard are listed in Table 1 and are referred to as SVOCs in the remainder of this document. 1.2 The SVOCs listed in Table 1 are present in a wide range of products and not limited to planar polyvinyl chloride (vinyl) indoor materials. This standard discusses specific planar polyvinyl chloride materials due to method development and associated quality control data produced from testing these materials. The materials inclusion in this standard does not indicate the SVOC source strength of specific polyvinyl chloride planar materials relative to other products. 1.3 This method describes the design of a 1 L environmental chamber with minimal exposed chamber walls. 1.4 This method measures the steady-state gas phase concentration of SVOCs in the chamber. Samples of products are tested at specified conditions of temperature, airflow rate, and elapsed time in a specially designed chamber with dry air. Air samples are collected periodically using sorbent sampling tubes at the chamber exhausts at controlled flow rates, and then analyzed by thermal desorption-gas chromatography-mass spectrometry (TD-GC-MS). 1.5 This method determines the SVOC convective gas-phase mass transfer coefficient across the material surface, hm, from the known dimethyl phthalate mass transfer coefficient in the chamber (1).2 1.6 Using the steady-state gas phase concentration and mass transfer coefficient, the method estimates the gas-phase concentration of SVOC in equilibrium with the material phase (y0) at a specified temperature. The obtained y0 data can be used to predict emissions in real indoor environments. However, exposure modeling is beyond t...
ASTM D8345-21 is classified under the following ICS (International Classification for Standards) categories: 13.040.20 - Ambient atmospheres; 83.140.99 - Other rubber and plastics products. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D8345-21 has the following relationships with other standards: It is inter standard links to ASTM D8142-23, ASTM D1356-20a, ASTM D1356-20, ASTM D6330-20, ASTM D6177-19, ASTM D6803-19, ASTM D6670-18, ASTM D5116-17, ASTM D8142-17e1, ASTM D8142-17, ASTM D7143-17, ASTM D7143-11(2016), ASTM D6196-15, ASTM D1356-15a, ASTM D1356-15. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D8345-21 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D8345 − 21
Standard Test Method for
Determination of an Emission Parameter for Phthalate
Esters and Other Non-Phthalate Plasticizers from Planar
Polyvinyl Chloride Indoor Materials for Use in Mass Transfer
Modeling Calculations
This standard is issued under the fixed designation D8345; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope centrationofSVOCinequilibriumwiththematerialphase(y )
at a specified temperature.The obtained y data can be used to
1.1 Planar polyvinyl chloride (vinyl) indoor materials can
predict emissions in real indoor environments. However, ex-
contain semi-volatile organic compounds (SVOCs), such as
posure modeling is beyond the scope of this method. For more
phthalate esters and other non-phthalate plasticizers, that can
information on mass transfer emission and exposure modeling
emit into indoor air. Phthalate esters and other non-phthalate
see Little et al. (2), Liang and Xu (1, 3), and Guo (4).
plasticizers that have been measured using this standard are
listedinTable1andarereferredtoasSVOCsintheremainder 1.7 The results for gas phase concentration change in the
of this document. chamber with time, steady-state gas phase SVOC concentra-
tions(y ),and y ,onlyrepresenttheconditionsspecifiedinthe
ss 0
1.2 TheSVOCslistedinTable1arepresentinawiderange
test method and are the result of assumptions built into the
ofproductsandnotlimitedtoplanarpolyvinylchloride(vinyl)
method such as instantaneous equilibrium at the source/air
indoor materials. This standard discusses specific planar poly-
interface. The results may not be representative of those
vinyl chloride materials due to method development and
collected under other test conditions (that is, temperature or
associated quality control data produced from testing these
flow rate) or comparable with other SVOC test methods.
materials. The materials inclusion in this standard does not
indicate the SVOC source strength of specific polyvinyl 1.8 The values stated in SI units are to be regarded as
chloride planar materials relative to other products. standard. No other units of measurement are included in this
standard.
1.3 Thismethoddescribesthedesignofa1Lenvironmental
1.9 This standard does not purport to address all of the
chamber with minimal exposed chamber walls.
safety concerns, if any, associated with its use. It is the
1.4 This method measures the steady-state gas phase con-
responsibility of the user of this standard to establish appro-
centration of SVOCs in the chamber. Samples of products are
priate safety, health, and environmental practices and deter-
tested at specified conditions of temperature, airflow rate, and
mine the applicability of regulatory limitations prior to use.
elapsed time in a specially designed chamber with dry air.Air
1.10 This international standard was developed in accor-
samples are collected periodically using sorbent sampling
dance with internationally recognized principles on standard-
tubesatthechamberexhaustsatcontrolledflowrates,andthen
ization established in the Decision on Principles for the
analyzed by thermal desorption-gas chromatography-mass
Development of International Standards, Guides and Recom-
spectrometry (TD-GC-MS).
mendations issued by the World Trade Organization Technical
1.5 This method determines the SVOC convective gas-
Barriers to Trade (TBT) Committee.
phase mass transfer coefficient across the material surface, h ,
m
from the known dimethyl phthalate mass transfer coefficient in 2. Referenced Documents
the chamber (1). 3
2.1 ASTM Standards:
1.6 Usingthesteady-stategasphaseconcentrationandmass D1193Specification for Reagent Water
transfer coefficient, the method estimates the gas-phase con- D1356Terminology Relating to Sampling and Analysis of
Atmospheres
This test method is under the jurisdiction of ASTM Committee D22 on Air
Quality and is the direct responsibility of Subcommittee D22.05 on Indoor Air.
Current edition approved March 1, 2021. Published December 2021. DOI: For referenced ASTM standards, visit the ASTM website, www.astm.org, or
10.1520/D8345-21. contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
The boldface numbers in parentheses refer to a list of references at the end of Standards volume information, refer to the standard’s Document Summary page on
this standard. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D8345 − 21
D5116Guide for Small-Scale Environmental Chamber De- 3.1.1 For definitions of terms commonly used for sampling
terminationsofOrganicEmissionsfromIndoorMaterials/ and analysis of atmospheres, refer to Terminology D1356. For
Products definitions and terms commonly used in volatile organic
D5337Practice for Flow RateAdjustment of Personal Sam- compound (VOC) emission testing from materials and prod-
pling Pumps uctsrefertoGuideD5116.Fordefinitionsandtermscommonly
D6007TestMethodforDeterminingFormaldehydeConcen- used in SVOC emission testing using a mass transfer frame-
trations in Air from Wood Products Using a Small-Scale work see Guide D8141.
Chamber
3.2 Definitions of Terms Specific to This Standard:
D6177Practice for Determining Emission Profiles of Vola-
3.2.1 loading factor, n—the ratio of the exposed test speci-
tile Organic Chemicals Emitted from Bedding Sets
men area to the chamber’s volume, usually expressed in
D6196Practice for Choosing Sorbents, Sampling Param- 2 3
m /m .
eters and Thermal Desorption Analytical Conditions for
Monitoring Volatile Organic Chemicals in Air
4. Summary of Test Method
D6330Practice for Determination of Volatile Organic Com-
4.1 A specially-designed stainless steel chamber is used to
pounds(ExcludingFormaldehyde)EmissionsfromWood-
rapidly measure SVOC additive emissions from planar poly-
Based Panels Using Small Environmental Chambers Un-
vinylchloridematerials.Thechamberdescribedinthismethod
der Defined Test Conditions
has a high area ratio of emission surface to sorption surfaces
D6670Practice for Full-Scale Chamber Determination of
(that is, chamber walls), reduces mass loss of SVOCs onto
Volatile Organic Emissions from Indoor Materials/
sampling pathways, and improves air mixing inside the cham-
Products
ber. This chamber has a reduced time to reach steady-state of
D6803PracticeforTestingandSamplingofVolatileOrganic
SVOC emissions compared to standard small chambers like
Compounds (Including Carbonyl Compounds) Emitted
those described in Guide D5116.
from Architectural Coatings Using Small-Scale Environ-
mental Chambers
4.2 Specificplanarpolyvinylchloridematerials,vinylfloor-
D7143Practice for Emission Cells for the Determination of
ing and mattress coverings, were used to develop this method
Volatile Organic Emissions from Indoor Materials/
andproducetheprecisiondataforthisstandard.Assuch,these
Products materials are discussed in this standard. Identification of these
D8141Guide for Selecting Volatile Organic Compounds
materials does not indicate SVOC source strength relative to
(VOCs) and Semi-Volatile Organic Compounds (SVOCs) other planar materials.
EmissionTestingMethodstoDetermineEmissionParam-
4.3 Representative test materials are prepared and placed to
eters for Modeling of Indoor Environments
form the top and bottom of the chamber. Clean dry air is
D8142Test Method for Determining Chemical Emissions
suppliedtothechamberandpassesovertheexposedsurfaceof
from Spray Polyurethane Foam (SPF) Insulation using
the test material before reaching the exhaust ports. Chamber
Micro-Scale Environmental Test Chambers
conditions such as temperature and airflow rate are controlled.
2.2 EPA Methods:
4.4 Air samples are collected periodically using sorbent
Method 8270ESemivolatile Organic Compounds by Gas
tubes at the chamber exhaust ports at controlled air sampling
Chromatography/Mass Spectrometry (GC/MS)
flow rates.
2.3 ISO Methods:
4.5 Sorbent tubes with air samples are analyzed by thermal
ISO 16000-6Determination of volatile organic compounds
desorption-gas chromatography/mass spectrometry (TD-GC-
in indoor and test chamber air by active sampling on
MS) to identify and quantify SVOC compounds. Information
TenaxTAsorbent,thermaldesorptionandgaschromatog-
on TD-GC-MS analysis can be found in ISO 16000-6, Annex
raphy using MS or MS-FID
D. Information specific to SVOC analysis can be found in
2.4 CPSC Methods:
Liang and Xu (1).
Test Method CPSC-CH-C1001-09.4 Standard Operating
4.6 Knowing the steady-state SVOC concentration in the
Procedure for Determination of Phthalates
chamber(y )andtheconvectivemasstransfercoefficient(h ),
ss m
the gas-phase concentration of SVOCs in equilibrium with the
3. Terminology
material phase (y ) at specified temperature and flow rate can
3.1 Definitions:
be quantified. The gas-phase concentration of SVOCs in
equilibriumwiththematerialphase(y )isneededtoaccurately
model exposure to SVOCs in indoor environments.
AvailablefromUnitedStatesEnvironmentalProtectionAgency(EPA),William
Jefferson Clinton Bldg., 1200 Pennsylvania Ave., NW, Washington, DC 20460,
https://www.epa.gov/hw-sw846/sw-846-test-method-8270e-semivolatile-organic-
5. Significance and Use
compounds-gas-chromatographymass-spectrometry.
5.1 The conventional approach for characterizing VOC
Available from International Organization for Standardization (ISO), ISO
Central Secretariat, Chemin de Blandonnet 8, CP 401, 1214 Vernier, Geneva,
emissions from building materials and products (for example,
Switzerland, https://www.iso.org.
Test Methods D6007 and D8142; Practices D6177, D6330,
Available from United States Consumer Product Safety Commission (CPSC),
D6670, D6803, D7143; Guide D5116; and ISO 16000-6
4330 East-West Highway Bethesda, MD 20814, https://www.cpsc.gov/Business--
Manufacturing/Testing-Certification/Lab-Accreditation/Test-Methods. standards) results in modeling VOC emissions in the indoor
D8345 − 21
-1 -2
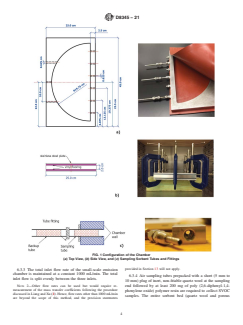
environment using area-specific emission rates (µg h m ). 6.1.2 As shown in Fig. 1, the thin chamber is positioned
TheseapproachesworkformostchemicalsclassifiedasVOCs, between two test sample sheets. The chamber is short semi-
because most VOC emissions are controlled by the internal cylindrical (40 cm in diameter×2cmin height) in shape.
mass transfer processes (diffusion of the chemical through the Exact dimensions of the chamber are shown in Fig. 1. The
material) and most VOCs sorb to minimal extent to chamber chamber walls are stainless steel. The top and bottom of the
walls. Hence, chamber area-specific emission rates can be chamber consist of two sheets of the tested sample. Air is
directly applied to models of indoor environments. injected into the circular side of the chamber and flows out the
portsontheflatside.Thedesignofthechambermaximizesthe
5.2 In contrast, chemicals classified as SVOCs will sorb
material emission area and minimizes the stainless steel
strongly to chamber walls and are controlled by the external
sorption area. Stainless steel plates (at least 2 mm thick) are
masstransferprocess(migrationthroughtheairboundarylayer
placed at the top and bottom of the chamber. Ten clamps or
on the material surface). When used for the equilibrium gas
screwsareusedtosealthechamber.Thechamberhasavolume
phase concentration of certain SVOCs above source materials,
2 3
of 1 L and a loading factor of 16 m /m . Users should ensure
conventional chamber emission characterization approaches
thattestmaterialsampleshaveanareathatextendsbeyondthe
are typically time-consuming taking up to several months due
perimeter of the chamber so that the edges of the material are
to sorption of analytes to chamber walls. Due to SVOC’s
notexposedtothestreamofairandthechambercanbesealed
external mass transfer limitation, the SVOC area-specific
-1 -2 properly.
emission rate (µg h m ) measured in a test chamber can be
6.1.3 Three inlets and six outlets enhance air flow mixing
different from that for the same material in a real indoor
inside the chamber and increase SVOC emissions (Fig. 1).
environment. To accurately model SVOC concentrations in
Eachinletshallhaveanelectronicmassflowcontrollercapable
indoor environments, a mass transfer approach to determine
of providing 333 mL/min of air (one third of the total airflow).
gasphaseconcentrationsinequilibriumwiththematerialphase
Outlet sampling flows shall also be controlled using electronic
is needed.
mass flow controllers.
5.3 Modeling emissions in a real environment using a mass
6.1.4 Construct the outlet ports to fit the used sorbent tubes,
transferframeworkrequiresknowledgeoftheconvectivemass
so that tubes can be directly inserted into the chamber without
transfer coefficient (h ), the initial SVOC concentration in the
m
any fittings. This avoids the loss of SVOCs to tubing and
material (C ), the diffusion coefficient in the material (D), and
o
fittings along the sampling pathway.
the concentration in the air immediately above the material
NOTE 1—The outlet ports can also be used to measure sorption of
surface(y ).Typically,theconvectivemasstransfercoefficient,
SVOCstostainlesssteelsurfacesandothersurfacesasdescribedbyLiang
h , and diffusion coefficient, D, can be estimated. The initial
m
and Xu (1), but it is not within the scope of this test method.
concentration in the material (C ) can be determined by means
o
6.1.5 Relatively thick (greater than 2 mm) planar polyvinyl
ofextraction.EPAMethod8270EandTestMethodCPSC-CH-
chloridematerialstypicallyserveasaneffectivegasketthatcan
C1001-09.4 can be used to determine bulk concentrations of
pass leakage tests. Polytetrafluoroethylene (PTFE) sheets (3
phthalates in materials. The unknown mass transfer emission
mm thick) are required for adequate sealing in tests of thinner
parameter required for exposure modeling in full-scale envi-
materials (1 mm thick, white layers in photo in Fig. 1b). Prior
ronments is the gas-phase concentration of SVOCs in equilib-
to testing determine if the chamber is leak free. The chamber
rium with the material phase (y ). This standard describes
can be considered sufficiently leak free if the outlet gas flow
procedures for rapidly determining y for phthalates from
rate is greater than 95 % of the inlet gas flow rate. Measure in
indoor planar polyvinyl chloride materials.
the flow rate in accordance with Practice D5337.
5.4 This method may be used to provide manufacturers,
6.2 Temperature-controlled Environmental Chamber:
builders, and end users with some of the input data (y )
6.2.1 A temperature-controlled environmental chamber or
required for models used to evaluating the impact of indoor
incubator must be used to ensure the chambers are operated at
planar, polyvinyl chloride materials on concentrations of in-
a constant temperature during the emission test. The small-
door SVOCs as well as for mass transfer exposure models.
scale emission chambers are placed in a 25 °C environmental
5.5 This method assumes that an instantaneous equilibrium
chamber that can evenly heat the chamber body and maintain
existsbetweengasphaseandmaterialsurface.Thisassumption
it at controlled temperatures with an accuracy of 61°Cand
has been made for a variety of SVOC mass transfer emission
precision of 61 °C at the set point. Interior chamber tempera-
and exposure models (see Little et al. (2), Liang and Xu (1, 3),
ture is verified using a NIST traceable device.
and Guo (4)). However, this assumption may be invalid under
6.3 Air Supply and Sampling System:
some environmental conditions.
6.3.1 Ultra-zero grade dry air either from a gas cylinder or
from a zero air generator is used to supply clean air to the
6. Apparatus
small-scale emission chambers. All tubing between the cylin-
6.1 Small-scale Emission Chamber:
der and the chambers should be 316 stainless steel or PTFE
6.1.1 The specially-designed emission chamber is made of where pressure appropriate.
316 stainless steel (SS) with an electropolished internal cham- 6.3.2 Massflowmetersormassflowcontrollersarerequired
ber surface. Chambers can also be used to measure sorption of for metering/setting the air flow rate through the emission
SVOCs to stainless steel surfaces and other materials/surfaces, chamber and air sampling tubes. An accuracy of 62 % and
but this is not within the scope of this method. precision of 63 % of the reading is needed.
D8345 − 21
FIG. 1 Configuration of the Chamber
(a) Top View, (b) Side View, and (c) Sampling Sorbent Tubes and Fittings
provided in Section 13 will not apply.
6.3.3 The total inlet flow rate of the small-scale emission
chamber is maintained at a constant 1000 mL/min. The total
6.3.4 Air sampling tubes prepacked with a short (5 mm to
inlet flow is split evenly between the three inlets.
10 mm) plug of inert, non-friable quartz wool at the sampling
NOTE 2—Other flow rates can be used but would require re- end followed by at least 200 mg of poly (2,6-diphenyl-1,4-
measurement of the mass transfer coefficients following the procedure
phenylene oxide) polymer resin are required to collect SVOC
discussed in Liang and Xu (1). Hence, flow rates other than 1000 mL/min
samples. The entire sorbent bed (quartz wool and porous
are beyond the scope of this method, and the precision statements
D8345 − 21
polymer sorbent) must fall within the heated zone of the as part of routine quality control, as described in Practice
thermal desorption unit. Sorbent tubes are commercially avail- D6196. Fig. 2 shows the schematic of a typical TD system
able and the most commonly used tubes are 8.9 cm long and operation.
6.4 mm outside diameter stainless steel tubes. Other tube sizes
NOTE3—Thetransferlinetemperatureusedtodevelopthismethodwas
and materials (glass) can be used if at least 180 mg of poly
250 °C. Optimization of different TD systems can result in different
(2,6-diphenyl-1,4-phenylene oxide) polymer resin is present
transfer line temperature requirements.
and breakthrough is checked on two tubes of the last set of
6.4.2 A GC-MS system is used to analyze SVOCs.
samples (see 10.2). Because only gas phase chemicals are
expected in chamber testing, a particle filter is not used.
7. Reagents and Materials
Connectbackuptubestooneofthethreeprimarysampletubes
7.1 Purity of Reagents—Reagent grade chemicals shall be
per sampling event to check for breakthrough (Fig. 1c).
used in all tests. Unless otherwise indicated, it is intended that
6.3.5 Three air sampling pumps are calibrated to a nominal
all reagents conform to the specifications of the Committee on
flow rate of 150 mL/min following Practice D5337, to ensure
Analytical Reagents of theAmerican Chemical Society where
high sorbent collection efficiencies of SVOCs and maintain 7
such specifications are available (5).
good flow distribution within the chamber. The flow rate of
7.2 Purity of Water—Unless otherwise indicated, references
pumps must be checked before and after sampling.The pumps
to water shall be understood to mean reagent water as defined
are placed outside the temperature-controlled environmental
by Type II of Specification D1193.
chamber with exhaust vents interfaced to a fume hood or other
exhaust system.
7.3 Standard Stock Solutions—An example list of potential
compounds for indoor planar polyvinyl chloride (vinyl) emis-
6.4 Chemical Analysis System:
sions determination is shown in Table 1. The compounds in
6.4.1 A thermal desorption (TD) system is required to
Table 1 have been measured emitting from historical vinyl
transfer SVOCs from the sorbent tube to a gas chromatograph
flooring and mattress covering samples. It is possible that the
(GC). In TD, the SVOCs are released from the sorbent by
compounds listed in Table 1 will not be present in currently
heating and are collected in a cold trap. Flash heating and
availablevinylflooringandmattresscoverings.Standardstock
purgingofthecoldtrapcarriesthesampletotheGCcolumnin
solutions of typical SVOCs are commercially available. In
a focused plug. Under this method, quartz wool is used at the
cases where standard stock solutions of SVOC chemicals are
inlet/outlet end of the cold trap to optimize retention, desorp-
unavailable for purchase, they can be prepared from pure
tionefficiencyandanalyterecoveryacrosstheSVOCvolatility
reagent and dilution solvents.
range. If a transfer line is used to transfer the sample from the
cold trap to the column, it should be uniformly heated, narrow
bore (to increase linear gas velocity and thus minimize risk of
ACS Reagent Chemicals, Specifications and Procedures for Reagents and
Standard-Grade Reference Materials, American Chemical Society, Washington,
analyte condensation onto surfaces) and inert (to avoid analyte
DC. For suggestions on the testing of reagents not listed by theAmerican Chemical
degradation). Analyte recovery through the entire thermal
Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset,
desorption process (desorption efficiency) –from the sample
U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharma-
tubetotransfer/injectionintotheGCcolumn–shouldbetested
copeial Convention, Inc. (USPC), Rockville, MD.
FIG. 2 Schematic Showing of a Typical TD System Operation
D8345 − 21
TABLE 1 List of SVOCs Measured Emitting from Historical Vinyl Flooring and Mattress Covering Samples
–1
Compounds Abbrev. CAS no. MW (g mol )
Di-n-butyl phthalate DnBP 84-74-2 278.4
Butyl benzyl phthalate BBP 85-68-7 312.4
Di(2-ethylhexyl) adipate DEHA 103-23-1 370.6
Di(2-ethylhexyl) phthalate DEHP 117-81-7 390.6
Di(2-ethylhexyl) isophthalate Iso-DEHP 137-89-3 390.6
Diisononyl phthalate DINP 28553-12-0 418.6
Diisononyl cyclohexane-1,2-dicarboxylate DINCH 474919-59-0 424.7
7.4 Hexane or Methanol—Solvent used to dilute standard contaminantsfromthecontainerair.Checkthetightnessofthe
solutions and cleaning apparatus. The solvents need to be end caps weekly using hand tightening and spanners.
regularly analyzed to monitor potential SVOC contamination.
8.2.3 Before sorbent tube sampling usage, one tenth of the
cleaned tubes should be randomly selected and analyzed to
7.5 Liquid Syringes—1 µL, 5 µL, 10 µL, 50 µL, and 100 µL
and 0.5 mLand 1 mLare used to prepare calibration solutions. verify that they do not contain target SVOCs. If any of the
tubes tested contain target SVOCs, all tubes must be tested
7.6 Nitrogen, ultra high-purity grade (99.999 %).
with only those tubes that are below detection limit values for
7.7 Air, ultra-zero grade or better.
target SVOCs used for sampling.
7.8 Helium, high-purity grade (best source).
8.3 Preparation of the Small-scale Emission Chamber:
7.9 Tube cleaner, instrument that heats sorbent tubes to a
8.3.1 Disconnectthechamberassemblyandcleanthecham-
predetermined temperature while flowing clean helium or
bercomponentsincludingthestainlesssteelplatesandfittings.
nitrogen through the tubes.
Use an alkali detergent followed by three separate water rinses
and three separate methanol rinses. Rinse all tubing with
8. Preparation of Apparatus
methanol. Dry the components thoroughly at an elevated
8.1 Preparation of SVOC Standards:
temperature of 80 °C for one hour.
8.1.1 If standard stock solutions of target SVOCs are not
8.3.2 Measure background SVOC concentrations for each
commercially available, prepare a standard stock solution of
emission chamber prior to using them for sample testing.
the these SVOCs by dissolving weighted amounts of the target
Background measurements are performed under normal oper-
compounds in hexane or methanol as described in Practice
ating conditions but without a material specimen in the
D6196.
chamber. Instead, PTFE sheets are used in the blank chambers
8.1.2 Prepare a working calibration standard mix from the
standard stock solution. Following guidance given in Practice as gaskets. Sample the chamber for 24 h in duplicate after 48
D6196, adjust the concentration of the target SVOCs in the h of steady gas flow and constant temperature. The chamber
standard mix solutions to reflect the range of mass expected in
canbeconsideredsufficientlycleaniftheconcentrationsinthe
samples. Dilute the standard stock solutions for each target
blank chamber are at least one tenth those found in samples.
SVOC to a concentration range of 2.5 mg/Lto 100 mg/L.This
Preliminary tests may be needed to determine these levels,
introducesacalibrationmassrangeof5ngto200ngbymeans
illustrative SVOC chamber concentrations can be found in (1,
ofa2µL injection.
6).
NOTE 4—Alternative mass ranges are also acceptable if more appropri-
8.4 Preparation of Test Materials:
ate for the samples and compounds being tested.
8.4.1 Cut new or used planar polyvinyl chloride materials
8.2 Preparation of Sorbent Tubes:
into two 45 cm × 25 cm sheets with a clean knife. Record the
8.2.1 Prepacked and pre-conditioned sorbent tubes must be
age and condition of the sample. Wrap the sample sheets
cleanedagainpriortousebyheatingto300°Cfor1hataflow
separately in clean aluminum foil and seal them in large
of 100 mL/min pure nitrogen or helium. Use a stainless steel
polyethylene terephthalate (PET) storage bags.
manifold inserted in an oven and mount the sorbent tubes with
8.4.2 During storage, protect samples from chemical con-
graphite or graphite (15 %)/polyimide (85 %) ferrules, check
tamination and from exposure to temperatures in excess of 25
the flows through each tube. Alternatively, a commercial tube
°C.
conditioningunitorthermaldesorptionunitcanbeusedforthe
cleaning. Desorb tubes at 300 °C for 20 min in cleaning mode
8.5 Preparation of Other Lab Equipment:
or as described in the TD manual. Freshly-packed but uncon-
8.5.1 Because laboratory equipment such as glassware and
ditionedsorbenttubesrequireextensivepreconditioningbefore
solvents may be contaminated with SVOCs, pre-wash all
use,typicallyforseveralhours.Followmanufacturer’sinstruc-
glassware. Use an alkali detergent followed by three separate
tions prior to use.
rinses with water and three separate rinses with methanol. Dry
8.2.2 Thecleanedsorbenttubesarefittedwithendcapswith
the glassware thoroughly at an elevated temperature of 80 °C
PTFE ferrules before sto
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...