ASTM D7614-20
(Test Method)Standard Test Method for Determination of Total Suspended Particulate (TSP) Hexavalent Chromium in Ambient Air Analyzed by Ion Chromatography (IC) and Spectrophotometric Measurements
Standard Test Method for Determination of Total Suspended Particulate (TSP) Hexavalent Chromium in Ambient Air Analyzed by Ion Chromatography (IC) and Spectrophotometric Measurements
SIGNIFICANCE AND USE
5.1 Hexavalent chromium is anthropogenic from a number of commercial and industrial sources. It readily penetrates biological membranes and has been identified as a carcinogen and industrial toxin. Hexavalent chromium is a known inhalation irritant and is associated with respiratory cancer (5).
5.2 Ambient atmospheric concentrations of hexavalent chromium are well below the detection limits of previous analytical methods utilized for the determination of hexavalent chromium (2).
5.3 Ambient atmospheric concentrations of hexavalent chromium provide a means of evaluating exposures in a manner that can be related to health-based risk levels. The data for samples collected in situ provide an improved basis for health assessments of potential exposures (5).
5.4 This test method provides step-by-step instructions for the sampling and analysis of total suspended ambient air particulates for hexavalent chromium.
5.5 This test method assumes that field and laboratory personnel are familiar with low volume ambient air sampling and hexavalent chromium analysis by ion chromatography with post-column derivatization. This method should not be performed for regulatory or compliance purposes until the field and laboratory personnel have demonstrated the ability to collect and analyze samples in such a manner as to pass the quality control requirements found in Section 13.
SCOPE
1.1 This test method specifies a procedure for the sampling and analysis of airborne particulate matter for hexavalent chromium in ambient air samples.
1.2 This method is applicable to the determination of masses of 0.40 to 20.0 ng of hexavalent chromium per sample without dilution. Detection limits vary by instrumentation. Some laboratories may be able to achieve lower detection limits. The lower limit of applicability for this method was determined in a 2019 multi-laboratory detection limit study (1).2
1.3 This method is applicable to hexavalent chromium measurement in the atmosphere from 0.019 to 0.926 ng/m3 assuming a 21.6 m3 sample volume. The lower range may be decreased with longer sampling times. The upper range can be increased using appropriate dilutions.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 29-Feb-2020
- Technical Committee
- D22 - Air Quality
- Drafting Committee
- D22.03 - Ambient Atmospheres and Source Emissions
Relations
- Effective Date
- 01-Mar-2020
- Effective Date
- 01-Feb-2024
- Effective Date
- 01-Sep-2020
- Effective Date
- 15-Mar-2020
- Effective Date
- 01-Aug-2019
- Effective Date
- 15-Aug-2018
- Effective Date
- 01-Jan-2018
- Effective Date
- 01-Nov-2017
- Effective Date
- 15-Oct-2015
- Effective Date
- 01-Jul-2015
- Effective Date
- 01-Dec-2014
- Effective Date
- 01-May-2014
- Effective Date
- 15-Jan-2014
- Effective Date
- 01-Dec-2011
- Effective Date
- 01-Oct-2011
Overview
ASTM D7614-20 is the Standard Test Method for the Determination of Total Suspended Particulate (TSP) Hexavalent Chromium in Ambient Air Analyzed by Ion Chromatography (IC) and Spectrophotometric Measurements. Published by ASTM International, this standard offers a systematic approach for sampling and analyzing airborne particulate matter for hexavalent chromium (Cr(VI)) in the ambient atmosphere. Hexavalent chromium is a hazardous air contaminant linked to carcinogenic and toxic effects, particularly from anthropogenic sources such as industrial processes. This method facilitates the detection of Cr(VI) at trace levels, contributing to air quality monitoring, public health risk assessments, and environmental compliance.
Key Topics
Health and Environmental Importance
Hexavalent chromium is a known carcinogen and industrial toxin that readily penetrates biological membranes, posing a significant inhalation hazard. Standardized detection in ambient air enables improved assessment of human exposure and associated health risks.Applicable Detection Ranges
The method is designed to quantify from 0.40 to 20.0 nanograms (ng) of hexavalent chromium per sample, with detection limits dependent on instrumentation. Atmospheric measurement is typically feasible in the 0.019 to 0.926 ng/m³ range, assuming a 21.6 m³ air sample.Sampling and Analytical Procedure
Air is sampled through an acid-washed, sodium bicarbonate-impregnated cellulose filter at a controlled flow rate. Collected particulates are extracted and analyzed using ion chromatography with post-column derivatization (using 1,5-diphenylcarbazide, DPC) and UV/Visible spectrophotometric detection.Quality Assurance
The standard emphasizes rigorous field and laboratory practices, including sample chain-of-custody, use of blanks, calibration, and proper equipment cleaning to minimize contamination and ensure data reliability.Minimization of Interconversion
Collection and analytical strategies are designed to minimize conversion between trivalent and hexavalent chromium, ensuring accurate measurement of the target analyte.
Applications
Ambient Air Monitoring
Regulatory agencies, environmental consultancies, and research institutions utilize this test method to monitor and report ambient levels of hexavalent chromium for air quality management.Health Risk Assessment
Data generated supports exposure evaluations and health-based risk assessments, particularly near industrial sites or urban areas with known chromium sources.Site Remediation and Compliance
The method is applicable for evaluating air quality prior to, during, and after site remediation activities involving chromium compounds, supporting compliance with environmental and occupational standards.Industrial Hygiene
While focused on ambient environments, the methodology can complement workplace air sampling and contribute to comprehensive assessments of chromium exposure.
Related Standards
ASTM D6832
Test Method for Determination of Hexavalent Chromium in Workplace Air by Ion Chromatography and Spectrophotometric Measurement.ASTM D4840
Guide for Sample Chain-of-Custody Procedures.ASTM D1193
Specification for Reagent Water.ASTM D1356 & D1357
Terminology and Practice for Sampling and Analysis of Atmospheres.ASTM D3195
Practice for Rotameter Calibration.EPA Methods and Guidance
Reference to Environmental Protection Agency (EPA) methods for complementary air monitoring protocols.
Practical Value
This ASTM standard streamlines the accurate and sensitive detection of airborne hexavalent chromium-a key element in environmental health studies and regulatory monitoring. The methodology supports trace-level quantification critical for compliance with increasingly stringent air quality and public health requirements. By following ASTM D7614-20, laboratories and field operators ensure the reliability, comparability, and legal defensibility of monitoring data related to this priority air toxicant.
Keywords: ASTM D7614-20, hexavalent chromium, total suspended particulates, ambient air, ion chromatography, spectrophotometric analysis, air quality, carcinogen monitoring, environmental compliance, trace metals, airborne contaminants
Buy Documents
ASTM D7614-20 - Standard Test Method for Determination of Total Suspended Particulate (TSP) Hexavalent Chromium in Ambient Air Analyzed by Ion Chromatography (IC) and Spectrophotometric Measurements
REDLINE ASTM D7614-20 - Standard Test Method for Determination of Total Suspended Particulate (TSP) Hexavalent Chromium in Ambient Air Analyzed by Ion Chromatography (IC) and Spectrophotometric Measurements
Get Certified
Connect with accredited certification bodies for this standard

NSF International
Global independent organization facilitating standards development and certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.

Kiwa BDA Testing
Building and construction product certification.
Sponsored listings
Frequently Asked Questions
ASTM D7614-20 is a standard published by ASTM International. Its full title is "Standard Test Method for Determination of Total Suspended Particulate (TSP) Hexavalent Chromium in Ambient Air Analyzed by Ion Chromatography (IC) and Spectrophotometric Measurements". This standard covers: SIGNIFICANCE AND USE 5.1 Hexavalent chromium is anthropogenic from a number of commercial and industrial sources. It readily penetrates biological membranes and has been identified as a carcinogen and industrial toxin. Hexavalent chromium is a known inhalation irritant and is associated with respiratory cancer (5). 5.2 Ambient atmospheric concentrations of hexavalent chromium are well below the detection limits of previous analytical methods utilized for the determination of hexavalent chromium (2). 5.3 Ambient atmospheric concentrations of hexavalent chromium provide a means of evaluating exposures in a manner that can be related to health-based risk levels. The data for samples collected in situ provide an improved basis for health assessments of potential exposures (5). 5.4 This test method provides step-by-step instructions for the sampling and analysis of total suspended ambient air particulates for hexavalent chromium. 5.5 This test method assumes that field and laboratory personnel are familiar with low volume ambient air sampling and hexavalent chromium analysis by ion chromatography with post-column derivatization. This method should not be performed for regulatory or compliance purposes until the field and laboratory personnel have demonstrated the ability to collect and analyze samples in such a manner as to pass the quality control requirements found in Section 13. SCOPE 1.1 This test method specifies a procedure for the sampling and analysis of airborne particulate matter for hexavalent chromium in ambient air samples. 1.2 This method is applicable to the determination of masses of 0.40 to 20.0 ng of hexavalent chromium per sample without dilution. Detection limits vary by instrumentation. Some laboratories may be able to achieve lower detection limits. The lower limit of applicability for this method was determined in a 2019 multi-laboratory detection limit study (1).2 1.3 This method is applicable to hexavalent chromium measurement in the atmosphere from 0.019 to 0.926 ng/m3 assuming a 21.6 m3 sample volume. The lower range may be decreased with longer sampling times. The upper range can be increased using appropriate dilutions. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 Hexavalent chromium is anthropogenic from a number of commercial and industrial sources. It readily penetrates biological membranes and has been identified as a carcinogen and industrial toxin. Hexavalent chromium is a known inhalation irritant and is associated with respiratory cancer (5). 5.2 Ambient atmospheric concentrations of hexavalent chromium are well below the detection limits of previous analytical methods utilized for the determination of hexavalent chromium (2). 5.3 Ambient atmospheric concentrations of hexavalent chromium provide a means of evaluating exposures in a manner that can be related to health-based risk levels. The data for samples collected in situ provide an improved basis for health assessments of potential exposures (5). 5.4 This test method provides step-by-step instructions for the sampling and analysis of total suspended ambient air particulates for hexavalent chromium. 5.5 This test method assumes that field and laboratory personnel are familiar with low volume ambient air sampling and hexavalent chromium analysis by ion chromatography with post-column derivatization. This method should not be performed for regulatory or compliance purposes until the field and laboratory personnel have demonstrated the ability to collect and analyze samples in such a manner as to pass the quality control requirements found in Section 13. SCOPE 1.1 This test method specifies a procedure for the sampling and analysis of airborne particulate matter for hexavalent chromium in ambient air samples. 1.2 This method is applicable to the determination of masses of 0.40 to 20.0 ng of hexavalent chromium per sample without dilution. Detection limits vary by instrumentation. Some laboratories may be able to achieve lower detection limits. The lower limit of applicability for this method was determined in a 2019 multi-laboratory detection limit study (1).2 1.3 This method is applicable to hexavalent chromium measurement in the atmosphere from 0.019 to 0.926 ng/m3 assuming a 21.6 m3 sample volume. The lower range may be decreased with longer sampling times. The upper range can be increased using appropriate dilutions. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D7614-20 is classified under the following ICS (International Classification for Standards) categories: 13.040.20 - Ambient atmospheres. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D7614-20 has the following relationships with other standards: It is inter standard links to ASTM D7614-12, ASTM E438-92(2024), ASTM D1356-20a, ASTM D1356-20, ASTM D1357-95(2019), ASTM D4840-99(2018)e1, ASTM E438-92(2018), ASTM E288-10(2017), ASTM D1356-15a, ASTM D1356-15, ASTM D1356-14b, ASTM D1356-14a, ASTM D1356-14, ASTM E438-92(2011), ASTM D1357-95(2011). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D7614-20 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D7614 − 20
Standard Test Method for
Determination of Total Suspended Particulate (TSP)
Hexavalent Chromium in Ambient Air Analyzed by Ion
Chromatography (IC) and Spectrophotometric
Measurements
This standard is issued under the fixed designation D7614; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 2. Referenced Documents
1.1 This test method specifies a procedure for the sampling 2.1 ASTM Standards:
and analysis of airborne particulate matter for hexavalent D1193 Specification for Reagent Water
chromium in ambient air samples. D1356 Terminology Relating to Sampling and Analysis of
Atmospheres
1.2 This method is applicable to the determination of
D1357 Practice for Planning the Sampling of the Ambient
masses of 0.40 to 20.0 ng of hexavalent chromium per sample
Atmosphere
without dilution. Detection limits vary by instrumentation.
D3195 Practice for Rotameter Calibration
Some laboratories may be able to achieve lower detection
D4840 Guide for Sample Chain-of-Custody Procedures
limits. The lower limit of applicability for this method was
E288 Specification for Laboratory Glass Volumetric Flasks
determined in a 2019 multi-laboratory detection limit study
E438 Specification for Glasses in Laboratory Apparatus
(1).
E1154 Specification for Piston or Plunger Operated Volu-
1.3 This method is applicable to hexavalent chromium
metric Apparatus
measurement in the atmosphere from 0.019 to 0.926 ng/m
assuming a 21.6 m sample volume. The lower range may be
3. Terminology
decreased with longer sampling times. The upper range can be
3.1 Definitions:
increased using appropriate dilutions.
3.1.1 For definitions of terms used in this test method, refer
1.4 The values stated in SI units are to be regarded as
to Terminology D1356.
standard. No other units of measurement are included in this
3.2 Definitions of Terms Specific to This Standard:
standard.
3.2.1 acid hardened filters, n—cellulose filters which have
1.5 This standard does not purport to address all of the
been acid-washed and solvent-rinsed by the manufacturer.
safety concerns, if any, associated with its use. It is the
3.2.2 cassette, n—a cartridge designed to contain a filter
responsibility of the user of this standard to establish appro-
through which air is pulled during sample collection. Cassettes
priate safety, health, and environmental practices and deter-
may be specific to a brand or model of sampler.
mine the applicability of regulatory limitations prior to use.
3.2.3 chain of custody (COC), n—a document that provides
1.6 This international standard was developed in accor-
for the traceable transfer of field samples to the analytical
dance with internationally recognized principles on standard-
laboratory.
ization established in the Decision on Principles for the
Development of International Standards, Guides and Recom- 3.2.4 eluent, n—the mobile phase used to transport the
mendations issued by the World Trade Organization Technical
sample through the ion chromatograph (IC) system, to include
Barriers to Trade (TBT) Committee. the UV/Vis detector.
3.2.5 field data sheet, n—a record that provides a reference
document for information directly related to the sample col-
This test method is under the jurisdiction of ASTM Committee D22 on Air
lection event, including pre- and post-calibration data.
Quality and is the direct responsibility of Subcommittee D22.03 on Ambient
Atmospheres and Source Emissions.
Current edition approved March 1, 2020. Published May 2020. Originally
approved in 2012. Last previous edition approved in 2012 as D7614 – 12. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
DOI:10.1520/D7614-20. contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
The boldface numbers in parentheses refer to a list of references at the end of Standards volume information, refer to the standard’s Document Summary page on
this standard. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D7614 − 20
3.2.6 quality assurance project plan (QAPP) or project biological membranes and has been identified as a carcinogen
plan, n—a document agreed upon by all stakeholders which and industrial toxin. Hexavalent chromium is a known inhala-
clearly delineates all aspects of the monitoring project, to tion irritant and is associated with respiratory cancer (5).
include siting of samplers, sampling days and frequency,
5.2 Ambient atmospheric concentrations of hexavalent
sampling and analytical protocols and quality assurance ele-
chromium are well below the detection limits of previous
ments.
analytical methods utilized for the determination of hexavalent
chromium (2).
4. Summary of Test Method (2-4)
5.3 Ambient atmospheric concentrations of hexavalent
4.1 This method captures ambient air particulate by pulling
chromium provide a means of evaluating exposures in a
a known volume of ambient air through an acid-washed
manner that can be related to health-based risk levels.The data
sodium bicarbonate-impregnated cellulose filter. A known
for samples collected in situ provide an improved basis for
volumeofairisdrawnatarateof5.0–16.0L/minfor24hours.
health assessments of potential exposures (5).
Particulate hexavalent chromium is stabilized on the impreg-
nated filter. 5.4 This test method provides step-by-step instructions for
the sampling and analysis of total suspended ambient air
4.2 After sampling, the chain of custody forms (COCs) are
particulates for hexavalent chromium.
completed and the filters are shipped to the laboratory in a
5.5 This test method assumes that field and laboratory
cooler with ice or cold packs that will keep filters at ≤0°C.
personnel are familiar with low volume ambient air sampling
Upon receipt, the filters are stored in a freezer at≤0°C prior to
and hexavalent chromium analysis by ion chromatography
sample extraction and analysis.
with post-column derivatization. This method should not be
4.3 The filters are extracted in 20 mM sodium bicarbonate.
performedforregulatoryorcompliancepurposesuntilthefield
The extract is analyzed by ion chromatography with
and laboratory personnel have demonstrated the ability to
post-column derivatization using a
collect and analyze samples in such a manner as to pass the
1,5-diphenylcarbazide (DPC) post-column reagent. Detection
quality control requirements found in Section 13.
is by ultraviolet/visible light (UV/Vis) detector set at 530 or
540 nm. The extract must be run within 24 hours from the end
6. Interferences
of extraction.
6.1 Sodium carbonate, when used to impregnate the sam-
4.4 The extract is analyzed by ion chromatography using a
pling filters, was observed to cause interferences with the
system comprised of a guard column, an analytical column, a
analysis (3).
post-column derivatization module including mixing coil, and
6.2 Higher concentrations of the sodium bicarbonate im-
a UV/Vis detector. During analysis, hexavalent chromium
pregnation solution (8.9) may cause flow restrictions during
exists as chromate due to the alkaline pH of the eluent. A
ambient air sampling (3).
minimum of 1000 µL of filtered extract is pumped through an
ion exchange column where the hexavalent chromium is
6.3 The use of an impregnated filter (11.2.2) of smaller pore
separated from other compounds.After separation through the
size has been shown to cause flow restrictions during ambient
column, the hexavalent chromium-containing eluent stream
air sampling (3).
mixes with the post-column reagent stream. The hexavalent
6.4 Several types of filters have been reported by multiple
chromium forms a complex with the
laboratories to contain trace amounts of hexavalent chromium
1,5 diphenylcarbazide (DPC) in the post-column reagent. The
which are detectable using this method. A variety of filter
combined liquid stream then flows through a UV/Vis detector
matrices including polyvinyl chloride (PVC), quartz, and
andtheresultantdiphenylcarbazonecomplexisdetectedat530
mixed cellulose esters (MCE) were found to have high
or 540 nm.
concentrations of hexavalent chromium and cannot be used for
6+
4.5 Hexavalent chromium is identified and quantified by
low level ambient Cr determinations (1, 4).
comparing its retention time and peak area to the correspond-
6.5 Conversion of trivalent chromium to hexavalent chro-
ing instrument responses of standard solutions of known
mium has been reported when the extraction temperature
concentration.
exceeds 24°C. This may be minimized by chilling the sonica-
4.6 Interconversion between trivalent and hexavalent chro-
tion bath water, or by using a mechanical shaker (wrist action
mium during sampling is minimized by using the sampling,
(7.9.1) or orbital (7.9.2)) at room temperature (4).
sample recovery, sample extraction, and analytical procedures
6.6 Conversion of trivalent to hexavalent chromium has
described in this standard.
been reported in unfiltered extracts stored at room temperature
4.7 Theholdtimebetweenthedateofsamplecollectionand
(6). This is minimized by filtering the extracts immediately
sample extraction is 21 days, stored frozen. The hold time
after extraction and storing them refrigerated at ≤4°C.
between sample extraction and sample analysis is 24 hours.
6.7 Trivalent iron, tetravalent titanium, pentavalent vana-
dium and hexavalent molybdenum form peaks on analytical
5. Significance and Use
systems utilizing Thermo AS7 separatory columns. Trivalent
5.1 Hexavalent chromium is anthropogenic from a number iron elutes off immediately prior to hexavalent chromium. In
of commercial and industrial sources. It readily penetrates large enough quantities, the trivalent iron may completely
D7614 − 20
obscure the hexavalent chromium peak. The peaks elute in the 7.1.2 Filter cassette, to hold the sample filter. All sampling
following chronological order: tetravalent titanium, hexavalent systemsshallensurethefilterdoesnotcontactanymetalatany
molybdenum, pentavalent vanadium, trivalent iron, hexavalent point in order to minimize the potential of hexavalent chro-
chromium (6). mium contamination.
7.1.2.1 Polytetrafluoroethylene (PTFE) filter cassette, all
6.8 Other published methods have reported copper, nickel,
components of this cassette which contact the sample stream
and mercury as interferents (7, 8).
are composed of PTFE. The inlet of the cassette is a mixing
6.9 Oxidizers such as acid fumes and reducing agents such
chamber to prevent particulate from accumulating only in the
as divalent iron are known to cause interconversion between
center of the filter. The filter support screen is composed of
the hexavalent and trivalent state (5, 9). The use of impreg-
PTFE.Theoutletofthecassettehasalargeenoughgasvolume
nated filters and room temperature extraction techniques may
toensureconsistentvacuumacrossthesurfaceofthefilter.The
minimize the amount of interconversion.
inletandoutlethalvesofthecassettearejoinedwithathreaded
6.10 In ambient airsheds, accuracy of data depends on coupling ring. The inlet and outlet stems are protected with
whether interconversion occurs in the airshed itself, that is, in vinyl caps. The filter is entirely encased within the cassette.
situ, or whether the conversion is a direct result of sampling or
During sampling, the cassette inlet faces the ground, and a
analytical processes (6). In the airshed, any interconversion glass funnel is attached to the inlet to prevent any precipitation
that occurs is not considered an interferent. Interconversion
from being entrained in the sample gas stream. See Fig. 2.
that is an artifact of sampling or analysis is considered an
7.1.2.2 FederalReferenceMethod(FRM)filtercassette,this
interferent. During sampling, a particle may be exposed to a
cassette is typically composed of a hard plastic such as high
much larger volume of ambient gasses than it would be if it
density polyethylene (HDPE) or acetal homopolymer resin.
were not trapped on a filter with gasses being drawn over its
The filter support screen is typically composed of stainless
surface. During analysis, some compounds may be solubilized
steel, although aluminum screens are available. The use of a
into the extract that, in the air shed, would be isolated from the
polyester drain disc between the filter and the metal support
hexavalent chromium. Once solubilized out of the particle(s)
screenensuresthatthefilterdoesnotcontactmetalatanypoint
and into the extraction solution, these interferent compounds
in time.
may then react with the hexavalent or trivalent chromium.
7.1.2.3 Secondary cassette containers, to minimize con-
Deducing whether interconversion is occurring in situ, during
tamination risks during shipment of cassettes. For PTFE
sampling, or during analysis, is not currently feasible, although
cassettes, this may consist of a 500 mL wide-mouth plastic
mechanisms are described in this method to attempt to limit
bottle.ForFRMcassettes,thismayconsistofananti-staticbag
interconversion.
or a clear plastic mailer, both designed to hold FRM cassettes.
Tertiary containers, if used, may consist of plastic freezer bags.
7. Apparatus
7.1.3 Filters,47mm,37mm,orothersuitablysizedashless,
7.1 Samplingsystem,capableofaccuratelysamplingatflow
cellulose filters. These filters must be acid-washed before use
rates of 5.0–16.0 L/min.
to remove any residual chromium. Filters which have been
acid-washed by the manufacturer are commercially available
NOTE 1—An example of a sampling system for ambient air consists of
and are commonly referred to as “acid hardened cellulose” by
anairinlet,acassettewithfilter,aflowmeter,avacuumpump,atimerand
a power supply as shown in Fig. 1.
the manufacturers.
7.1.4 Glass funnel, for use with the PTFE filter cassettes.
7.1.1 Sampling pumps, with an adjustable flow rate, capable
(See Fig. 2.)
of maintaining a consistent and constant flow rate over a
sampling period of up to 24 hours. Sampling pump flow rates 7.1.5 Flow control device, capable of controlling and mea-
must be calibrated before sampling begins. (See Practice suring selected sample gas flow rates to within 62 %. Rotame-
D3195 and Section 10.) ter control devices must be calibrated against a primary
FIG. 1 Hexavalent Chromium Sampling System in Ambient Air
D7614 − 20
FIG. 2 Diagram of Hexavalent Chromium PTFE Filter Cassette Assembly
standard (that is, a flowmeter whose accuracy is traceable to a 7.2.1 Pump, capable of delivering a constant flow of eluent
primary standard, see Practice D3195 and Section 10.) at the rate recommended by the manufacturer.
7.1.6 Elapsed timer, to be placed in line with the sample
7.2.2 Guard column,placedbeforetheseparatorycolumnto
pump to determine the total amount of time the sampler was in
remove particulate and organic contaminants from samples.
operation.
7.2.3 Separatory column, packed with a high capacity, high
7.1.7 Freezer, for storage of filters before and after sam-
efficiency, hydrophobic, anion exchange resin capable of sepa-
pling. Freezer temperatures must be maintained below 0°C at
rating hexavalent chromium from other metallic cations.
all times. A field freezer is not required, but highly recom-
7.2.4 Post-column reagent module, capable of delivering a
mended. The laboratory must have a freezer.All freezers must
constant flow of post-column reagent at one third of the rate of
have a means of monitoring the freezer temperature 7 days per
the eluent flow rate. The module may consist of a pneumatic
week, such as a max/min thermometer. If a max/min thermom-
delivery system or a pump, as long as the ratio of post-column
eterisused,itmustbeNIST-traceableandwithinitscalibration
expiration date. reagentflowtoeluentflowisconsistentandtheeluentflowrate
7.1.8 Cooler and cold packs (or dry ice), for transport of
is three-times the post-column reagent flow rate. It is impera-
filters to and from the sampling site. Cold packs may be any
tive that the ratio of eluent to post-column reagent be 3:1 to
form which can maintain a temperature of <0°C for the
ensure that the pH upon mixing of the two liquid streams is
duration of time that the filters are contained in the cooler.
such that the diphenylcarbazide can complex to the fullest
7.1.9 Flexible tubing, for use with sampling system (7.1).
extent possible during post-column derivatization.
7.1.10 Calibration system, soap bubble, rotameter, or mass
7.2.5 Reaction coil, capable of mixing two flowing liquid
flow calibration system to calibrate flow meters (10.1).
streams with minimal band spreading.
7.2 Analytical system, ion chromatograph, with the follow-
7.2.6 UV/Vis detector, flow through cell with UV/Vis absor-
ing components:
bance detector. The detection wavelength for hexavalent chro-
NOTE 2—A wide variety of instrumentation is commercially available. mium following this method is 530 or 540 nm, whichever has
Follow the instrument manufacturer’s guidelines for allowable instrument
the greatest response for a 1 ng/mL standard on a given ion
component operating pressures and other instrument-specific operational
chromatography system.
requirements.
D7614 − 20
7.2.7 Injection valve,withnon-metallicflowpathcomposed 7.11 Forceps,Polytetrafluoroethylene(PTFE)orplastic,for
of a largely inert material such as polyethylethylketone handling filters. Clean forceps prior to each use with reagent
(PEEK). water or ethanol.
7.2.8 Sample loop, minimum volume of 1 mL (1000 µL).
7.12 Thermometer or thermocouple, NIST-traceable, for
7.2.9 Autosampler with suitable autosampler vials and
measuringtemperatureofthelocalizedenvironmentbeforeand
caps, to provide consistent operation with better precision of
after sample extraction.
retention times.
7.2.10 Acquisition software, as provided by instrument 7.13 Extract storage containers, disposable plastic
manufacturer. (polystyrene, polypropylene, or polyethylene) tubes with
tightly fitting caps and a minimum working volume of 10 mL.
7.3 Fully adjustable, air-displacement pipets, for small-
volume dispensing of aqueous fluids of moderate viscosity and
7.14 Plastic syringe with Luer-Lok fitting and liquid sealed
density. Pipets should comply with Specification E1154 for
plunger, 10 mL, for filtering extracts after extraction.
piston operated volumetric devices.
7.15 Luer-Lok syringe filter, 0.2 µm pore size, for filtering
7.4 Volumetric flasks, made of borosilicate glass, 100 mL,
extracts after extraction.
200 mL, 1 L, and 2 L, which comply with Specification E288
7.16 pH strips, full range, for checking pH of cleaned filters
and Specification E438.
if following filter cleaning Option 1.
7.5 Analytical balance, for reagent preparation. Must have
accurate readability to 0.1 mg. 7.17 Drain Discs (only required if using FRM cassettes),
polyester, chemically inert, binder free, for preventing filter
7.6 Extraction vessels with caps, disposable plastic
from contacting metal support screen if using FRM cassettes.
(polystyrene, polypropylene, or polyethylene) tubes or cups
with tightly fitting caps and a minimum working volume of 10
8. Reagents
mL.Vessels must have sufficient depth to ensure that filters are
fully submerged in extraction solution during extraction.
8.1 Reagent grade chemicals shall be used in all tests.
Unless otherwise indicated, it is intended that all reagents
7.7 Petri dishes, disposable plastic, for storage of the
conform to the specifications of the Committee on Analytical
acid-washed, sodium bicarbonate-impregnated filters.
Reagents of the American Chemical Society where such
7.8 Impregnation area, with clean airshed and surfaces such
specifications are available. Other grades may be used, pro-
as:
vided it is first ascertained that the reagent is of sufficiently
7.8.1 Option1—Nitrogenpurgedgloveboxes(2)oneboxto
high purity to permit its use without lessening the accuracy of
be used to prepare the filters before sampling, the second box
the determination.
to be used to prepare the filters for post-sampling extraction
8.2 Purity of Water—Unless otherwise indicated, references
and analysis. The boxes must be airtight with a double-layered
to water shall be understood to mean reagent water as defined
closed gasket system and contain suspended plastic-coated
by Type I of Specification D1193.
screens to hold filters during preparation. Purge the boxes with
ultra-pure nitrogen.
8.3 Reagent Water, ASTM Type I, ≥18.2 MΩ.
7.8.2 Option 2—Dedicated filter handling room, maintained
8.4 Ammonium sulfate ((NH ) SO ), 99.999 % purity based
largely dust free, equipped with HEPA filtered HVAC system
4 2 4
on trace metals.
and HEPAfiltered laminar flow hood. Similar to that described
in EPA PM2.5 Guidance Document 2.12 (10).
8.5 Ammonium hydroxide (NH OH), reagent grade,
7.8.3 Option 3—Any clean area in which filters are cleaned, 3
28.0–30.0 % NH4, specific gravity 0.899 g/cm .
impregnated,dried,andhandledinsuchamannerastopassthe
8.6 1,5-diphenylcarbazide (DPC), crystalline. When fully
10 % prescreening requirements described in 11.2.2.5.
dissolved in methanol, no visible particulate should be present
7.9 Ultrasonicator, to be used for reagent preparation,
and solution should be colorless.
standard preparation when standards are not purchased
commercially, and filter extraction if using extraction Option 1
8.7 Methanol (CH OH), HPLC grade, greater than 99.9 %
(11.5.2.1). purity, specific gravity 0.79 g/cm .
7.9.1 (Optional) wrist action shaker, for filter extraction
8.8 Sulfuric acid (H SO ), concentrated, specific gravity
2 4
Option 2, operating at 385 oscillations/minute with a tilt of
1.84 g/cm .
7.5°.
7.9.2 (Optional) orbital shaker, for filter extraction Option 8.9 Sodium bicarbonate (NaHCO ), 99.5+ % purity.
3, operating at a minimum of 200 RPM.
7.10 Disposable PVC gloves, for prevention of sample
contamination during sampling and analysis. Nitrile and latex
ACS Reagent Chemicals, Specifications and Procedures for Reagents and
Standard-Grade Reference Materials, American Chemical Society, Washington,
gloves react with the post-column DPC reagent to form a color
DC. For suggestions on the testing of reagents not listed by theAmerican Chemical
that absorbs at 530 or 540 nm (6). Use of gloves made from
Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset,
any material other than PVC must be carefully considered with
U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharma-
regard to the likelihood of contamination of the filter. copeial Convention, Inc. (USPC), Rockville, MD.
D7614 − 20
8.10 Potassium dichromate (K Cr O ), 99.99+ % purity mix thoroughly. Depending on the laboratory environment, the
2 2 7
based on trace metals, crystalline. For use in preparing stan- intermediate primary standard solution can be stable for up to
twelve months.
dards. Dry at 105°C for 1 hour then cool in a desiccator prior
to use. Alternatively, potassium chromate (K Cr O ) may be
8.14.3 Hexavalent Chromium Calibration Solutions, to be
2 2 4
used (see Note 3). prepared in 20 mM sodium bicarbonate extraction solution. A
minimum of six standards are prepared to span the range of
8.11 Sodium Bicarbonate Extraction Solution (20 mM), in a
0.04 to 2.0 ng/mLby diluting appropriate volumes of the 1000
2 L volumetric flask, dissolve 3.36 g of sodium bicarbonate
ng/mL intermediate standard solution (8.14.2) with 20 mM
(8.9) in reagent water (8.3). Dilute to volume with reagent
sodium bicarbonate extraction solution (8.11). Depending on
water and mix thoroughly.
the laboratory environment, the calibration standard solutions
can be stable for up to twelve months.
8.12 Eluent, 250 mM ammonium sulfate (8.4)/100 mM
ammonium hydroxide (8.5)—Ina2L volumetric flask, dis-
NOTE4—Calibrationstandardconcentrationsbelow0.04ng/mLmaybe
solve 66 g of ammonium sulfate in approximately 1500 mL
used if the laboratory’s analytical system has a lower detection limit than
reagent water (8.3). Add 13 mL of ammonium hydroxide. that determined in the 2019 multi-laboratory study (1). For NELAC
accredited laboratories, the lowest calibration standard must be at the
Dilutetovolumewithreagentwaterandmixthoroughly.Other
lowest concentration reported to the client without qualifiers (11).
eluents or eluent concentrations are acceptable if all quality
control samples meet their acceptance criteria. 8.14.4 Hexavalent Chromium Intermediate Secondary Stan-
dard Solution, 1000 ng/mL, to be prepared in 20 mM sodium
8.13 Post-Column Derivatization Reagent, in a 500 mL
bicarbonate extraction solution. In a 100 mL volumetric flask,
volumetric flask, add approximately 300 mL of reagent water
add approximately 75 mL extraction solution (8.11) and 100
(8.3), then carefully add 14 mL of concentrated sulfuric acid
µL of the secondary source stock standard solution (8.14.1)
(8.8) and mix thoroughly. Allow to cool after mixing. In a 50
pending on the laboratory environment, the working interme-
mL volumetric flask, dissolve 0.25 g of 1,5-diphenylcarbazide
diate standard solution can be stable for up to twelve months.
(8.6) in 50 mLof HPLC-grade methanol (8.7). Sonication may
8.14.5 Hexavalent Chromium Initial and Continuing Cali-
be used to facilitate DPC dissolution. Add DPC-methanol
bration Verification Standard Solution, concentration at or
solution to sulfuric acid solution. Dilute to 500 mL with
below the mid-point of the calibration curve, to be prepared in
reagent water and mix thoroughly. Depending on laboratory
20 mM sodium bicarbonate extraction solution. In a 100 mL
conditions, this reagent may be stable for up to seven days. It
volumetric flask, add approximately 75 mLextraction solution
is suspected that exposure to UV light can increase the rate of
(8.11) and the appropriate amount of the intermediate second-
degradation of this reagent. Do not use if the reagent is darker
ary source standard solution (8.14.4). Dilute to volume with 20
than dark honey in color.
mMsodiumbicarbonatesolutionandmixthoroughly.Depend-
ing on laboratory environment, the secondary working stan-
8.14 Standard Solutions:
dard solution can be stable for up to twelve months.
8.14.1 Hexavalent Chromium Primary and Secondary Stock
6+ 8.14.6 Trivalent Chromium Stock Standard Solution (~1000
Standard Solutions (~1000 µg/mL Cr ), stock hexavalent
3+
µg/mL Cr ), stock trivalent chromium standards are available
chromium standards are available commercially or can be
commercially. The standard must be verified to contain negli-
prepared by dissolving 0.283 g of potassium dichromate (8.10)
6+
gible amounts of Cr prior to use for this method.
in 20 mM sodium bicarbonate extraction solution (8.11)ina
8.14.7 Trivalent Chromium Working Standard Solution,
100mLvolumetricflask.Dilutetovolumewith20mMsodium
1000 ng/mL, to be prepared in reagent water. In a 100 mL
bicarbonate extraction solution and mix thoroughly.
volumetric flask, add approximately 75 mLreagent water (8.3)
6+
8.14.1.1 Prepare two separate 1000 µg/mL Cr stock solu-
and 100 µL of the trivalent chromium stock standard solution
tionsorobtaintwocommerciallypreparedstocksolutionsfrom
(8.14.6). Dilute to volume with reagent water and mix thor-
separate sources.When a second manufacturer is not available,
oughly.Dependingonthelaboratoryenvironment,theworking
a primary stock solution from the same manufacturer, but from
standard solution can be stable for up to twelve months. A
a different manufacturing lot, may be used. The primary stock
small amount of concentrated nitric acid (<1% volume-to-
standard is used exclusively for calibration standards. The 3+ 6+
volume) may be added to prevent conversion of Cr to Cr .
secondary stock standard is used for all quality control stan-
8.15 Nitric Acid (HNO ), concentrated, trace metals grade,
dards and samples (Section 13) (11).
specific gravity 1.4 (g/cm ).
NOTE 3—Potassium chromate (K CrO ) may be used as an alternative
2 4
8.16 Acid Bath Solution (10 % Nitric Acid), ina1L
to potassium dichromate for the preparation of the hexavalent chromium
volumetric flask, add approximately 500 mL reagent water
stock standard solution. 0.373 g of K CrO are needed to make ~100 mL
2 4
of ~1000 µg/mL Cr6. (8.3) and 50 mL concentrated nitric acid (8.15), in that order.
Dilute to volume with reagent water and mix well. This
8.14.2 Hexavalent Chromium Intermediate Primary Stan-
solutionisonlyrequiredifusingfiltercleaningprotocolOption
dard Solution, 1000 ng/mL, to be prepared in 20 mM sodium
1(11.2.1.1).
bicarbonate extraction solution. In a 100 mL volumetric flask,
add approximately 75 mL extraction solution (8.11) and 100 8.17 Sodium Bicarbonate Impregnation Solution (0.12M),
µL of the primary source stock standard solution (8.14.1). in a 500 mL volumetric flask, dissolve 5.0 g of sodium
Dilute to volume with 20 mM sodium bicarbonate solution and bicarbonate (8.9) in reagent water (8.3). Sonication may be
D7614 − 20
used to facilitate dissolution. Dilute to volume with reagent 9.5.5.2 Program the sampler to initiate flow through the
water and mix thoroughly. entire sampling system.Adjust the sample collection flow rate
to the intended flow rate to be used during sampling.
9. Sampling 9.5.5.3 Allow the flow to continue for approximately two
minutes while the system attains operating temperature. After
9.1 Collect samples using a pre-prepared filter (11.2.2)
two minutes, verify that the flow rate matches the intended
containedinacassette(7.1.2)suitablefortheSamplingSystem
flow rate from 9.5.5.2; re-adjust if necessary. Document the
being employed. (See 11.1 through 11.4.) Wear gloves (7.10)
flow rate on the field data sheet.
when handling the filter cassettes, both pre- and post-sample
9.5.5.4 Program the sampler to collect a 24-hour sample on
collection.
the designated sample date.
9.2 For each sample, use a flow control device to maintain
9.5.6 Sample Recovery:
a relatively constant sample flow rate of 5–16 L/min over 24
9.5.6.1 Wear gloves (7.10) to remove the filter cassette
hours. The flow device can be a mass flow controller or a
(7.1.2.2) from the sampler. Retrieve the sample as soon as
rotameter.
possible and no later than 12 hours after the sampling system
9.3 Ensure the connecting lines between the filter cassette
has completed the run. If removing the filter from the filter
(7.1.2) and the sampling pump (7.1.1), if any, are as short as
cassette in the field, wear gloves and use only PTFE or plastic
possible to minimize the system residence time.
forceps (8.11).
9.4 Prior to use, calibrate all sampling system components
NOTE 5—Timely recovery of samples from the sampler is necessary to
3+ 6+
described in Section 10, “Calibration and Standardization.” If
prevent possible interconversion between Cr and Cr . To avoid
recovery timing issues, alternative sampling days or sampling start and
a rotameter is used, calibrate it in the field to determine true
end times may be used if approved by the regulator or the facility, or both,
readings (10.1).
or if specified as such in a QAPP(that is, Sampler 1 start time 0700 hours,
9.5 The following steps are provided for operation of a
Sampler 2 start time 0730 hours, Sampler 3 start time 0800 hours).
typical sampling system while collecting a sample:
9.5.6.2 For systems equipped with rotameters (7.1.5), verify
9.5.1 Assemble the sampling system.
the final flow by turning on the system and taking a reading
9.5.2 If performing field loading of filters into cassettes
after a two-minute warm-up period.
(7.1.2), wear gloves (7.10) and use PTFE or plastic forceps
9.5.6.3 Document the final flow, sample recovery date, and
(8.11) when removing filters from petri dishes and placing
total elapsed run time on the field data sheet.
them into filter cassettes. Load the filters as described in 11.3.
9.5.6.4 Record the start and end time of the sample collec-
9.5.3 Document all required site information on the field
tion event and the corresponding flow rate on the sampling
data sheet. Include, at a minimum, the following: site location,
field data sheet. Record the sample event name, sample type,
operator, filter set-up date, scheduled collection date, initial
location, and collection date and time on the field data sheet as
rotameter or mass flow controller reading, programmed start
well as any remaining required information and observations
and end times, and any additional comments deemed neces-
pertaining to the samples. Place the completed data sheet (if
sary.
paper sheets are used) in the cooler with the samples. If
9.5.4 Sample Collection Using PTFE Cassettes (7.1.2.1)
electronic field data sheets are used, save the file to an
(see Fig. 2):
appropriate device to avoid accidental deletion.
9.5.4.1 Remove the caps from the inlet and outlet of the
9.5.6.5 Carefully remove the sample filter cassette (7.1.2.2)
pre-loaded filter cassette. Attach the funnel stem (7.1.4)tothe
and funnel (if used) (7.1.4) from the sampling system and
inlet fitting of the cassette with tubing and tighten securely.
gently place the cassette in a clean secondary container
9.5.4.2 Connect the outlet of the cassette (7.1.2) and funnel
(7.1.2.3). Limit any jarring or sudden movements of the
assembly to the PTFE tube (7.1.9) that connects to the sample
cassettes while removing it from the samplers. See Note 6.
pump (7.1.1). If a collocated sample is scheduled, connect the
Place the secondary container containing the sample cassette
outlet of the collocated cassette to the collocated PTFE
into a cooler containing ice packs or dry ice.
connecting tube. (See Note 17.)
9.5.4.3 Program the sampler to initiate flow through the NOTE 6—Limitation of shaking, jarring, or jostling of the filter or filter
cassette during the sample recovery process is to prevent the loss of
entire sampling system.Adjust the sample collection flow rate
particulate matter from the sample filter.
to the intended flow rate to be used during sampling.
9.5.4.4 Allow the flow to continue for approximately two 9.5.6.6 If a freezer (7.1.7) is available for storage in the
minutes while the system attains operating temperature. After field, the cooler temperature is acceptable as long as the transit
two minutes, verify that the flow rate matches the intended time of the filter cassette between removal from the sampling
flow rate from 9.5.4.3; re-adjust if necessary. Document the system to placement into the field freezer is less than one hour
and the ice packs are still ≤0°C or dry ice (if used) is still
flow rate on the field data sheet.
9.5.4.5 Program the sampler to collect a 24-hour sample on visibly present.
the designated sample date. 9.5.6.7 If a freezer (7.1.7) is not available in the field, the
9.5.5 Sample Collection Using FRM Cassettes (7.1.2.2): cooler (7.1.8) must maintain a temperature of ≤0°C for the
9.5.5.1 Equip the sampling system with a pre-loaded FRM duration of the time that the filter or cassette is contained
cassette per the Manufacturer’s guidelines for the sampling within the cooler, whether the cooler is being used for storage
system being employed. in the field or shipment to the laboratory.
D7614 − 20
9.5.6.8 Ship the filters or filter cassettes to the laboratory as to Section 13 for acceptance criterion. If the ICV fails to meet
described in 11.4. the acceptance criterion, seek and rectify the cause of the
failure, then recalibrate.
10. Calibration and Standardization 10.2.6 Analyze an initial calibration blank (ICB) immedi-
ately after the ICV. Refer to Section 13 for acceptance
10.1 Sampling Calibration, required for samplers equipped
criterion. If the ICB fails to meet the acceptance criterion, seek
with rotameters.
and rectify the cause of the failure, then recalibrate.
10.1.1 Calibrate sample air flow rate using a primary
method of calibration at the beginning and end of sampling
11. Procedure
period.
11.1 Wheneverfiltersarehandled,usecleanPTFEorplastic
10.1.2 Use a rotameter, a soap bubbler or a mass flow
forceps (8.11) and wear disposable gloves (7.10). Complete all
calibrator system for calibration (7.1.10). Generic procedures
filter preparation, including cleaning of filters, impregnation of
are provided in 10.1.3 through 10.1.6.
filters, and cassette loading and unloading, in the clean
10.1.3 Wear gloves (7.10) during calibration to prevent
impregnation area (7.8). The impregnation area is demonstra-
sampler contamination.
10.1.4 Installafiltercassette(7.1.2)containingafilterinthe bly clean when the impregnation lot checks have no detectable
amounts of hexavalent chromium (11.2.2.5).
same manner as performed during routine sampling. Void this
filter when calibration is complete.
11.2 Filter Preparation:
10.1.5 Leak check the system:
11.2.1 Clean Filters:
10.1.5.1 Turn on the pump (7.1.1) and allow it to stabilize.
NOTE 7—The cleaning options below were validated during a 2019
10.1.5.2 Check the system for leaks by blocking the flow at
multi-laboratorystudy.Atotalof16blankfilterspreparedbytwodifferent
thecassetteinlet(PTFEcassettes(7.1.2.1))orsamplingsystem
laboratories using Option 1 and eight blank filters prepared by one
inlet (FRM cassettes (7.1.2.2)) and visually checking that flow
laboratory using Option 2 were analyzed by all three laboratories for a
is not occurring. If flow is detected, seek and eliminate the total of 24 blank filters analyzed. All blank filters returned results below
the detection limits of the laboratories performing the analysis (1).
leak(s).
10.1.5.3 Repeat the leak check until no flow is detected.
11.2.1.1 Option 1 for Cleaning Non-Acid Hardened Filters:
10.1.6 Calibrate the flow controller (7.1.5).
(1) Soak filters in fresh acid bath solution (8.16) for a
10.1.6.1 Turn on the sampler pump and allow it to stabilize.
minimum of 16 and a maximum of 24 hours.
10.1.6.2 After the flow rate has stabilized, attach the cali-
(2) Rinse filters thoroughly by soaking in reagent water
bration device to the cassette inlet (for PTFE cassettes) or
(8.3).Multipleexchangesofreagentwatermaybenecessaryto
sampling system inlet (for FRM cassettes).
fully rinse the acid from the filters.
10.1.6.3 Adjust the pump (7.1.1) so that the flow rate is set
(3) Check the pH of the filters by placing a pH strip (7.16)
to the intended flow rate to be used during sampling.
on top of a wet rinsed filter. If the pH of the wet filter matches
10.1.6.4 Record at least three calibration readings and the
thepHofthereagentwater(8.3),thefilterisfullyrinsed.Ifthe
average of the readings in a sampler log book. Additionally,
pH of the wet filter is lower than the pH of the reagent water,
record the sampler ID, date, time, and initials of the person
repeat the reagent water rinse. Discard the tested filter(s).
performing the calibration.
(4) Dry the filters in the impregnation area (7.8) until
10.1.6.5 Repeat steps 10.1.6.3 and 10.1.6.4 with two flow
opaque, stiff, and curled.
rates below the intended flow rate to be used during sampling,
NOTE 8—If the filters are not completely dry before placing them in the
and one flow rate above intended flow rate to be used during
impregnation solution, the solution can become diluted and the filters will
sampling, for a total of at least twelve flow readings. For
not collect samples as efficiently.
example, if the sampling flow rate during sampling is intended
11.2.1.2 Option 2 for Cleaning Acid Hardened Filters.
to be 15 L/min, the flow controller should be calibrated at 13,
(1) Place filters in a clean glass vessel with tightly fitting
14, 15 and 16 L/min, with three readings at each flow rate.
lid, such as a glass dish with a snap-on plastic lid equipped
10.2 Analytical Calibration: with a silicone gasket.
10.2.1 Prepare the initial calibration standards as described
(2) Add 0.12M sodium bicarbonate impregnation solution
in 8.14.1 through 8.14.3. (8.17) until all filters are submerged and move freely in the
10.2.2 Determine the instrument response in peak area for solution.
each hexavalent chromium standard (8.14.3) using the proce- (3) Place the lid on the container tightly such that no
dure described in 11.6.
sonicator water can enter the container. Sonicate for 15
10.2.3 Using least squares regression, prepare a calibration minutes.
curve using a linear plot of the standard concentration versus (4) Remove from sonicator and pour off the impregnation
peak area. solution.
10.2.4 Calibrate instrument (7.2) at the frequency required (5) Repeat steps 11.2.1.2 (2) through 11.2.1.2 (4) a second
by the QAPP, or, at a minimum, every time a fresh DPC time, for a total of two rounds of sonication.
reagent is made. 11.2.2 Impregnate Filters:
10.2.5 Analyze an initial calibration verification (ICV 11.2.2.1 Soak the filters in the sodium bicarbonate impreg-
(8.14.5)) immediately after calibration, at a concentration at or nation solution (8.17) overnight or for a minimum of 8 hours.
below the mid-point of the calibration curve (≤1 ng/mL). Refer 11.2.2.2 Discard the impregnation solution.
D7614 − 20
11.2.2.3 In the impregnation area (7.8), place the filters in a 11.3.2 FRM Cassette Loading and Unloading:
single layer on a clean surface. Dry the filters until opaque, 11.3.2.1 To load a filter into the cassette, separate the top of
stiff, and curled. the cassette from the bottom of the cassette. This may be
11.2.2.4 Place dried filters into Petri dishes (7.7) labeled achieved by use of a specialty piece of equipment designed for
with a unique impregnation batch identifier. Place the petri the purpose, or by prying the top and bottom of the cassette
dishes into secondary containers. Store in a freezer at ≤0°C apart by hand.
until needed.
11.3.2.2 Place a polyester drain disc (7.17) on top of the
filter screen.
NOTE 9—The use of freezers with metallic shelving or surfaces is not
11.3.2.3 Place an impregnated filter (11.2.2) on the drain
recommended due to potential for filter contamination.
disc (7.17), ensuring that the filter does not contact the metal
NOTE10—Filtersarestoredinafreezeruntilshippedtothefieldorused
to prepare quality control elements (13.3 and 13.4) during analysis. The
support screen, then place the other half of the cassette on top
filters are frozen to prevent sodium bicarbonate from reacting with
of the filter.
possible interfering substances present in air.
11.3.2.4 Securely seat the top of the cassette into the bottom
11.2.2.5 Analyze 10 % of the impregnated filters as de-
of the cassette.
scribedinsteps11.5and11.6toverifycleanliness.Ifanyofthe
11.3.2.5 Place the loaded cassette in a secondary container
verificationfiltersfromtheimpregnationbatchhasadetectable
such as an anti-static bag or plastic mailer (7.1.2.3). Place the
amount of hexavalent chromium, discard the entire batch and
secondary containers together in a tertiary container such as a
prepare a new batch.
sealed plastic bag for additional protection from contamina-
tion. Store and ship at ≤0°C.
11.3 Cassette loading and unloading. Post-sampling filters
11.3.2.6 To unload the filter from the cassette, separate the
shouldbehandledonlywithPTFEorplasticforceps(8.11)and
cassette as in step 11.3.2.1, ensuring that the filter does not
only on their edges where no sample deposit exists to prevent
contact the metal support screen. Push the support screen up
transfer of particulate from the filter onto the forceps. Unless
from behind, and carefully remove the filter without removing
specified in a QAPP, the decision of whether filter cassette
the drain disc. Place the sample filter into an extraction vessel
loadingandunloadingwillbeperformedinthelaboratoryorin
(7.6) and cap tightly. Discard the drain disc. If extraction is not
the field is determined by agreement between the parties
to be performed immediately, store the filters at ≤0°C.
performing sampling and those performing analysis.
11.4 Shipment and Storage of the Filters:
NOTE 11—Typically, the laboratory will have more control over the
cleanliness of the environment in which filter loading and unloading 11.4.1 Ship the filters or cassettes in a cooler packed with
occurs.
frozen ice packs. Dry ice may be substituted for ice packs,
however, “wet ice” (that is, frozen water) should not be used.
11.3.1 PTFE Cassette Loading and Unloading:
This shipping protocol applies to shipments from the
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D7614 − 12 D7614 − 20
Standard Test Method for
Determination of Total Suspended Particulate (TSP)
Hexavalent Chromium in Ambient Air Analyzed by Ion
Chromatography (IC) and Spectrophotometric
Measurements
This standard is issued under the fixed designation D7614; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This test method specifies a procedure for the sampling and analysis of airborne particulate matter for hexavalent chromium
in ambient air samples.
1.2 The method involves drawing 21.6 m ambient air (at 15 L/min for 24 hours) through a sodium bicarbonate cellulose acid
washed filter. Atmospheric hexavalent chromium is stabilized on the alkaline coated filter.
1.3 This method uses ion chromatography with post-column derivatization with 1,5-diphenylcarbazide (DPC) and a
Ultraviolet/Visible (UV/VIS) detector.
1.2 This method is applicable to the determination of masses of 0.100.40 to 20.0 ng of hexavalent chromium per sample without
dilution. Detection limits vary by instrumentation. Some laboratories may be able to achieve lower detection limits. The lower limit
of applicability for this method was determined in a 2019 multi-laboratory detection limit study (1).
1.3 This method is applicable forto hexavalent chromium measurement in the atmosphere from 0.004 ng/m0.019 to 0.926
3 3
ng/m assuming a 21.6 m sample volume. The range lower range may be decreased with longer sampling times. The upper range
can be increased using appropriate dilutions.
1.6 Interconversion of trivalent and hexavalent chromium during sampling is minimized to the extent possible by using these
sampling procedures.
1.7 The corresponding method for workplace air samples is ASTM Test Method D6832.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D4840 Guide for Sample Chain-of-Custody Procedures
D1193 Specification for Reagent Water
D1356 Terminology Relating to Sampling and Analysis of Atmospheres
D1357 Practice for Planning the Sampling of the Ambient Atmosphere
D3195 Practice for Rotameter Calibration
This test method is under the jurisdiction of ASTM Committee D22 on Air Quality and is the direct responsibility of Subcommittee D22.03 on Ambient Atmospheres
and Source Emissions.
Current edition approved April 1, 2012March 1, 2020. Published May 2012May 2020. DOI:10.1520/D7614-12.Originally approved in 2012. Last previous edition
approved in 2012 as D7614 – 12. DOI:10.1520/D7614-20.
The boldface numbers in parentheses refer to a list of references at the end of this standard.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D7614 − 20
D3586D4840 Test Method for Chromium in Workplace Atmospheres (Colorimetric Method)Guide for Sample Chain-of-
Custody Procedures (Withdrawn 1990)
D5281E288 Test Method for Collection and Analysis of Hexavalent Chromium in Ambient AtmospheresSpecification for
Laboratory Glass Volumetric Flasks
D6832E438 Test Method for the Determination of Hexavalent Chromium in Workplace Air by Ion Chromatography and
Spectrophotometric Measurement Using 1,5-diphenylcarbazideSpecification for Glasses in Laboratory Apparatus
E1154 Specification for Piston or Plunger Operated Volumetric Apparatus
3. Terminology
3.1 Definitions—Definitions:
3.1.1 For definitions of terms used in this test method, refer to Terminology D1356.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 acid hardened filters, n—cellulose filters which have been acid-washed and solvent-rinsed by the manufacturer.
3.2.2 cassette, n—a cartridge designed to contain a filter through which air is pulled during sample collection. Cassettes may
be specific to a brand or model of sampler.
3.2.3 chain of custody (COC), n—a document that provides for the traceable transfer of field samples to the analytical
laboratory.
3.2.4 eluent—eluent, n—the mobile phase used to transport the sample through the ion exchange column.chromatograph (IC)
system, to include the UV/Vis detector.
3.2.5 field data sheet, n—a record that provides a reference document for information directly related to the sample collection
event, including pre- and post-calibration data.
3.2.6 resolution—quality assurance project plan (QAPP) or project plan, n—the ability of a column to separate constituents
under specified test conditions.a document agreed upon by all stakeholders which clearly delineates all aspects of the monitoring
project, to include siting of samplers, sampling days and frequency, sampling and analytical protocols and quality assurance
elements.
4. Summary of Test Method (1, (2-42, )3, 4)
4.1 This method captures ambient air particulate by pulling a known volume of ambient air through an acid-washed sodium
bicarbonate-impregnated cellulose filter. A known volume of air is drawn through a sodium bicarbonate coated cellulose filter at
a rate of 9.0 to 16.0 5.0–16.0 L/min for 24 hours. Particulate hexavalent chromium is stabilized on the impregnated filter.
4.2 After sampling, the chain-of-custody forms are appropriately labeled chain of custody forms (COCs) are completed and the
filters are placed shipped to the laboratory in a cooler with ice for shipment to the laboratory. or cold packs that will keep filters
at ≤0°C. Upon receipt, the filters are placedstored in a freezer before preparation for at ≤0°C prior to sample extraction and
analysis.
4.3 The filters are extracted in 20 mM sodium bicarbonate. The extract is analyzed by ion chromatography with
post-column derivatization using a
1,5-diphenylcarbazide (DPC) post-column reagent. Detection is by ultraviolet/visible light (UV/Vis) detector set at 530 or 540 nm.
The extract must be run within 24 hours from the end of extraction.
4.4 The filters are extracted in sodium bicarbonate by means of sonication for one hour. The extract is analyzed by ion
chromatography using a system comprised of a guard column, an analytical column, a post-column derivatization module, module
including mixing coil, and a UV/VIS detector. In the analysis procedure, UV/Vis detector. During analysis, hexavalent chromium
exists as chromate due to the near neutral alkaline pH of the eluent. A minimum of 1000 μL of filtered extract is pumped through
an ion exchange column where the hexavalent chromium is separated from other compounds. After separation through the column,
the hexavalent chromium-containing eluent stream mixes with the post-column reagent stream. The hexavalent chromium forms
a complex with the
1,5-diphenylcarbazide (DPC) which can be 1,5 diphenylcarbazide (DPC) in the post-column reagent. The combined liquid stream
then flows through a UV/Vis detector and the resultant diphenylcarbazone complex is detected at 530 or 540 nm.
4.4 One blank filter, per 10 samples prepared, is also desorbed and analyzed in accordance with 4.3.
4.5 Hexavalent chromium is identified and quantified by comparing its retention time and peak area to the corresponding
standard solutions.instrument responses of standard solutions of known concentration.
4.6 Interconversion between trivalent and hexavalent chromium during sampling is minimized by using the sampling, sample
recovery, sample extraction, and analytical procedures described in this standard.
4.7 The hold time between the date of sample collection and sample extraction is 21 days, stored frozen. The hold time between
sample extraction and sample analysis is 24 hours.
D7614 − 20
5. Significance and Use
5.1 Hexavalent chromium is anthropogenic from a number of commercial and industrial sources. It readily penetrates biological
membranes and has been identified as an industrial toxic and cancer-causing substance.a carcinogen and industrial toxin.
Hexavalent chromium is a known inhalation irritant and is associated with respiratory cancer ((45).).
5.2 Ambient atmospheric concentrations of hexavalent chromium are well below the detection limits of standard sampling
methods, including Test Methodsprevious analytical methods utilized for the D3586 anddetermination of D5281 (hexavalent
chromium (52).).
5.3 Ambient atmospheric concentrations of hexavalent chromium providesprovide a means of evaluating exposures in a manner
that can be related to health-based risk levels. Collecting actual monitoring data provides The data for samples collected in situ
provide an improved basis for health assessments of potential exposures ((25).).
5.4 This test method provides step-by-step instructions for the sampling and analysis of hexavalent chromium collected on
sodium bicarbonate coated cellulose filters exposed to ambient air.total suspended ambient air particulates for hexavalent
chromium.
5.5 This test method assumes that field and laboratory personnel are familiar with low volume ambient air sampling and
hexavalent chromium analysis by ion chromatography with post-column derivatization. This method should not be performed for
regulatory or compliance purposes until the field and laboratory personnel have demonstrated the ability to collect and analyze
samples in such a manner as to pass the quality control requirements found in Section 13.
6. Interferences
6.1 Sodium carbonate, ifwhen used as the stabilizing medium for the hexavalent chromium to impregnate the sampling filters,
was observed to cause interferences with the analysis ((43).).
6.2 Higher concentrations of the sodium bicarbonate impregnatingimpregnation solution (8.9) may cause flow restrictions
during ambient air sampling ((43).).
6.3 The use of an impregnated filter (11.2.2) of smaller pore size has been shown to cause definite flow restrictions during
ambient air sampling.sampling (3).
6.4 Several types of filters have been determined reported by multiple laboratories to contain trace amounts of hexavalent
chromium which will leach out with time.are detectable using this method. A variety of filters filter matrices including polyvinyl
chloride (PVC), Quartz,quartz, and mixed cellulose esters (MCE) were found to have high concentrations of hexavalent chromium
6+
and can not cannot be used for low level ambient methods.Cr Cellulosedeterminations filters(1 can be acid washed and alkali
treated in order to stabilize and retain the hexavalent chromium following method specifications (, 4).).
6.5 Conversion of trivalent chromium to hexavalent chromium has been reported when the extraction temperature exceeds
24°C. This may be minimized by chilling the sonication bath water, or by using a mechanical shaker (wrist action (7.9.1) or orbital
(7.9.2)) at room temperature (4).
6.6 Conversion of trivalent to hexavalent chromium has been reported in unfiltered extracts stored at room temperature (6). This
is minimized by filtering the extracts immediately after extraction and storing them refrigerated at ≤4°C.
6.7 Trivalent iron, tetravalent titanium, pentavalent vanadium and hexavalent molybdenum form peaks on analytical systems
utilizing Thermo AS7 separatory columns. Trivalent iron elutes off immediately prior to hexavalent chromium. In large enough
quantities, the trivalent iron may completely obscure the hexavalent chromium peak. The peaks elute in the following
chronological order: tetravalent titanium, hexavalent molybdenum, pentavalent vanadium, trivalent iron, hexavalent chromium (6).
6.8 Other published methods have reported copper, nickel, and mercury as interferents (7, 8).
6.9 Oxidizers such as acid fumes and reducing agents such as divalent iron are known to cause interconversion between the
hexavalent and trivalent state (5, 9). The use of impregnated filters and room temperature extraction techniques may minimize the
amount of interconversion.
6.10 In ambient airsheds, accuracy of data depends on whether interconversion occurs in the airshed itself, that is, in situ, or
whether the conversion is a direct result of sampling or analytical processes (6). In the airshed, any interconversion that occurs is
not considered an interferent. Interconversion that is an artifact of sampling or analysis is considered an interferent. During
sampling, a particle may be exposed to a much larger volume of ambient gasses than it would be if it were not trapped on a filter
with gasses being drawn over its surface. During analysis, some compounds may be solubilized into the extract that, in the air shed,
would be isolated from the hexavalent chromium. Once solubilized out of the particle(s) and into the extraction solution, these
interferent compounds may then react with the hexavalent or trivalent chromium. Deducing whether interconversion is occurring
in situ, during sampling, or during analysis, is not currently feasible, although mechanisms are described in this method to attempt
to limit interconversion.
D7614 − 20
7. Apparatus
7.1 Sampling System,system, capable of accurately and precisely sampling 9.0 to 16.0 sampling at flow rates of 5.0–16.0 L/min.
NOTE 1—An example of a sampling system for ambient air consists of a filteran air inlet, a cassette with filter, a flow meter, a vacuum gage/pump,pump,
a timer and a power supply as shown in Fig. 1. In operation, ambient air is drawn through the filter assembly with a vacuum pump at a fixed flow rate
between 9 to 16 L/min.
7.1.1 Sampling pumps, with an adjustable flow rate, capable of maintaining a flow rate between 9.0 and 16.0 L/min through
consistent and constant flow rate over a sampling period of up to 24 hours. Sampling pump flow rates shallmust be calibrated
before sampling begins. (See Practice D3195 and Section 10.)
7.1.2 Polytetrafluoroethylene (PTFE) filter holder, Filter cassette, needed for some sampling systems.to hold the sample filter.
All sampling systems shall have PTFE screens ensure the filter does not contact any metal at any point in order to minimize the
potential of hexavalent chromium contamination.
7.1.2.1 Polytetrafluoroethylene (PTFE) filter cassette, all components of this cassette which contact the sample stream are
composed of PTFE. The inlet of the cassette is a mixing chamber to prevent particulate from accumulating only in the center of
the filter. The filter support screen is composed of PTFE. The outlet of the cassette has a large enough gas volume to ensure
consistent vacuum across the surface of the filter. The inlet and outlet halves of the cassette are joined with a threaded coupling
ring. The inlet and outlet stems are protected with vinyl caps. The filter is entirely encased within the cassette. During sampling,
the cassette inlet faces the ground, and a glass funnel is attached to the inlet to prevent any precipitation from being entrained in
the sample gas stream. See Fig. 2.
7.1.2.2 Federal Reference Method (FRM) filter cassette, this cassette is typically composed of a hard plastic such as high density
polyethylene (HDPE) or acetal homopolymer resin. The filter support screen is typically composed of stainless steel, although
aluminum screens are available. The use of a polyester drain disc between the filter and the metal support screen ensures that the
filter does not contact metal at any point in time.
7.1.2.3 Secondary cassette containers, to minimize contamination risks during shipment of cassettes. For PTFE cassettes, this
may consist of a 500 mL wide-mouth plastic bottle. For FRM cassettes, this may consist of an anti-static bag or a clear plastic
mailer, both designed to hold FRM cassettes. Tertiary containers, if used, may consist of plastic freezer bags.
7.1.3 Filters, 47 mm mm, 37 mm, or other suitably sized ashless, cellulose filters. These filters must be acid washed acid-washed
before use to remove any residual chromium. Filters which have been acid-washed by the manufacturer are commercially available
and are commonly referred to as “acid hardened cellulose” by the manufacturers.
NOTE 2—Finding filters that are free of Chromium can be challenging because of the low detections determined when following this method. Cellulose
filters can be acid washed and sodium bicarbonate treated in order to stabilize the hexavalent chromium following method specifications (4).
7.1.4 Sample pump:
7.1.4 Glass funnel assembly, funnel, for use with the PTFE filter holders usedcassettes. (See Fig. 2with Sampling System Type
A. These funnels are used to protect the filters from precipitation. .)
7.1.5 Flow control device, capable of controlling and measuring selected volumetric sample gas flow rates to within 62 %.
Rotameter control devices shouldmust be calibrated against a primary standard (that is, a flowmeter whose accuracy is traceable
to a primary standard. (Seestandard, see Practice D3195 and Section 10.)
7.1.6 Elapsed timer, to be placed in line with the sample pump to assist in detection of electrical interruptions that could occur
over the 24 hour time interval.determine the total amount of time the sampler was in operation.
7.1.7 Freezer, for storage of filters before and after sampling. Freezer temperatures must be maintained below -18°C.0°C at all
times. A field freezer is not required, but highly recommended. The laboratory must have a freezer. All freezers must have a means
of monitoring the freezer temperature 7 days per week, such as a max/min thermometer. If a max/min thermometer is used, it must
be NIST-traceable and within its calibration expiration date.
7.1.8 Ice cooler, Cooler and cold packs (or dry ice), for transport of filters to and from the sampling site. Cold packs may be
any form which can maintain a temperature of <0°C for the duration of time that the filters are contained in the cooler.
7.1.9 Flexible tubing, for use with flow calibration (see sampling system (10.1.47.1).
FIG. 1 Hexavalent Chromium Sampling System in Ambient Air
D7614 − 20
FIG. 32 Ion Chromatography Determination Diagram of Hexavalent Chromium PTFE Filter Cassette Assembly
7.1.10 Calibration system, soap bubble, rotameter, or mass flow calibration system Calibration system, soap bubble, rotameter
or mass flow calibration system to calibrate flow meters (see (10.1).
7.2 Analytical System: system, ion chromatograph, with the following components:
NOTE 2—A wide variety of instrumentation is commercially available. Follow the instrument manufacturer’s guidelines for allowable instrument
component operating pressures and other instrument-specific operational requirements.
7.2.1 Pump, capable of delivering a constant flow of eluent at the rate recommended by the manufacturer.
7.2.2 Guard column, placed before the separatory column to remove particulate and organic contaminants from samples.
7.2.3 Separatory column, packed with a high capacity, high efficiency, hydrophobic, anion exchange resin capable of separating
hexavalent chromium from other metallic cations.
7.2.4 Post-column reagent module, capable of delivering a constant flow of post-column reagent at one third of the rate of the
eluent flow rate. The module may consist of a pneumatic delivery system or a pump, as long as the ratio of post-column reagent
flow to eluent flow is consistent and the eluent flow rate is three-times the post-column reagent flow rate. It is imperative that the
ratio of eluent to post-column reagent be 3:1 to ensure that the pH upon mixing of the two liquid streams is such that the
diphenylcarbazide can complex to the fullest extent possible during post-column derivatization.
7.2.5 Ion chromatographReaction coil, shall have the following components:capable of mixing two flowing liquid streams with
minimal band spreading.
7.2.1.1 Pump, capable of delivering a constant flow of in the range of 1 to 5 mL/min (millimetre/minute) at a pressure of 15
to 150 MPa.
7.2.1.2 Guard column, placed before the separator column to remove particulate contaminants and highly adsorptive compounds
from samples, prolonging analytical column life.
7.2.1.3 Separator column, packed with a high capacity, high efficiency, hydrophobic, anion exchange column
7.2.1.4 Post column reagent module, capable of delivering 0 to 2 mL/min of reagent against a backpressure of up to 40 kPa.
7.2.1.5 Reaction coil, capable of mixing two flowing streams with minimal band spreading.
7.2.1.6 UV/VIS detector, low volume, flow through visible absorbance detector with a nonmetallic 1-cm flow path. The detection
wavelength for hexavalent chromium is 530 nm.
7.2.1.7 Injection valve, to ensure compatibility with aqueous and reverse-phase eluents and protect sensitive samples from
metallic contamination. Sample loops of up to 1 mL will provide enhanced detection limits.
7.2.1.8 Autosampler (optional), to provide consistent operation with precise quality assurance.
D7614 − 20
7.2.1.9 Acquisition software, where the instrument is controlled and data are collected and processed using the instrument
chromatography software and computer.
7.2.6 UV/Vis detector, flow through cell with UV/Vis absorbance detector. The detection wavelength for hexavalent chromium
following this method is 530 or 540 nm, whichever has the greatest response for a 1 ng/mL standard on a given ion chromatography
system.
7.2.7 Injection valve, with non-metallic flow path composed of a largely inert material such as polyethylethylketone (PEEK).
7.2.8 Sample loop, minimum volume of 1 mL (1000 μL).
7.2.9 Autosampler with suitable autosampler vials and caps, to provide consistent operation with better precision of retention
times.
7.2.10 Acquisition software, as provided by instrument manufacturer.
7.3 Fully adjustable, air-displacement pipets, for small-volume dispensing of aqueous fluids of moderate viscosity and density.
Pipets should comply with ISO 648,Specification E1154 for laboratory glassware/pipettes.piston operated volumetric devices.
7.4 Glassware, Volumetric flasks, made of borosilicate glass, which should comply with ISO 3585 for100 mL, 200 mL, 1 L, and
2 L, which comply with Specification E288 borosilicate glass.and Specification E438.
7.4.1 Volumetric flasks, 100 mL, 200 mL, 1 L, and 2 L, which should comply with ISO 1042 for laboratory glassware/one-mark
volumetric flasks.
7.5 Analytical balance, to provide reliable performance andfor reagent preparation. Must have accurate readability to 0.0010.1
mg.
7.6 Centrifuge tubesExtraction vessels with caps, 14 mL disposable polystyrene round bottom tubes with snap caps for sample
preparation.disposable plastic (polystyrene, polypropylene, or polyethylene) tubes or cups with tightly fitting caps and a minimum
working volume of 10 mL. Vessels must have sufficient depth to ensure that filters are fully submerged in extraction solution during
extraction.
7.7 Petri dishes, 47 millimetre (mm) disposable plastic units to be used in the disposable plastic, for storage of the acid washed,
sodium bicarbonate coated acid-washed, sodium bicarbonate-impregnated filters.
7.8 Nitrogen purged Glove box (2), Impregnation area, one box to be use to prepare the filters before sampling. The second box
should be used to prepare the filters for analysis. The boxes should be sealed airtight with a double-layered closed gasket system
and contain suspended plastic coated screens to hold filters during preparation. The boxes should be purged with ultra-pure
nitrogen.with clean airshed and surfaces such as:
7.8.1 Option 1—Nitrogen purged glove boxes (2) one box to be used to prepare the filters before sampling, the second box to
be used to prepare the filters for post-sampling extraction and analysis. The boxes must be airtight with a double-layered closed
gasket system and contain suspended plastic-coated screens to hold filters during preparation. Purge the boxes with ultra-pure
nitrogen.
7.8.2 Option 2—Dedicated filter handling room, maintained largely dust free, equipped with HEPA filtered HVAC system and
HEPA filtered laminar flow hood. Similar to that described in EPA PM2.5 Guidance Document 2.12 (10).
7.8.3 Option 3—Any clean area in which filters are cleaned, impregnated, dried, and handled in such a manner as to pass the
10 % prescreening requirements described in 11.2.2.5.
7.9 Ultrasonicator, to be used for standard preparation.reagent preparation, standard preparation when standards are not
purchased commercially, and filter extraction if using extraction Option 1 (11.5.2.1).
7.9.1 (Optional) wrist action shaker, for filter extraction Option 2, operating at 385 oscillations/minute with a tilt of 7.5°.
7.9.2 (Optional) orbital shaker, for filter extraction Option 3, operating at a minimum of 200 RPM.
7.10 Disposable PVC gloves, for sample handling and prevention of sample contamination.contamination during sampling and
analysis. Nitrile and latex gloves react with the post-column DPC reagent to form a color that absorbs at 530 or 540 nm (6). Use
of gloves made from any material other than PVC must be carefully considered with regard to the likelihood of contamination of
the filter.
7.11 Forceps, Polytetrafluoroethylene (PTFE) or plastic, for handling filters. Clean forceps prior to each use with reagent water
or ethanol.
7.12 Thermometer or thermocouple, NIST-traceable, for measuring temperature of the localized environment before and after
sample extraction.
7.13 Extract storage containers, disposable plastic (polystyrene, polypropylene, or polyethylene) tubes with tightly fitting caps
and a minimum working volume of 10 mL.
7.14 Plastic syringe with Luer-Lok fitting and liquid sealed plunger, 10 mL, for filtering extracts after extraction.
7.15 Luer-Lok syringe filter, 0.2 μm pore size, for filtering extracts after extraction.
7.16 pH strips, full range, for checking pH of cleaned filters if following filter cleaning Option 1.
D7614 − 20
7.17 Drain Discs (only required if using FRM cassettes), polyester, chemically inert, binder free, for preventing filter from
contacting metal support screen if using FRM cassettes.
8. Reagents
8.1 Reagent grade chemicals shall be used in all tests. Unless otherwise indicated, it is intended that all reagents conform to the
specifications of the Committee on Analytical Reagents of the American Chemical Society where such specifications are available.
Other grades may be used, provided it is first ascertained that the reagent is of sufficiently high purity to permit its use without
lessening the accuracy of the determination.
8.2 Purity of Water—Unless otherwise indicated, references to water shall be understood to mean reagent water as defined by
Type I of Specification D1193.
8.3 Reagent Water, ASTM Type I, ≥18.2 MΩ.
8.4 Ammonium sulfate ((NH ) SO ), 99.999 % purity based on trace metals.
4 2 4
8.5 Ammonium hydroxide (NH OH), reagent grade, 28.0–30.0 % NH4, specific gravity 0.899 g/cm .
8.6 1,5-diphenylcarbazide (DPC), crystalline. When fully dissolved in methanol, no visible particulate should be present and
solution should be colorless.
8.7 Methanol (CH OH), HPLC grade, greater than 99.9 % purity, specific gravity 0.79 g/cm .
8.8 Sulfuric acid (H SO ), concentrated, specific gravity 1.84 g/cm .
2 4
8.9 Sodium bicarbonate (NaHCO ), 99.5+ % purity.
8.10 Potassium dichromate (K Cr O ), 99.99+ % purity based on trace metals, crystalline. For use in preparing standards. Dry
2 2 7
at 105°C for 1 hour then cool in a desiccator prior to use. Alternatively, potassium chromate (K Cr O ) may be used (see Note
2 2 4
3).
8.11 Sodium Bicarbonate Extraction Solution (20 mM), in a 2 L volumetric flask, dissolve 3.36 g of sodium bicarbonate (8.9)
in reagent water (8.3). Dilute to volume with reagent water and mix thoroughly.
8.12 Eluent, 250 mM ammonium sulfate (8.4)/100 mM ammonium hydroxide (8.5)—In a 2 L volumetric flask, dissolve 66 g
of ammonium sulfate in approximately 1500 mL reagent water (8.3). Add 13 mL of ammonium hydroxide. Dilute to volume with
reagent water and mix thoroughly. Other eluents or eluent concentrations are acceptable if all quality control samples meet their
acceptance criteria.
8.13 Post-Column Derivatization Reagent, in a 500 mL volumetric flask, add approximately 300 mL of reagent water (8.3), then
carefully add 14 mL of concentrated sulfuric acid (8.8) and mix thoroughly. Allow to cool after mixing. In a 50 mL volumetric
flask, dissolve 0.25 g of 1,5-diphenylcarbazide (8.6) in 50 mL of HPLC-grade methanol (8.7). Sonication may be used to facilitate
DPC dissolution. Add DPC-methanol solution to sulfuric acid solution. Dilute to 500 mL with reagent water and mix thoroughly.
Depending on laboratory conditions, this reagent may be stable for up to seven days. It is suspected that exposure to UV light can
increase the rate of degradation of this reagent. Do not use if the reagent is darker than dark honey in color.
8.14 For the analysis of low level hexavalent chromium, only use reagents of the highest recognized analytical grade and water
as specified in (see 8.1.1).Standard Solutions:
8.1.1 Water, complying with the requirements of ASTM Type I deionized water (DI water) as specified in Specification D1193
(>18 MΩ-cm).
8.1.2 Ammonium sulfate (NH )SO , 99.999 % purity based on trace metals, specific gravity 1.77 (g/mL).
4 4
8.1.3 Ammonium hydroxide (NH )OH, reagent grade, 28.0–30.0 % NH basis, specific gravity 0.99 (g/cm ).
4 4
8.1.4 1,5-diphenylcarbazide (C N O), ACS crystalline (DPC).
13 4
8.1.5 Methanol (CH OH), HPLC grade, 0.2 micron filtered, greater than 99.9 % purity, 0.79 (g/cm ).
8.1.6 Sulfuric acid (H SO ), concentrated, 99.999 % purity based on trace metals, specific gravity 1.84 (g/mL).
2 4
8.1.7 Sodium bicarbonate (NaHCO ), greater than 99.5 % purity.
8.1.8 Potassium dichromate (K Cr O ), 99.99+ % purity based on trace metals, crystalline. Before use it should be dried at
2 2 7
105°C for 1 hour then cooled in a dessicator. See 8.1.12.1 for instructions on preparing standard solutions.
8.1.9 Sodium Bicarbonate Extraction Solution (20 mM), dissolve 3.36 grams (g) of sodium bicarbonate (see 8.1.7) in DI water
(see 8.1.1) in a 2.0 L volumetric flask. Mix well and dilute to mark. Stopper and mix thoroughly.
The last approved version ACS Reagent Chemicals, Specifications and Procedures for Reagents and Standard-Grade Reference Materials,of this historical standard is
referenced on www.astm.org. American Chemical Society, Washington, DC. For suggestions on the testing of reagents not listed by the American Chemical Society, see Analar
Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharmacopeial Convention, Inc.
(USPC), Rockville, MD.
The boldface numbers in parentheses refer to a list of references at the end of this standard.
D7614 − 20
8.1.10 Eluent Stock, 250 mM ammonium sulfate (see 8.1.2) and 100 mM ammonium hydroxide (see 8.1.3): dissolve 66 g of
ammonium sulfate in ~1000 mL DI water (see 8.1.1) in a 2 L volumetric flask. Add 7 mL of ammonium hydroxide and dilute to
volume with DI water. Stopper and mix thoroughly.
8.1.11 Post-Column Derivatizing Reagent (PCR), in a 50 mL volumetric flask, dissolve 0.25 gm of 1,5-diphenylcarbazide (see
8.1.4) in 50 mL of HPLC-grade methanol (see 8.1.5). Sonicate until DPC goes into solution. In a 500 mL volumentric flask add
approximately 300 mL of DI water (see 8.1.1). Carefully add 14 mL of 99.999 % sulfuric acid (see 8.1.6) to the DI water, allow
to cool after mixing. Add DPC-methanol solution to sulfuric acid solution. Dilute to 500 mL with DI water, stopper and mix
thoroughly. This reagent is stable for four or five days. To minimize waste, it should be prepared in 500 mL quantities as needed.
6+
8.14.1 Hexavalent Chromium Standard Solutions: Primary and Secondary Stock Standard Solutions (~1000 μg/mL Cr ), stock
hexavalent chromium standards are available commercially or can be prepared by dissolving 0.283 g of potassium dichromate
(8.10) in 20 mM sodium bicarbonate extraction solution (8.11) in a 100 mL volumetric flask. Dilute to volume with 20 mM sodium
bicarbonate extraction solution and mix thoroughly.
6+ 6+
8.14.1.1 Hexavalent Chromium Stock Standard Solution (~1000 μg/mL Cr ),—Prepare two separate 1000 μg/mL Cr stock
hexavalent chromium standards are available commercially or can be prepared by diluting 0.283 grams of potassium dichromate
(seesolutions or obtain two commercially prepared stock solutions from separate sources. When a second manufacturer is not
available, a primary stock solution from the same manufacturer, but from a different manufacturing lot, may be used. The primary
stock standard is used exclusively for 8.1.8) with DI water (seecalibration standards. The secondary stock 8.1.1) in a 100 mL
volumetric flask. Dilute to volume withstandard is used for all quality control standards and samples (Section 1320) mM
(11sodium ).bicarbonate extraction solution, stopper and mix thoroughly.
NOTE 3—Potassium chromate (K CrO ) canmay be used as an alternative to potassium dichromate for the preparation of the hexavalent chromium
2 4
stock standard solution. 0.373 g of K CrO are needed to make ~100 mL of ~1000 μg/mL Cr6.
2 4
NOTE 4—Two primary stock solutions should be prepared and/or obtained from separate sources. One is to be used exclusively for the calibration
standards and the other for the laboratory control samples (LCS) and calibration verification.
8.1.12.2 Hexavalent Chromium Working Standard Solution, to be prepared in the 20 mM sodium bicarbonate extraction
solution. The working standard solution is at 1000 ng/mL. Dilute 100 μL of the stock standard solution (see 8.1.12.1) with
extraction solution (see 8.1.9) in a 100 mL volumetric flask. Dilute to volume, stopper and mix thoroughly. The working standard
solution is stable for up to one month.
8.1.12.3 Hexavalent Chromium Calibration Solutions, to be prepared in the 20 mM sodium bicarbonate extraction solution.
Standards are prepared in 0.05, 0.1, 0.2, 0.5, 1.0 and 2.0 ng/L concentrations by diluting appropriate volumes of the 1000 ng/mL
working standard solution (see 8.1.12.2) with the 20 mM sodium bicarbonate extraction solution (see 8.1.9). The calibration
standards are prepared after the working stock standard is prepared. They are stable for one month and should be analyzed every
day samples are prepared.
8.14.2 Hexavalent Chromium Intermediate Primary Standard Solution, 1000 ng/mL, to be prepared in 20 mM sodium
bicarbonate extraction solution. In a 100 mL volumetric flask, add approximately 75 mL extraction solution (8.11) and 100 μL of
the primary source stock standard solution (8.14.1). Dilute to volume with 20 mM sodium bicarbonate solution and mix
thoroughly. Depending on the laboratory environment, the intermediate primary standard solution can be stable for up to twelve
months.
8.14.3 Hexavalent Chromium Calibration Solutions, to be prepared in 20 mM sodium bicarbonate extraction solution. A
minimum of six standards are prepared to span the range of 0.04 to 2.0 ng/mL by diluting appropriate volumes of the 1000 ng/mL
intermediate standard solution (8.14.2) with 20 mM sodium bicarbonate extraction solution (8.11). Depending on the laboratory
environment, the calibration standard solutions can be stable for up to twelve months.
NOTE 4—Calibration standard concentrations below 0.04 ng/mL may be used if the laboratory’s analytical system has a lower detection limit than that
determined in the 2019 multi-laboratory study (1). For NELAC accredited laboratories, the lowest calibration standard must be at the lowest concentration
reported to the client without qualifiers (11).
8.14.4 Hexavalent Chromium Intermediate Secondary Standard Solution, 1000 ng/mL, to be prepared in 20 mM sodium
bicarbonate extraction solution. In a 100 mL volumetric flask, add approximately 75 mL extraction solution (8.11) and 100 μL of
the secondary source stock standard solution (8.14.1) pending on the laboratory environment, the working intermediate standard
solution can be stable for up to twelve months.
8.14.5 Nitric Acid (HNOHexavalent Chromium Initial ), and Continuing Calibration Verification Standard Solution,
concentrated, 70 % redistilled, 99.999+ % purity based concentration at or below the mid-point of the calibration curve, to be
prepared in 20 mM sodium bicarbonate extraction solution. In a 100 mL volumetric flask, add approximately 75 mL extraction
solution (8.11) and the appropriate amount of the intermediate secondary source standard solution (8.14.4on trace metals, specific
gravity 1.4 (g/mL).). Dilute to volume with 20 mM sodium bicarbonate solution and mix thoroughly. Depending on laboratory
environment, the secondary working standard solution can be stable for up to twelve months.
8.1.13.1 Acid Bath Solution (10 % Nitric Acid), add 50 mL 99.999+ % nitric acid (see 8.1.13) to approximately 500 mL DI water
(see 8.1.1) in a volumetric flask. Dilute to mark with DI water, stopper and mix well.
3+
8.14.6 Trivalent Chromium Stock Standard Solution (~1000 μg/mL Cr ), stock trivalent chromium standards are available
6+
commercially. The standard must be verified to contain negligible amounts of Cr prior to use for this method.
D7614 − 20
8.14.7 Sodium Bicarbonate Impregnating Solution (1.2 M), Trivalent Chromium Working Standard Solution, dissolve 5 g of
sodium bicarbonate (see 1000 ng/mL, to be prepared in reagent water. In a 100 mL volumetric flask, add approximately 75 mL
reagent water (8.1.78.3) in DI water (seeand 100 μL of 8.1.1) in a 500 L volumetric flask.the trivalent chromium stock standard
solution (8.14.6 Mix well and dilute to mark. Stopper ). Dilute to volume with reagent water and mix thoroughly. Depending on
the laboratory environment, the working standard solution can be stable for up to twelve months. A small amount of concentrated
3+ 6+
nitric acid (<1% volume-to-volume) may be added to prevent conversion of Cr to Cr .
8.15 Nitric Acid (HNO ), concentrated, trace metals grade, specific gravity 1.4 (g/cm ).
8.16 Acid Bath Solution (10 % Nitric Acid), in a 1 L volumetric flask, add approximately 500 mL reagent water (8.3) and 50
mL concentrated nitric acid (8.15), in that order. Dilute to volume with reagent water and mix well. This solution is only required
if using filter cleaning protocol Option 1 (11.2.1.1).
8.17 Sodium Bicarbonate Impregnation Solution (0.12M), in a 500 mL volumetric flask, dissolve 5.0 g of sodium bicarbonate
(8.9) in reagent water (8.3). Sonication may be used to facilitate dissolution. Dilute to volume with reagent water and mix
thoroughly.
9. Sampling
9.1 Samples are collected using an individual filter apparatusCollect samples using a pre-prepared filter (11.2.2) contained in
a cassette (7.1.2) suitable for the Sampling System being employed. (See 11.1 through 11.4.) Wear gloves (7.10and flow control
device.) when handling the filter cassettes, both pre- and post-sample collection.
9.2 A flow control device(s) is used For each sample, use a flow control device to maintain a relatively constant sample flow
rate through each sample filter over a specific sampling period. of 5–16 L/min over 24 hours. The flow device can be a mass flow
controller or a rotameter. A nominal flow rate of 9.0 to 16.0 L/min is applied for sample collection.
9.3 During operation, the control device is programmed to activate and deactivate the components of the sample collection
system, consistent with the beginning and end of the sample collection period. The Ensure the connecting lines between the filter
assembly cassette (7.1.2) and the sampling pump should(7.1.1 be kept ), if any, are as short as possible to minimize the system
residence time. If a rotameter is used, it should be calibrated in the field to determine true readings (see 10.1).
9.4 Prior to use, calibrate all sampling system components described in Section 10, “Calibration and Standardization.” If a
rotameter is used, calibrate it in the field to determine true readings (10.1).
9.5 The following steps are provided for operation of a typical collectionsampling system while collecting a samplesample:
9.5.1 Set the sampling system to the desired sample collection flow rate(s) (that is, referencing the corresponding ambient
calibration curve(s) and considering the desired total volume of ambient air to be sampled and the sampling period for each
sampling event).Assemble the sampling system.
9.5.2 If performing field loading of filters into cassettes (7.1.2), wear gloves (7.10) and use PTFE or plastic forceps (8.11) when
removing filters from petri dishes and placing them into filter cassettes. Load the filters as described in 11.3.
9.5.3 Document all required site information on the field data sheet. Include, at a minimum, the following: site location,
operator, filter set-up date, scheduled collection date, initial rotameter or mass flow controller reading, programmed start and end
times, and any additional comments deemed necessary.
9.5.4 Sample Collection Using PTFE Cassettes With(7.1.2.1 disposable gloves, attach the prepared filter assembly to the inlet
) (see Fig. 2) of the probe or place the prepared filter into the filter holder, depending on the type of sampler.):
NOTE 5—One filter to collect a single sample, two filters for collocated samples.
9.5.4.1 Remove the caps from the inlet and outlet of the pre-loaded filter cassette. Attach the funnel stem (7.1.4) to the inlet
fitting of the cassette with tubing and tighten securely.
9.5.4.2 Connect the outlet of the cassette (7.1.2) and funnel assembly to the PTFE tube (7.1.9) that connects to the sample pump
(7.1.1). If a collocated sample is scheduled, connect the outlet of the collocated cassette to the collocated PTFE connecting tube.
(See Note 17.)
9.5.4.3 Program the sampler to initiate flow through the entire sampling system. Adjust the sample collection flow rate to the
intended flow rate to be used during sampling.
9.5.4.4 Allow the flow to continue for approximately two minutes while the system attains operating temperature. After two
minutes, verify that the flow rate matches the intended flow rate from 9.5.4.3; re-adjust if necessary. Document the flow rate on
the field data sheet.
9.5.4.5 Program the sampler to collect a 24-hour sample on the designated sample date.
9.5.5 Sample Collection Using FRM Cassettes (7.1.2.2):
9.5.5.1 Equip the sampling system with a pre-loaded FRM cassette per the Manufacturer’s guidelines for the sampling system
being employed.
9.5.5.2 Program the sampler to initiate flow through the entire sampling system. Adjust the sample collection flow rate to the
intended flow rate to be used during sampling.
D7614 − 20
9.5.5.3 Allow the flow to continue for approximately two minutes while the system attains operating temperature. After two
minutes, verify that the flow rate matches the intended flow rate from 9.5.5.2; re-adjust if necessary. Document the flow rate on
the field data sheet.
9.5.5.4 Program the sampler to collect a 24-hour sample on the designated sample date.
9.5.6 Record the start and end time of the collection event and the corresponding flow rate onto the sampling field data sheet
and calculate an average flow rate.Sample Recovery:
9.5.6.1 Wear gloves (7.10) to remove the filter cassette (7.1.2.2) from the sampler. Retrieve the sam
...



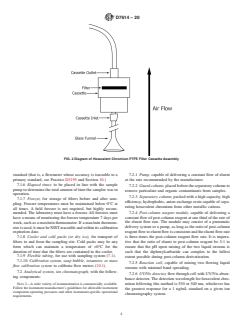




Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...