ISO/TS 19218-1:2011/Amd 1:2013
(Amendment)Medical devices — Hierarchical coding structure for adverse events — Part 1: Event-type codes — Amendment 1
Medical devices — Hierarchical coding structure for adverse events — Part 1: Event-type codes — Amendment 1
Dispositifs médicaux — Structure de codage pour la cause et le type d'événement défavorable — Partie 1: Codes de type d'événement — Amendement 1
General Information
- Status
- Withdrawn
- Publication Date
- 07-Jan-2013
- Withdrawal Date
- 07-Jan-2013
- Current Stage
- 9599 - Withdrawal of International Standard
- Start Date
- 02-Oct-2020
- Completion Date
- 12-Feb-2026
Relations
- Effective Date
- 04-Dec-2021
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ISO/TS 19218-1:2011/Amd 1:2013 is a technical specification published by the International Organization for Standardization (ISO). Its full title is "Medical devices — Hierarchical coding structure for adverse events — Part 1: Event-type codes — Amendment 1". This standard covers: Medical devices — Hierarchical coding structure for adverse events — Part 1: Event-type codes — Amendment 1
Medical devices — Hierarchical coding structure for adverse events — Part 1: Event-type codes — Amendment 1
ISO/TS 19218-1:2011/Amd 1:2013 is classified under the following ICS (International Classification for Standards) categories: 03.120.10 - Quality management and quality assurance; 11.040.01 - Medical equipment in general. The ICS classification helps identify the subject area and facilitates finding related standards.
ISO/TS 19218-1:2011/Amd 1:2013 has the following relationships with other standards: It is inter standard links to ISO/TS 19218-1:2011. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ISO/TS 19218-1:2011/Amd 1:2013 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
TECHNICAL ISO/TS
SPECIFICATION 19218-1
First edition
2011-05-15
AMENDMENT 1
2013-01-15
Medical devices — Hierarchical coding
structure for adverse events —
Part 1:
Event-type codes
AMENDMENT 1
Dispositifs médicaux — Structure de codage pour la cause et le type
d’événement défavorable —
Partie 1: Codes de type d’événement
AMENDEMENT 1
Reference number
ISO/TS 19218-1:2011/Amd.1:2013(E)
©
ISO 2013
ISO/TS 19218-1:2011/Amd.1:2013(E)
Foreword
ISO (the International Organization for Standardization) is a worldwide federation of national standards bodies
(ISO member bodies). The work of preparing International Standards is normally carried out through ISO
technical committees. Each member body interested in a subject for which a technical committee has been
established has the right to be represented on that committee. International organizations, governmental and
non-governmental, in liaison with ISO, also take part in the work. ISO collaborates closely with the International
Electrotechnical Commission (IEC) on all matters of electrotechnical standardization.
International Standards are drafted in accordance with the rules given in the ISO/IEC Directives, Part 2.
The main task of technical committees is to prepare International Standards. Draft International Standards
adopted by the technical committees are circulated to the member bodies for voting. Publication as an
International Standard requires approval by at least 75 % of the member bodies casting a vote.
In other circumstances, particularly when there is an urgent market requirement for such documents, a technical
committee may decide to publish other types of document:
— an ISO Publicly Available Specification (ISO/PAS) represents an agreement between technical experts in
an ISO working group and is accepted for publication if it is approved by more than 50 % of the members
of the parent committee casting a vote;
— an ISO Technical Specification (ISO/TS) represents an agreement between the members of a technical
committee and is accepted for publication if it is approved by 2/3 of the members of the committee
casting a vote.
An ISO/PAS or ISO/TS is reviewed after three years in order to decide whether it will be confirmed for a further
three years, revised to become an International Standard, or withdrawn. If the ISO/PAS or ISO/TS is confirmed,
it is reviewed again after a further three years, at which time it must either be transformed into an International
Standard or be withdrawn.
Attention is drawn to the possibility that some of the elements of this document may be the subject of patent
rights. ISO shall not be held responsible for identifying any or all such patent rights.
Amendment 1 to ISO/TS 19218-1:2011 was prepared by Technical Committee ISO/TC 210, Quality management
and corresponding general aspects for medical devices.
ii © ISO 2013 – All rights reserved
ISO/TS 19218-1:2011/Amd.1:2013(E)
Medical devices — Hierarchical coding structure for adverse
events —
Part 1:
Event-type codes
AMENDMENT 1
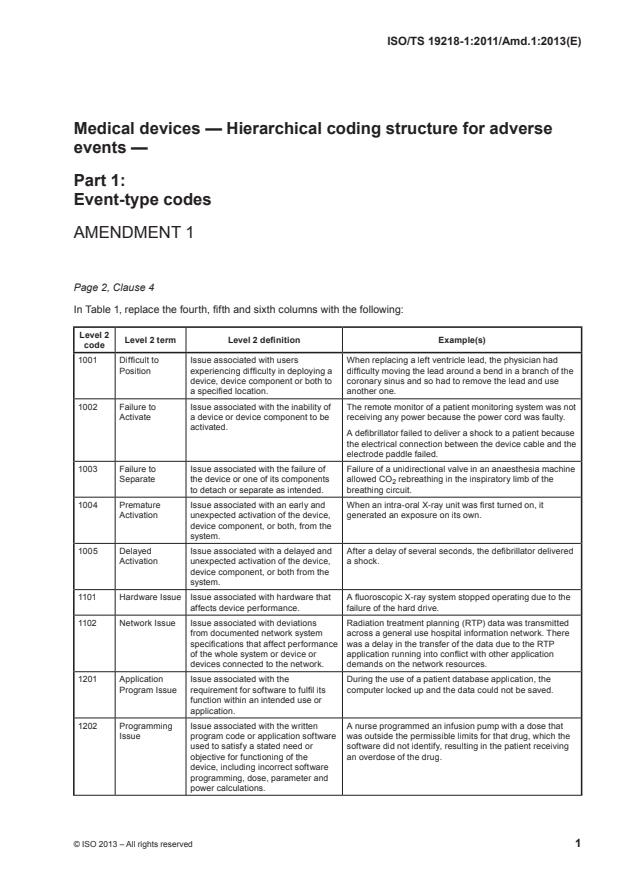
Page 2, Clause 4
In Table 1, replace the fourth, fifth and sixth columns with the following:
Level 2
Level 2 term Level 2 definition Example(s)
code
1001 Difficult to Issue associated with users When replacing a left ventricle lead, the physician had
Position experiencing difficulty in deploying a difficulty moving the lead around a bend in a branch of the
device, device component or both to coronary sinus and so had to remove the lead and use
a specified location. another one.
1002 Failure to Issue associated with the inability of The remote monitor of a patient monitoring system was not
Activate a device or device component to be receiving any power because the power cord was faulty.
activated.
A defibrillator failed to deliver a shock to a patient because
the electrical connection between the device cable and the
electrode paddle failed.
1003 Failure to Issue associated with the failure of Failure of a unidirectional valve in an anaesthesia machine
Separate the device or one of its components allowed CO rebreathing in the inspiratory limb of the
to detach or separate as intended. breathing circuit.
1004 Premature Issue associated with an early and When an intra-oral X-ray unit was first turned on, it
Activation unexpected activation of the device, generated an exposure on its own.
device component, or both, from the
system.
1005 Delayed Issue associated with a delayed and After a delay of several seconds, the defibrillator delivered
Activation unexpected activation of the device, a shock.
device component, or both from the
system.
1101 Hardware Issue Issue associated with hardware that A fluoroscopic X-ray system stopped operating due to the
affects device performance. failure of the hard drive.
1102 Network Issue Issue associated with deviations Radiation treatment planning (RTP) data was transmitted
from documented network system across a general use hospital information network. There
specifications that affect performance was a delay in the transfer of the data due to the RTP
of the whole system or device or application running into conflict with other application
devices connected to the network. demands on the network resources.
1201 Application Issue associated with the During the use of a patient database application, the
Program Issue requirement for software to fulfil its computer locked up and the data could not be saved.
function within an intended use or
application.
1202 Programming Issue associated with the written A nurse programmed an infusion pump with a dose that
Issue program code or application software was outside the permissible limits for that drug, which the
used to satisfy a stated need or software did not identify, resulting in the patient receiving
objective for functioning of the an overdose of the drug.
device, including incorrect software
programming, dose, parameter and
power calculations.
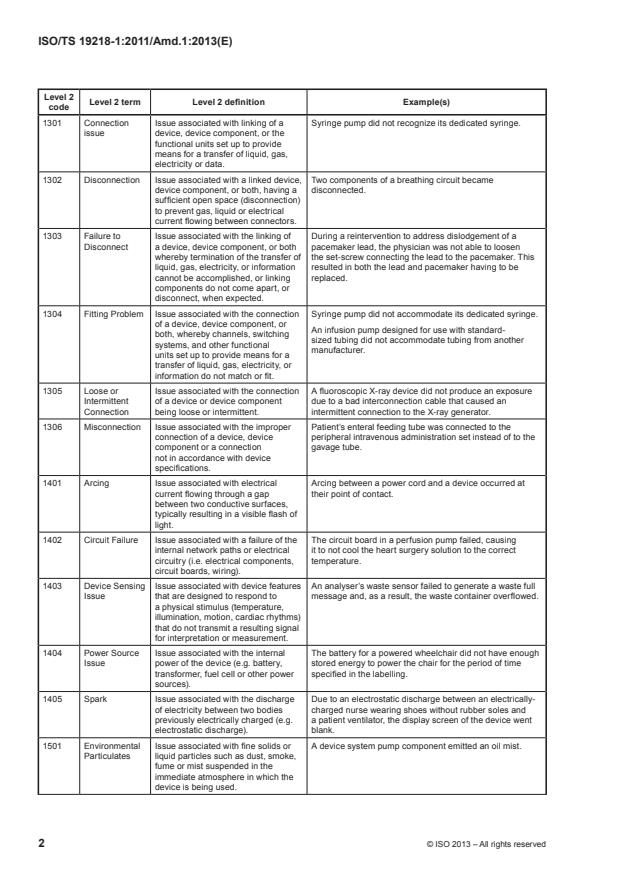
ISO/TS 19218-1:2011/Amd.1:2013(E)
Level 2
Level 2 term Level 2 definition Example(s)
code
1301 Connection Issue associated with linking of a Syringe pump did not recognize its dedicated syringe.
issue device, device component, or the
functional units set up to provide
means for a transfer of liquid, gas,
electricity or data.
1302 Disconnection Issue associated with a linked device, Two components of a breathing circuit became
device component, or both, having a disconnected.
sufficient open space (disconnection)
to prevent gas, liquid or electrical
current flowing between connectors.
1303 Failure to Issue associated with the linking of During a reintervention to address dislodgement of a
Disconnect a device, device component, or both pacemaker lead, the physician was not able to loosen
whereby termination of the transfer of the set-screw connecting the lead to the pacemaker. This
liquid, gas, electricity, or information resulted in both the lead and pacemaker having to be
cannot be accomplished, or linking replaced.
components do not come apart, or
disconnect, when expected.
1304 Fitting Problem Issue associated with the connection Syringe pump did not accommodate its dedicated syringe.
of a device, device component, or
An infusion pump designed for use with standard-
both, whereby channels, switching
sized tubing did not accommodate tubing from another
systems, and other functional
manufacturer.
units set up to provide means for a
transfer of liquid, gas, electricity, or
information do not match or fit.
1305 Loose or Issue associated with the connection A fluoroscopic X-ray device did not produce an exposure
Intermittent of a device or device component due to a bad interconnection cable that caused an
Connection being loose or intermittent. intermittent connection to the X-ray generator.
1306 Misconnection Issue associated with the improper Patient’s enteral feeding tube was connected to the
connection of a device, device peripheral intravenous administration set instead of to the
component or a connection gavage tube.
not in accordance with device
specifications.
1401 Arcing Issue associated with electrical Arcing between a power cord and a device occurred at
current flowing through a gap their point of contact.
between two conductive surfaces,
typically resulting in a visible flash of
light.
1402 Circuit Failure Issue associated with a failure of the The circuit board in a perfusion pump failed, causing
internal network paths or electrical it to not cool the heart surgery solution to the correct
circuitry (i.e. electrical components, temperature.
circuit boards, wiring).
1403 Device Sensing Issue associated with device features An analyser’s waste sensor failed to generate a waste full
Issue that are designed to respond to message and, as a result, the waste container overflowed.
a physical stimulus (temperature,
illumination, motion, cardiac rhythms)
that do not transmit a resulting signal
for interpretation or measurement.
1404 Power Source Issue associated with the internal The battery for a powered wheelchair did not have enough
Issue power of the device (e.g. battery, stored energy to power the chair for the period of time
transformer, fuel cell or other power specified in the labelling.
sources).
1405 Spark Issue associated with the discharge Due to an electrostatic discharge between an electrically-
of electricity between two bodies charged nurse wearing shoes without rubber soles and
previously electrically charged (e.g. a patient ventilator, the display screen of the device went
electrostatic discharge). blank.
1501 Environmental Issue associated with fine solids or A device system pump component emitted an oil mist.
Particulates liquid particles such as dust, smoke,
fume or mist suspended in the
immediate atmosphere in which the
device is being used.
2 © ISO 2013 – All rights reserved
ISO/TS 19218-1:2011/Amd.1:2013(E)
Level 2
Level 2 term Level 2 definition Example(s)
code
1502 Fumes or Issue associated with the visibility, Due to inadequate room ventilation, an abnormally high
Vapours odour or toxicity of an ambient vapour concentration of carbon dioxide in the room caused an IVD
or gas which affects the operation of autoanalyser being used to measure blood carbon dioxide
the device. levels to generate erroneous test results.
1503 Inadequate Issue associated with inadequate or As a result of the user storing the test strips in a plastic
Storage inappropriate storage of the device. bag instead of the original container, the glucose monitor
reported erroneous readings that resulted in unnecessary
treatment.
1504 Loss of Power Issue associated with the failure A patient was being transported by helicopter. The intra-
of primary power provided by the aortic balloon pump was plugged into a power inverter that
facility, e.g. electrical, gas, fluid failed, which resulted in loss of power to the balloon pump.
pressure.
1601 Migration Issue associated with an undesired After a stenting procedure was completed, it was
of Device movement of a device, device determined that the stent migrated and no longer
or Device component, or both, related to its completely covered the lesion.
Component movement away from or dislodging
from a source.
1602 Osseo- Issue association with Due to loosening of the connection between the hip
disintegration interconnection between bone and an implant and the femur, the patient required revision to
Issue implanted device. address persistent pain.
1701 Component Issue associated with the When the bulb in a phototherapy lamp burned out, the
or Accessory incompatibility of any device, neonatal intensive care unit nurse replaced it with a bulb
Incompatibility device component, or both, while that did not meet the manufacturer’s specifications. The
being operated in the same use lamp overheated and burned the baby’s skin.
environment thereby leading to a
dysfunction between the device and
its components.
1702 Device-Device Issue associated with the Users of a newly distributed enhanced algorithm found the
Incompatibility incompatibility of two or more devices algorithm was incompatible with the electrocardiograph’s
while being operated in the same operating software, resulting in operational errors.
use environment thereby leading to a
dysfunction of more than one device.
1703 Patient-Device Issue associated with the interaction During a procedure to replace a right ventricular lead,
Incompatibility between the patient’s physiology or the placement was not successful due to the size of the
anatomy and the device that affects patient’s vein.
patient or device (e.g. biocompatibility
or immunological issues).
1801 Deflation Issue Issue associated with the inability of After the balloon of a percutaneous transluminal
a device, device component, or both, angioplasty (PTA) balloon dilatation catheter was inflated,
to release its contents. it could not be deflated without surgical intervention.
1802 Improper Flow Issue associated with the An infusion pump delivered a larger volume of drug than
or Infusion unsubstantiated regulation and programmed to deliver.
delivery of therapy, e.g. air, gas,
The total parenteral nutrition solution was improperly
drugs or fluids into a device or a
mixed and, when the bag was connected for infusion,
patient under positive pressure that is
the pump was unable to deliver the solution because it
being generated by a pump.
clogged the tubing.
1803 Inflation Issue Issue associated with the inability of a During a blood pressure reading, the limb cuff continued to
device, device component, or both, to inflate to a level beyond normal practice.
expand or enlarge with the intended
inflation agent (e.g. saline or air).
1804 No Flow Issue arising from the device failing A ventilator alarmed due to a valve stuck in a closed
to deliver the specified liquid or gas. condition blocking flow of oxygen to the patient.
1805 Excessive Flow Issue associated with an overdose The infusion pump operator inadvertently entered an
or Overinfusion of delivery therapy, such as drugs or inappropriately high value for the volume of drug to be
fluids being delivered into a device or infused.
a patient under positive pressure.
ISO/TS 19218-1:2011/Amd.1:2013(E)
Level 2
Level 2 term Level 2 definition Example(s)
code
1806 Insufficient Issue associated with an underdose During a phacofragmentation procedure, the viscous gas
Flo
...


Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...